Abstract
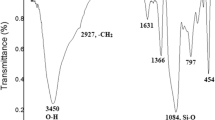
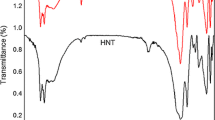
This research work focuses on the potential usefulness of iron ion-exchanged Indian bentonite for one-pot synthesis of 3,4-dihydropyrimidine-2(1H)-thiones. Our goal was to produce mesoporous Fe3+-exchanged Indian clay (Fe3+-ExIC) and its supported forms through a wet impregnation technique. The physicochemical properties of the catalysts, such as XRD, FTIR, TGA, physisorption studies, SEM, and acidity, were investigated. It was found that Fe3+-ExIC catalysts are efficient and reusable acidic heterogeneous catalysts for the synthesis of 3,4-dihydropyrimidine-2(1H)-thiones by refluxing aldehyde, methyl acetoacetate and thiourea with acetonitrile in cyclocondensation. An optimized reaction condition was found for preparing 5-methoxycarbonyl-6-methyl-4-phenyl-3,4-dihydropyrimidin-2(1H)-thione. A possible mechanism for the synthesis of 3,4-dihydropyrimidine-2(1H)-thiones has been proposed. Melting point, FTIR, and proton NMR were used to identify the products. Adequate catalyst activity was maintained over a period of six cycles, and a reduction in activity was attributed to a decrease in the acidity of the recycled catalyst.













Similar content being viewed by others
Data availability
Data will be made available upon reasonable request.
References
K.S. Atwal, B.N. Swanson, S.E. Unger, D.M. Floyd, S. Moreland, A. Hedberg, B.C. O’Reilly, J. Med. Chem. 34, 806 (1991)
C.O. Kappe, Eur. J. Med. Chem. 35, 1043 (2000)
W.M.B.I.A. Nagwa, M. Abdelazeem Farid, M. Sroor, M.A. Tantawy, Polycycl. Aromat. Compd. 43, 5840 (2023)
M. Brands, R. Endermann, R. Gahlmann, J. Krüger, S. Raddatz, Bioorg. Med. Chem. Lett. 13, 241 (2003)
H.A. Stefani, C.B. Oliveira, R.B. Almeida, C.M.P. Pereira, R.C. Braga, R. Cella, V.C. Borges, L. Savegnago, C.W. Nogueira, Eur. J. Med. Chem. 41, 513 (2006)
N. Hosseini Nasab, H. Raza, R.S. Shim, M. Hassan, A. Kloczkowski, S.J. Kim, J. Mol. Struct. 1286, 135638 (2023)
A. Kumar, R.A. Maurya, Tetrahedron Lett. 48, 4569 (2007)
N. Mohammadian, B. Akhlaghinia, Res. Chem. Intermed. 43, 3325 (2017)
G. Van Der Heijden, E. Ruijter, R.V.A. Orru, Synlett 24, 666 (2013)
M.M. Anastas, P.T. Kirchhoff, Acc. Chem. Res. 35, 686 (2002)
R.A. Sheldon, Green Chem. 9, 1273 (2007)
P. Beigiazaraghbelagh, A. Poursattar Marjani, Res. Chem. Intermed. 50, 485 (2024)
S. Bibak, A. Poursattar Marjani, Sci. Rep. 13, 17894 (2023)
A. Poursattar Marjani, F. Asadzadeh, A. Danandeh Asl, Appl. Organomet. Chem. 37, 1 (2023)
A. Cornelis, P. Laszlo, P. Pennetreau, Clay Miner. 18, 437 (1983)
G. Nagendrappa, Appl. Clay Sci. 53, 106 (2011)
S. Bikas, A. Poursattar Marjani, S. Bibak, H. Sarreshtehdar Aslaheh, Sci. Rep. 13, 2564 (2023)
J.M. Adams, K. Martin, R.W. McCabe, J. Incl. Phenom. 5, 663 (1987)
S.D. Salim, K.G. Akamanchi, Catal. Commun. 12, 1153 (2011)
J. Lal, M. Sharma, S. Gupta, P. Parashar, P. Sahu, D.D. Agarwal, J. Mol. Catal. A Chem. 352, 31 (2012)
B. Vijayakumar, G.R. Rao, J. Porous Mater. 19, 491 (2012)
P. Gupta, S. Paul, J. Mol. Catal. A Chem. 352, 75 (2012)
