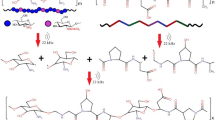
Abstract
Graft copolymers of collagen and polymethyl methacrylate were synthesized in a suspension of an acetic acid dispersion of fish collagen and methyl methacrylate in the presence of tributylborane. The copolymers were characterized by IR spectroscopy, gel permeation chromatography, and differential scanning calorimetry. The glass transition temperature is determined for the obtained copolymers. Changes in the synthesis temperature in a range of 25–60 °C did not significantly affect the fraction and molecular weight of the graft polymethyl methacrylate. Microphotographs obtained by scanning electron microscopy and atomic force microscopy indicate the destruction of the fibrillous structure of collagen in the copolymer at the synthesis temperature higher than 25 °C. The hydrophilicity and light transmission of films of the synthesized copolymer samples were studied.
Similar content being viewed by others
References
J. Furukawa, T. Tsuruta, S. Inoue, J. Polym. Sci., Part A: Polym. Chem., 1967, 26, 234; DOI: https://doi.org/10.1002/pol.1957.1202611312.
G. A. Kolesnikov, L. S. Fedorova, Bull. Acad. Sci. USSR, Div. Chem. Sci., 1957, 2, 251; DOI: https://doi.org/10.1007/BF01170563.
M. Yu. Zaremski, E. S. Garina, M. E. Gurskii, Yu. N. Bubnovb, Polym. Sci., Ser. B, 2013, 55, 304; DOI: https://doi.org/10.1134/S1560090413050072.
M. Yu. Zaremskii, V. V. Odintsova, A. V. Plutalova, M. E. Gurskii, Yu. N. Bubnov, Polym. Sci., Ser. B, 2018, 60, 162; DOI: https://doi.org/10.1134/S1560090418020082.
Ch. Lv, Yu. Du, X. Pan, J. Polym. Sci., Part A: Polym. Chem., 2020, 58, 14; DOI: https://doi.org/10.1002/pola.29477.
R. L. Timmins, O. R. Wilson, A. J. D. Magenau, J. Polym. Sci., 2020, 58, 1463; DOI: https://doi.org/10.1002/pol.20200089.
D. V. Ludin, Yu. L. Kuznetsova, S. D. Zaitsev, Polym. Sci., Ser. B, 2016, 58, 503; DOI: https://doi.org/10.1134/S1560090416050079.
N. L. Pegeev, L. L. Semenycheva, N. B. Valetova, Y. O. Matkivskaya, T. I. Liogon’kaya, A. V. Mitin, Y. A. Kurskii, J. Organomet. Chem., 2020, 922, 121396; DOI: https://doi.org/10.1016/j.jorganchem.2020.121396.
M. F. Sonnenschein, S. P. Webb, P. E. Kastl, D. J. Arriola, B. M. Wendt, D. R. Harrington, N. G. Rondan, Macromolecules, 2004, 37, 7974; DOI: https://doi.org/10.1021/ma040095f.
S. Liu, Z. Zheng, M. Li, X. Wang, J. Appl. Polym. Sci., 2012, 125, 3335; DOI: https://doi.org/10.1002/app.34232.
T. C. Chung, W. Janvikul, H. L. Lu, J. Am. Chem. Soc., 1996, 118, 705; DOI: https://doi.org/10.1021/ja9527737.
D. F. Grishin, L. L. Semyonycheva, Russ. Chem. Rev., 2001, 70, 425; DOI: https://doi.org/10.1070/RC2001v070n05ABEH000635.
V. A. Dodonov, Yu. L. Kuznetsova, A. A. Skatova, Polym. Sci., Ser. B, 1999, 41, 261.
V. A. Dodonov, A. A. Skatova, Yu. L. Kuznetsova, I. I. Brezhneva, Polym. Sci., Ser. A, 2000, 42, 967.
V. A. Dodonov, T. I. Starostina, V. A. Kuropatov, Yu. B. Malysheva, Yu. L. Kuznetsova, A. S. Buzina, Russ. J. Appl. Chem., 2017, 90, 77; DOI: https://doi.org/10.1134/s1070427217010128.
S. Fujisawa, Y. Kadoma, Polymers, 2010, 2, 575; DOI: https://doi.org/10.3390/polym2040575.
D. V. Ludin, S. D. Zaitsev, Yu. L. Kuznetsova, A. V. Markin, A. E. Mochalova, E. V. Salomatina, J. Polym. Res., 2017, 24, 117; DOI: https://doi.org/10.1007/s10965-017-1280-x.
