Abstract
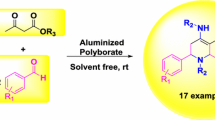
The present work reports a catalytic application of aluminized polyborate for the solvent-free one pot four-component green, efficient and cost-effective synthesis of polydroquinoline derivatives. The aluminized polyborate was synthesized from readily available non-toxic, inexpensive boric acid and aluminium trichloride and characterized by IR, XRD, SEM and EDAX techniques. The polyhydroquinolines were successfully synthesized by well-known Hantzsch condensation reaction of differently substituted aldehydes, dimedone, ethylacetoacetate and NH4OAc at 100 °C under solvent-free condition. The catalyst is stable and easy to use with some significant features such as good acidic catalytic activity, inexpensive and eco-friendly nature. Along with this, excellent product yields, short reaction time, clean reaction, operational simplicity, solvent-free condition and non-column purification are some more additional aspects of this protocol.
Graphic abstract










Similar content being viewed by others
References
S. Tu, F. Fang, C. Miao, H. Jian, Y. Feng, D. Shi, X. Wang, Tetrahedron Lett. 44, 6153 (2003)
M.K. Chaudhari, S. Hussian, J. Mol. Catal. A Chem. 269, 214 (2007)
T.B. Nguyen, J. Sorres, M.Q. Tran, L. Ermolenko, A. Al-Mourabit, Org. Lett. 14, 3202 (2012)
A. Shahrisa, S. Esmati, M.G. Nazari, J. Chem. Sci. 124, 927 (2012)
S. Chandrashekar, K. Gopalaiah, Tetrahedron Lett. 43, 2455 (2002)
K.S. Indalkar, C.K. Khatri, G.U. Chaturbhuj, J. Chem. Sci. 129, 141 (2017)
D.S. Rekunge, C.K. Khatri, G.U. Chaturbhuj, Montash Chem. 148, 2019 (2017)
M.S. Patil, C. Mudaliar, G.U. Chaturbhuj, Tetrahedron Lett. 58, 2859 (2017)
C.K. Khatri, G.U. Chaturbhuj, J. Iran. Chem. Soc. 14, 2513 (2017)
A.S. Mali, C.S. Potnis, G.U. Chaturbhuj, J. Iran. Chem. Soc. 15, 1399 (2018)
A. Zhu, R. Liu, C. Dua, L. Lia, RSC Adv. 7, 6679 (2017)
K.C. Nicolau, T. Montagnon, S.A. Snyder, Chem. Commun. 5, 551 (2003)
A. Maleki, R. Paydar, React. Funct. Polym. 109, 120 (2016)
V. Polshettiwar, R.S. Varma, Green Chem. 12, 743 (2010)
M.T. Vladu, Chem. Soc. Rev. 43, 588 (2014)
T. Godfraind, R. Miller, M. Wibo, Pharmacol. Rev. 38, 321 (1986)
R.A. Jonis, D.J. Triggle, J. Med. Chem. 26, 775 (1983)
D.M. Stout, A.I. Meyers, Chem. Rev. 82, 223 (1982)
D.J. Triggle, Drug Dev. Res. 58, 5 (2003)
H.S. Chung, W.S. Woo, J. Nat. Prod. 64, 1579 (2001)
O.A. El-Sayed, T.M. Al-Turki, H.M. Al-Dattiri, B.A. Al-Bassam, M.E. Hussein, Boll. Chim. Farm. 10, 227 (2004)
H. Gevariya, B. Desai, V. Vora, A. Shah, Heterocycl. Commun. 7, 481 (2011)
