Abstract
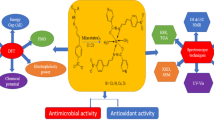
Sixteen Co(II), Ni(II), Cu(II) and Zn(II) complexes were synthesized by four Schiff base ligands 4-((4-(benzyloxy)-2-hydroxybenzylidene)amino)-[1,1'-biphenyl]-3-ol (H2L1), 6-((4-(benzyloxy)-2-hydroxybenzylidene)amino)-4,6-dichloro-3-methylphenol (H2L2), 2-((4-(benzyloxy)-2-hydroxybenzylidene)amino)-6-chloro-4-nitrophenol (H2L3), 5-(benzyloxy)-2-(((2-hydroxyphenyl)imino)methyl)phenol (H2L4) obtained from condensation reaction of 4-(benzyloxy)-2-hydroxybenzaldehyde with various aminophenol derivatives and well characterized by various spectroscopic techniques (FT-IR, UV–Vis, NMR, ESR, SEM, fluorescence), mass spectrometry, elemental analysis (CHN) and physical studies (TGA, XRD, molar conductance, melting point). The various characterization data suggested that the Schiff base ligands coordinated via nitrogen of azomethine and deprotonated phenolic oxygen atoms (ONO) with metal ions in 1:1 molar ratio having general formula [M(L1−4)(H2O)2(CH3COO)]. Low values of molar conductance of compounds suggested their non-electrolytic nature. The thermal decomposition of complexes indicated that metal complexes are stable up to 150 °C and give metal oxide as end product. Using SEM analysis, the surface morphology of the synthesized compound was determined. The compounds (1–20) were evaluated for their in vitro antioxidant activity and found that the synthesized metal(II) complexes are highly potent and also show good efficiency for decolourizing the purple-coloured solution of DPPH compared to free Schiff base ligands and Cu(II) complexes were most potent having IC50 value from 2.98 to 3.89 µM range. The compounds (1–20) were assayed for their in vitro antimicrobial activities against four bacterial strains (S. aureus, B. subtilis, P. aeruginosa, E. coli) and two fungal strains (A. niger, C. albicans) by serial dilution method, and it was found that the metal(II) complexes are more noxious than free Schiff base ligands. The antimicrobial activity results showed that the complexes 10, 11, 14 and 15 were most active compounds. Antifungal activity of complex 11 (Cu(L2)(CH3COO)(H2O) against C. albicans was found to be comparable to standard drug. The molecular docking of ligand H2L2 (2) and its Cu(II) complex (11) with enzyme C. albicans sterol 14-alpha demethylase suggested the hydrophobic binding. Furthermore, in silico study stressed that the compounds may be used as orally active drugs.
Graphical abstract

The synthesized compounds (1–20) were screened for in vitro antimicrobial activity. Copper complex (11) was found to be the most potent antimicrobial agent and which is held by hydrophobic interactions in the active site of the enzyme C. albicans sterol 14-alpha demethylase.