Yu. L. Kuznetsova, E. A. Morozova, A. S. Vavilova, A. V. Markin, O. N. Smirnova, N. S. Zakharycheva, D. V. Lyakaev, L. L. Semenycheva, Polym. Sci. D, 2020, 13, 453; DOI: https://doi.org/10.31044/1994-6260-2020-0-1-22-29.
Yu. L. Kuznetsova, K. S. Sustaeva, A. S. Vavilova, A. V. Markin, D. V. Lyakaev, A. V. Mitin, L. L. Semenycheva, J. Organomet. Chem., 2020, Article Number: 121431, DOI: https://doi.org/10.1016/j.jorganchem.2020.121431.
M. A. Uromicheva, Yu. L. Kuznetsova, N. B. Valetova, A. V. Mitin, L. L. Semenycheva, O. N. Smirnova, Proceedings of Universities. Applied Chemistry and Biotechnology, 2021, 11, 16; DOI: https://doi.org/10.21285/2227-2925-2021-11-1-16-25.
H. Okamura, A. Sudo, T. Endo, J. Polym. Sci., Part A: Polym. Chem., 2009, 47, 6163; DOI: https://doi.org/10.1002/pola.23659.
B. M. Mikhailov, Yu. N. Bubnov, Boroorganicheskie soedineniya v organicheskom sinteze [Organoboron Compounds in Organic Synthesis], Moscow, Nauka, 1977, 515 pp. (in Russian).
V. M. Oliveira, C. R. D. Assis, B. A. M. Costa, R. C. A. Neri, F. T. Monte, H. M. S. C. V. Freitas, R. C. P. França, J. Santos, R. S. Bezerra, A. L. F. Porto, J. Molecular Structure, 2021, 1224, 129023; DOI: https://doi.org/10.1016/j.molstruc.2020.129023.
W. L. F. Armarego, C. C. L. Chai, Purification Laboratory Chemicals, Elsevier, Oxford, 2012, 1024.
L. L. Semenycheva, M. V. Astanina, J. L. Kuznetsova, N. B. Valetova, E. V. Geras’kina, O. A. Tarankova, Pat. RF, 2015, 31, 1.
D. Ludin, Yu. Voitovich, E. Salomatina, Yu. Kuznetsova, I. Grishin, I. Fedushkin, S. Zaitsev, Macromol. Res., 2020; DOI: https://doi.org/10.1007/s13233-020-8111-3.
K. Mahmood, L. Muhammad, F. Ariffin, H. Kamilah, A. Razak, S. Sulaiman, Food Science and Quality Management, 2016, 56, 47; DOI: Journals/index.php/FSQM/article/view/33411/34351.
R. Schrieber, H. Gareis, Gelatin Handbook. Theory and Industrial Practice, Wiley-VCH Verlag GmbH&Co. KGaA, Weinheim, Germany, 2007.
F. Subhan, M. Ikram, A. Shehzad, A. Ghafoor, J. Food Sci. Technol., 2015, 528, 4703; DOI: https://doi.org/10.1007/s13197-014-1652-8.
L. L. Semenycheva, Yu. O. Matkivskaia, N. B. Valetova, Yu. O. Chasova, N. L. Pegeev, A. L. Eloyan, A. A. Moikin, Yu. A. Kursky, Russ. Chem. Bull., 2017, 66, 1660; DOI: https://doi.org/10.1007/s11172-017-1938-x.
F. Casanova, M. A. Mohammadifar, M. Jahromi, H. O. Petersen, J. J. Sloth, K. L. Eybye, S. Kobbelgaard, G. Jakobsen, F. Jessen, Int. J. Biol. Macromol., 2020, 156, 918; DOI: https://doi.org/10.1016/j.ijbiomac.2020.04.047.
I. Ruiz-Rizaldos, J. Baviera-Sabater, J. Ortega-Usobiaga, J. Archivos de la Sociedad Espanola de Oftalmologia, 2017, 92, 233; DOI: https://doi.org/10.1016/j.oftal.2016.11.002.
Funding
This work was carried out using the Center for Collective Use “Novel Materials and Resource Saving Technologies.”
This work was financially supported by the Ministry of Science and Higher Education of the Russian Federation (base part of state assignment, Project No. 0729-2020-0039).
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 2, pp. 389–398, February, 2022.
No human or animal subjects were used in this research.
The authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Kuznetsova, Y.L., Morozova, E.A., Sustaeva, K.S. et al. Tributylborane in the synthesis of graft copolymers of collagen and polymethyl methacrylate. Russ Chem Bull 71, 389–398 (2022). https://doi.org/10.1007/s11172-022-3424-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-022-3424-3