Abstract
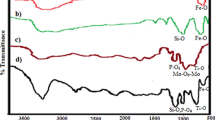
In this research, 14-aryl-14-H-dibenzo[a,j]xanthene derivatives have successfully prepared through one pot reaction of the aldehyde derivatives and 2-naphthol employing the as-fabricated NiCuFe2O4 magnetic nanocatalyst under ultrasound irradiation. The structure of achieved derivatives was identified by using FT-IR, 1H NMR and melting point analyses. Moreover, FT-IR, XRD, FE-SEM, EDX, and VSM techniques were applied in order to verify the structure of NiCuFe2O4 magnetic nanoparticles. This protocol involves noticeable aspects such as; facile workup, saving costs, excellent yields, and reusability of the catalyst applying an external magnet. Furthermore, the used nanocatalyst was recycled and reused in five runs with slightly reduction in its catalytic activity.
Graphical abstract
In this research, 14-aryl-14-H-dibenzo[a,j]xanthene compounds have successfully prepared through the one pot reaction of diverse aldehyde derivatives and 2-naphthol employing the as-fabricated NiCuFe2O4 magnetic nanocatalyst under ultrasonic irradiation. The structure of achieved derivatives was corroborated using FT-IR, 1H NMR and melting point techniques.














Similar content being viewed by others
Data availability
Electronic supplementary material contains 1H NMR, FT-IR and microscopy data.
References
L.B. Santos, R.S. Assis, J.A. Barreto, M.A. Bezerra, C.G. Novaes, V.A. Lemos, TrAC. Trends Anal. Chem. 146, 116478 (2022)
M. Khorasani, H. Naeimi, Synth. Commun. 52, 1917 (2022)
Martins MTM, Dias FRF, de Moraes RSM, da Silva MFV, Lucio KR, D’Oliveira Góes K, do Nascimento PA, da Silva ASS, Ferreira VF, Cunha AC, Chem. Rec. 22, (2022). https://doi.org/10.1002/tcr.202100251
H. Yazdani, S.E. Hooshmand, R.S. Varma, Org. Chem. Front. 9, 4178 (2022)
H. Liu, P. Kanjilal, S. Thayumanavan, Polym. Int. 71, 562 (2022)
A.N. Dadhania, V.K. Patel, D.K. Raval, C. R. Chim. 15, 378 (2012)
Y. Song, Y. Yang, L. Wu, N. Dong, S. Gao, H. Ji, X. Du, B. Liu, G. Chen, Molecules 22, 517 (2017)
S. De, S. Das, A. Girigoswami, Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 61, 1821 (2005)
A. Tabara, C. Yamane, M. Abe, M. Seguchi, Cellulose 18, 45 (2011)
Quintás D, Garcı́a A, Domı́nguez D (2003) Tetrahedron Lett. 44, 9291
K. Chibale, M. Visser, D. van Schalkwyk, P.J. Smith, A. Saravanamuthu, A.H. Fairlamb, Tetrahedron 59, 2289 (2003)
J. Abdi, A.J. Sisi, M. Hadipoor, A. Khataee, J. Hazard. Mater. 424, 127558 (2022)
S.H. Mosavi, R. Zare-Dorabei, M. Bereyhi, ChemistrySelect 6, 4432 (2021)
G. Cravotto, P. Cintas, Chem. Soc. Rev. 35, 180 (2006)
X. Cheng, A. Lei, T.-S. Mei, H.-C. Xu, K. Xu, C. Zeng, CCS Chem. 4, 1120 (2022)
X. Sui, L. Zhang, J. Li, K. Doyle-Davis, R. Li, Z. Wang, X. Sun, Adv. Energy Mater. 12, 2102556 (2022)
R. Cheng, L. Yin, Y. Wen, B. Zhai, Y. Guo, Z. Zhang, W. Liao, W. Xiong, H. Wang, S. Yuan, J. Jiang, C. Liu, J. He, Nat. Commun. 13, 5241 (2022)
H. Qin, Y. He, P. Xu, D. Huang, Z. Wang, H. Wang, Z. Wang, Y. Zhao, Q. Tian, C. Wang, Adv. Colloid Interface Sci. 294, 102486 (2021)
S.B. Narang, K. Pubby, J. Magn. Magn. Mater. 519, 167163 (2021)
D. Hong, Y. Yamada, T. Nagatomi, Y. Takai, S. Fukuzumi, J. Am. Chem. Soc. 134, 19572 (2012)
Y. Ge, X. Qin, A. Li, Y. Deng, L. Lin, M. Zhang, Q. Yu, S. Li, M. Peng, Y. Xu, X. Zhao, M. Xu, W. Zhou, S. Yao, D. Ma, J. Am. Chem. Soc. 143, 628 (2021)
R. Kumar, G.C. Nandi, R.K. Verma, M.S. Singh, Tetrahedron Lett. 51, 442 (2010)
P. Kumari, V. Yathindranath, S.M.S. Chauhan, Synth. Commun. 38, 637 (2008)
M. Fathollahi, S. Rostamizadeh, A.M. Amani, Comb. Chem. High Throughput Screen. 21, 5 (2018)
G.H. Mahdavinia, S. Rostamizadeh, A.M. Amani, Z. Emdadi, Ultrason. Sonochem. 16, 7 (2009)
H. Naeimi, Z.S. Nazifi, J. Chin. Chem. Soc. 60, 1113 (2013)
J. Safari, P. Aftabi, M. Ahmadzadeh, M. Sadeghi, Z. Zarnegar, J. Mol. Struct. 1142, 33 (2017)
B. Das, B. Ravikanth, R. Ramu, K. Laxminarayana, B.V. Rao, J. Mol. Catal. A Chem. 255, 74 (2006)
R. Tayebee, S. Tizabi, Chin. J. Catal. 33, 962 (2012)
A. Khosropour, M. Khodaei, H. Moghannian, Synlett 2005, 0955 (2005)
H. Naeimi, S. Lahouti, RSC Adv. 7, 2555 (2017)
N.G. Khaligh, F. Shirini, Ultrason. Sonochem. 22, 397 (2015)
K. Tabatabaeian, M.A. Zanjanchi, M. Mamaghani, A. Dadashi, Res. Chem. Intermed. 42, 5049 (2016)
A. Marandi, N. Koukabi, M.A. Zolfigol, Res. Chem. Intermed. 47, 3145 (2021)
Acknowledgements
The authors are so thankful to Kashan University for supporting this work by Grant No. 159148/81.
Author information
Authors and Affiliations
Contributions
Azin Alipour and Hossein Naeimi wrote the main manuscript text and Azin Alipour prepared all of the figures. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Not applicable.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
In accordance with the copyright transfer or open access rules.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Alipour, A., Naeimi, H. Design, fabrication and characterization of magnetic nickel copper ferrite nanocomposites and their application as a reusable nanocatalyst for sonochemical synthesis of 14-aryl-14-H-dibenzo[a,j]xanthene derivatives. Res Chem Intermed 49, 2705–2723 (2023). https://doi.org/10.1007/s11164-023-04981-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-023-04981-0