Similar content being viewed by others
References
S. Ershad, L. Sagathforoush, G. Karim-Nezhad, S. Kangari, Int. J. Electrochem. Sci. 4, 846 (2009)
U. Kendur, G.H. Chimmalagi, S.M. Patil, K.B. Gudasi, C.S. Frampton, C.V. Mangannavar, I.S. Muchchandi, J. Mol. Struct. 1153, 299 (2018)
W.H. Mahmoud, R.G. Deghadi, G.G. Mohamed, Appl. Organomet. Chem. 30, 221 (2016)
P. Sen, L.S. Mpeta, J. Mack, T. Nyokong, J. Lumin. 224, 117262 (2020)
S. Zhu, S. Zhai, Z. Chen, H. Su, C. Zhang, R. Liu, H. Zhu, Dyes Pigments 182, 108591 (2020)
T.H. Sanatkar, A. Khorshidi, E. Sohouli, J. Janczak, Inorganica Chim. Acta 506, 119537 (2020)
S. Gurusamy, K. Krishnaveni, M. Sankarganesh, V. Sathish, P. Thanasekaran, A. Mathavan, J. Mol. Liq. 325, 115190 (2021)
M. Azam, S.M. Wabaidur, M. Alam, A. Trzesowska-Kruszynska, R. Kruszynski, S.I. Al-Resayes, M.R. Khan, Polyhedron 195, 114991 (2021)
D. Majumdar, Y. Agrawal, R. Thomas, Z. Ullah, M.K. Santra, S. Das, D. Mishra, Appl. Organomet. Chem. 34, e5269 (2020)
C.X. Hiller, U. Hübner, S. Fajnorova, T. Schwartz, Sci. Total Environ. 685, 596 (2019)
S.S. Saleem, M. Sankarganesh, P.A. Jose, J.D. Raja, Inorg. Chem. Commun. 124, 108396 (2021)
R.N. Asha, M. Sankarganesh, N. Bhuvanesh, B.R. Nayagam, J. Mol. Struct. 1250, 131692 (2022)
A. Mahal, R. Abu-El-Halawa, S.A. Zabin, M. Ibrahim, M. Al-Refai, T. Kaimari, World J. Org. Chem. 3, 1 (2015)
Y. Deswal, S. Asija, D. Kumar, D.K. Jindal, G. Chandan, V. Panwar, N. Kumar, Res. Chem. Intermed., 1 (2021)
M. Shebl, M. Saif, A.I. Nabeel, R. Shokry, J. Mol. Struct. 1118, 335 (2016)
A. Mumtaz, T. Mahmud, M.R. Elsegood, G.W. Weaver, J. Nucl. Med. Radiat. Ther 7, 2 (2016)
M.A. Malik, O.A. Dar, P. Gull, M.Y. Wani, A.A. Hashmi, Med. Chem. Comm 9, 409 (2018)
M.M.Y. Kuddushi, M.A.H. Malek, V.L. Patidar, M.S. Patel, R.K. Patel, R.H. Dave, Int. J. Recent Sci. Res. 9, 26026 (2018)
D.G. Kokare, K. Naik, A. Nevrekar, A. Kotian, V. Kamat, V.K. Revankar, Appl. Organomet. Chem. 30, 181 (2016)
J. Devi, S. Kumar, D. Kumar, D. K. Jindal, Y. Poornachandra, Res. Chem. Intermed., 1 (2021)
M. Azam, S.I. Al-Resayes, A. Trzesowska-Kruszynska, R. Kruszynski, F. Shakeel, S.M. Soliman, S.M Wabaidur, J. Mol. Struct., 1201, 127177 (2020)
M. Sankarganesh, P.R. Adwin Jose, J. Dhaveethu Raja, R. Vijay Solomon, C. Dorothy Sheela, S. Gurusamy, J. Biomol Struct. Dyn., 1 (2021)
A. Madrid Villegas, L. Espinoza Catalán, I. Montenegro Venegas, J. Villena García, H. Carrasco Altamirano, Molecules 16, 4632 (2011)
Y.S. Moon, W.S. Choi, E.S. Park, I.K. Bae, S.D. Choi, O. Paek, S.E. Lee, Toxins 8, 240 (2016)
L. Deswal, V. Verma, D. Kumar, A. Kumar, M. Bhatia, Y. Deswal, A. Kumar, Future Med. Chem. 13, 975 (2021)
MarvinSketch 19.19.0, 2019, ChemAxon
J.-M. Yang, C.-C. Chen, Proteins Struct. Funct. Bioinf. 55, 288 (2004)