R. Tayebee, M.M. Amini, M. Ghadamgahi, M. Armaghan, J. Mol. Catal. A Chem. 366, 266 (2013)
F. Tamaddon, S. Moradi, J. Mol. Catal. A Chem. 370, 117 (2013)
A. Rajack, K. Yuvaraju, C. Praveen, Y.L.N. Murthy, J. Mol. Catal. A Chem. 370, 197 (2013)
J. Safari, S. Gandomi-Ravandi, J. Mol. Catal. A Chem. 373, 72 (2013)
G. Kour, M. Gupta, S. Paul, Rajnikant, V.K. Gupta, J. Mol. Catal. A Chem. 392, 260 (2014)
K. Kouachi, G. Lafaye, S. Pronier, L. Bennini, S. Menad, J. Mol. Catal. A Chem. 395, 210 (2014)
Y. Titova, O. Fedorova, G. Rusinov, A. Vigorov, V. Krasnov, A. Murashkevich, V. Charushin, Catal. Today 241, 270 (2015)
A. Mobinikhaledi, N. Foroughifar, A. Khajeh-Amiri, React. Kinet. Mech. Catal. 117, 59 (2016)
D. Elhamifar, D. Elhamifar, F. Shojaeipoor, J. Mol. Catal. A Chem. 426, 198 (2017)
M. Sheykhan, A. Yahyazadeh, L. Ramezani, Mol. Catal. 435, 166 (2017)
J. Safaei-Ghomi, M. Tavazo, G.H. Mahdavinia, Ultrason. Sonochem. 40, 230 (2018)
E. Dezfoolinezhad, K. Ghodrati, R. Badri, SILICON 11, 1593 (2019)
R. Esmaeili, L. Kafi-Ahmadi, S. Khademinia, J. Mol. Struct. 1216, 128124 (2020)
F. Ramezani Gomari, S. Farahi, H. Arvinnezhad, Iran. J. Chem. Chem. Eng. 40, 888 (2021)
B. Mohammadi, F.K. Behbahani, G.B. Marandi, B. Mirza, Russ. J. Org. Chem. 58, 1319 (2022)
F. Mohamadpour, Sci. Rep. 13, 13142 (2023)
F. Mohamadpour, Polycycl. Aromat. Compd. (2023)
M. Nikpassand, L.Z. Fekri, M. Gharib, O. Marvi, Lett. Org. Chem. 9, 745 (2013)
M. Kancherla, V. Badathala, J. Porous Mater. 24, 1187 (2017)
B. Vijayakumar, G.R. Rao, J. Porous Mater. 19, 233 (2012)
V. Singh, R. Ratti, S. Kaur, J. Mol. Catal. A Chem. 334, 13 (2011)
X.X. Zheng, Z.P. Fang, Z.J. Dai, J.M. Cai, L.J. Shen, Y.F. Zhang, C.T. Au, L.L. Jiang, Inorg. Chem. 59, 4483 (2020)
S. Sadjadi, M.M. Heravi, M. Malmir, Res. Chem. Intermed. 43, 6701 (2017)
Acknowledgements
BVK is thankful to the SERB, Ministry of DST, Government of India for sanctioning the research project (Project No. SB/FT/CS-089/2012). The authors thank Vel Tech High Tech Dr. RR Dr.SR Engineering College for providing research facilities.
Funding
The SERB, Ministry of DST, Government of India had provided financial support for this study (Project No. SB/FT/CS-089/2012).
Author information
Authors and Affiliations
Contributions
MK: Experimental and Characterization work analysis; KS: Data analysis, Visualization, and Characterization work analysis; VB: Conceptualization, Investigation, Writing-Original Draft, Funding Acquisition.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no competing interests to declare that are relevant to the content of this article.
Ethical approval and consent to participate
Does not require any ethical approval since we could not use any animals or Humans for this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kancherla, M., Seku, K. & Badathala, V. An efficient three-component one-pot synthesis of 3, 4-dihydropyrimidin-2-(1H)-thione derivatives with a mesoporous iron ion exchanged Indian clay catalyst. Res Chem Intermed 50, 2307–2324 (2024). https://doi.org/10.1007/s11164-024-05247-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-024-05247-z