Dassault Systèmes BIOVIA, Discovery studio visualizer v17.2.0.16349, San Diego: DassaultSystèmes, (2016)
E.F. Pettersen, T.D. Goddard, C.C. Huang, E.C. Meng, G.S. Couch, T.I. Croll, J.H. Morris, T.E. Ferrin, Protein Sci. 30(1), 70 (2021)
S.H.S. Saleem, M. Sankarganesh, J.D. Raja, P.R.A. Jose, A. Sakthivel, T.C. Jeyakumar, R.N. Asha, J. Saudi Chem. Soc. 25, 101225 (2021)
J. Devi, J. Yadav, D. Kumar, D.K. Jindal, B. Basu, Appl. Organomet. Chem. 34, e5815 (2020)
J. Devi, M. Yadav, A. Kumar, A. Kumar, Chem. Pap. 72, 2479 (2018)
A. Sharma, M. Shah, J. Appl. Chem. 3, 62 (2013)
S.S. Jawoor, S.A. Patil, S.S. Toragalmath, J Coord Chem. 71, 271 (2018)
M. Yadav, S. Sharma, J. Devi, J. Chem Sci 133, 1 (2021)
J. Devi, J. Yadav, N. Singh, Res. Chem. Intermed. 45, 3943 (2019)
N. Revathi, M. Sankarganesh, J. Dhaveethu Raja, G.G. Vinoth Kumar, A. Sakthivel, R. Rajasekaran, J. Biomol. Struct. Dyn. 39, 3012 (2021)
A.N.M. Alaghaz, Y.A. Ammar, H.A. Bayoumi, S.A. Aldhlmani, J. Mol. Struct. 1074, 359 (2014)
C.J. Dhanaraj, I.U. Hassan, J. Johnson, J. Joseph, R.S. Joseyphus, J. Photochem. Photobiol. B Biol. 162, 115 (2016)
M.M. Omar, H.F. Abd El-Halim, E.A. Khalil, Appl. Organomet. Chem. 31, e3724 (2017)
J. Devi, M. Yadav, D.K. Jindal, D. Kumar, Y. Poornachandra, Appl. Organomet. Chem. 33, e5154 (2019)
H.A. El-Boraey, O.A. El-Gammal, Spectrochim. Acta A Mol. Biomol. Spectrosc. 138, 553 (2015)
N.A. El-Ghamaz, A.Z. El-Sonbati, M.A. Diab, A.A. El-Bindary, G.G. Mohamed, S.M. Morgan, Spectrochim. Acta A Mol. Biomol. Spectrosc. 147, 200 (2015)
J.R. Anacona, J.L. Rodriguez, J. Camus, Spectrochim. Acta A Mol. Biomol. Spectrosc. 129, 96 (2014)
S. Velumania, X. Mathew, P.J. Sebastian, S.K. Narayandass, D. Mangalaraj, Sol Cells 76, 347 (2003)
M.I. Khan, A. Khan, I. Hussain, M.A. Khan, S. Gul, M. Iqbal, F. Khuda, Inorg. Chem. Commun. 35, 104 (2013)
A.N.M.A. Alaghaz, S.A. Aldulmani, Appl. Organomet. Chem. 33, e5135 (2019)
H.F. El-Shafiy, M. Shebl, J. Mol. Struct. 403, 1156 (2018)
M. Shebl, M. Saif, A.I. Nabeel, R. Shokry, J. Mol. Struct. 335, 1118 (2016)
A.N.M. Alaghaz, H.A. Bayoumi, Y.A. Ammar, S.A. Aldhlmani, J. Mol. Struct. 1035, 383 (2013)
A.S. El-Tabl, M. Mohamed Abd El-Waheed, M.A. Wahba, A.E.H. Abou El-Fadl, Bioinorg Chem Appl., (2015)
K. Buldurun, N. Turan, A. Savci, N.C. Olak, J. Saudi Chem. Soc. 23, 205 (2019)
D.P. Singh, R. Kumar, M. Kamboj, V. Grover, K. Jain, J. Coord. Chem. 34, 233 (2008)
S. Ali, V. Singh, P. Jain, V. Tripathi, J. Saudi Chem. Soc. 23, 52 (2019)
D. Sathyanarayana, Electronic absorption spectroscopy and related techniques, First, Universities Press, New Delhi, (2001)
A. Lever, Inorganic Electronic Spectroscopy, Second (Elsevier, Amsterdam, 1984)
D. Billing, B. Hathaway, J. Chem. Phys. 50, 1476 (1969)
A.N.M. Alaghaz, J. Mol. Struct. 1072, 103 (2014)
N. Zhang, Y.H. Fan, C.F. Bi, X. Zhang, P.F. Zhang, Z.Y. Zhang, X. Li, J. Inorg. Organomet. Polym. 23, 1492 (2013)
A. Majumder, G.M. Rosair, A. Mallick, N. Chattopadhyay, S. Mitra, Polyhedron 25, 1753 (2006)
S. Basak, S. Sen, S. Banerjee, S. Mitra, G. Rosair, M.G. Rodriguez, Polyhedron 26, 5104 (2007)
D.B. Shpakovsky, C.N. Banti, E.M. Mukhatova, Y. Gracheva, V.P. Osipova, N.T. Berberova, T.A. Albov, D.V. Antonenko, L.A. Aslanov, E.R. Milaev, S.K. Hadjikakoub, Dalton Trans. 43, 6880 (2014)
S. Jabbar, I. Shahzadi, R. Rehman, H. Iqbal, Q. Ul-Ain, A. Jamil, R. Kousar, S. Ali, S. Shahzadi, M.A. Choudhary, M. Shahid, Q.M. Khan, S.K. Sharma, K. Qanungo, J. Coord. Chem. 65, 572 (2012)
M. Sirajuddin, S. Ali, F.A. Shah, M. Ahmad, M.N. Tahir, J. Iran. Chem. Soc. 11, 297 (2013)
T.A. Nibila, P.P. Soufeena, P. Periyat, K.K. Aravindakshan, J. Mol. Struct 1231, 129938 (2021)
M. Pervaiz, M. Yousaf, I. Ahmad, A. Munawar, Z. Saeed, A. Adnan, T. Gulzar, S. Kirn, M.T. Kamal, A. Ahmad, Spectrochim. Acta A Mol. Biomol Spectrosc. 18, 1386 (2018)
A. Rambabu, M. Pradeep Kumar, N. Ganji, S. Daravath, Shivaraj, J. Biomol. Struct. Dyn., 38, 307 (2020)
A. Rambabu, M.P. Kumar, S. Tejaswi, N. Vamsikrishna, Shivaraj, J. Photochem. Photobiol. B. Biol. 165, 147 (2016)
J. Devi, S. Sharma, S. Kumar, D.K. Jindal, P.P. Dutta, D. Kumar, Res. Chem. Intermed. 47, 2433 (2021)
L. John, R.S. Joseyphus, I.H. Joe, SN Appl. Sci. 2, 1 (2020)
J. Devi, N. Batra, R. Malhotra, Spectrochim. Acta A 97, 397 (2012)
P. Debnath, A. Das, K.S. Singha, T. Yama, S.S.K. Singh, R.J. Butcher, L. Sieron, W. Maniukiewicz, Inorg. Chim. Acta 498, 119 (2019)
A. Mai-Prochnow, M. Clauson, J. Hong, A.B. Murphy, Sci. Rep. 6, 38610 (2016)
Z. Breijyeh, B. Jubeh, R. Karaman, Molecules 25, 1340 (2020)
A. Daina, O. Michielin, V. Zoete, Sci. Rep. 7, 42717 (2017)
J.M. Pallicer, M. Rosés, C. Ràfol, E. Bosch, R. Pascual, A. Port, ADMET DMPK 2, 107 (2014)
Y. Zhao, M.H. Abraham, J. Lee, A. Hersey, N.C. Luscombe, G. Beck, B. Sherborne, I. Cooper, Pharm. Res. 19, 1446 (2002)
Acknowledgements
The author Mr. Sanjeev Kumar (SRF) expresses his sincere thanks to the CSIR, New Delhi, India (File No. 09/752(0095)/2019-EMR-I), for financial assistance. The authors are also thankful to Dr. A.P.J. Abdul Kalam Central Instrumentation laboratory and Department of Chemistry, Guru Jambheshwar University of Science & Technology, Hisar (Haryana), for providing facilities to carry out this research work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Devi, J., Kumar, S., Kumar, B. et al. Synthesis, structural analysis, in vitro antioxidant, antimicrobial activity and molecular docking studies of transition metal complexes derived from Schiff base ligands of 4-(benzyloxy)-2-hydroxybenzaldehyde. Res Chem Intermed 48, 1541–1576 (2022). https://doi.org/10.1007/s11164-021-04644-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-021-04644-y