A. Hilgeroth, Mini-Rev. Med. Chem. 2, 235 (2002)
A. Maleki, A.R. Akbarzade, A.R. Bhat, J. Nanostruct. Chem. 7, 309 (2017)
A. Maleki, N. Hamidi, S. Maleki, J. Rahimi, Appl. Organomet. Chem. 32, 4245 (2018)
J. Davarapanah, M. Ghahremani, O. Najafi, J. Mol. Struct. 1177, 525 (2019)
P. Choudhury, P. Ghosh, B. Basu, Mol. Divers. 24, 283 (2020)
T. Tamoradi, M. Ghadermazi, A. Ghorbani-Choghamarani, Catal. Lett. 149, 2645 (2019)
P. Mondal, S. Chatterjee, P. Sarkar, A. Bhaumik, C. Mukhopadhyay, ChemistrySelect 4, 11701 (2019)
S.J. Yu, S. Wu, X.M. Zhao, C.W. Lu, Res. Chem. Intermed. 43, 3121 (2017)
A. Ghorbani-Choghmarani, Z. Heidarnezhad, B. Tahmasbi, G. Azadi, J. Iran. Chem. Soc. 15, 2281 (2018)
T. Tamoradi, S.M. Mousavi, Polyhedron 175, 114211 (2020)
T. Tamoradi, A. Ghorbani-Choghamarani, M. Ghadermazi, H. Veisi, Solid State Sci. 91, 96 (2019)
A. Yaghoubi, M.G. Dekamin, B. Karimi, Catal. Lett. 147, 2656 (2017)
B. Maleki, H. Alinezhad, H. Atharifar, R. Tayebee, A.V. Mofrad, Org. Prep. Proced. Int. 51, 301 (2019)
R.R. Harale, P.V. Shitre, B.R. Sathe, M.S. Shingare, Res. Chem. Intermed. 43, 3237 (2017)
T. Momeni, M.M. Heravi, T. Hosseinnejad, M. Mirzaei, V. Zadsirjan, J. Mol. Struc. 1199, 127011 (2020)
M.A. Ashraf, Z. Liu, W. Peng, C. Gao, Catal. Lett. 150, 683 (2020)
S. Hashemi-Uderji, M. Abdollahi-Alibeik, R.J. Ranjbar-Karimi, J. Porous Mater. 26, 467 (2019)
S.C. Jadhvar, H.M. Kasraliker, S.V. Goswami, A.V. Chakrawar, S.R. Bhusare, Res. Chem. Intermed. 43, 7211 (2017)
N.G. Khaligh, Chin. J. Catal. 35, 1036 (2014)
G.B. Dharma Rao, S. Nagakalyan, G.K. Prasad, RSC Adv. 7, 3611 (2017)
B. Sakram, B. Sonyanaik, K. Ashok, S. Rambhau, Res. Chem. Intermed. 42, 7651 (2016)
S.J. Saghanezhad, M.H. Sayahi, I. Imanifar, M. Mombeni, S.D. Hamood, Res. Chem. Intermed. 43, 6521 (2017)
I. Sheout, R. Boulcina, B. Boudjemaa, T. Boumoud, A. Debache, Synth. Commun. 47, 1185 (2017)
R. Ghafouri-Nejad, M. Hajjami, React. Kinet. Mech. Cat. 129, 371 (2020)
A. Ghorbani-Choghamarani, M. Mohammadi, S. Lotfi, Z. Taherinia, Res. Chem. Intermed. 45, 5705 (2019)
M. Abdollahi-Alibeik, S.S. Hoseinikhah, J. Iran. Chem. Soc. 13, 1339 (2016)
Y. Chen, Z. Zhang, W. Jiang, M. Zhang, Y. Li, Mol. Divers. 23, 421 (2019)
A. Ghorbani-Choghamarani, P. Moradi, B. Tahmasbi, J. Iran. Chem. Soc. 16, 511 (2019)
A. Maleki, R. Rahimi, S. Maleki, N. Hamidi, RSC Adv. 4, 29765 (2014)
A. Maleki, V. Eskandarpour, J. Rahimi, N. Hamidi, Carbohydr. Polym. 208, 251 (2019)
M. Aziz, A. Maleki, F. Hakimpoor, R.F. Haji, M. Ghassemi, J. Rahimi, Lett. Org. Chem. 15, 753 (2018)
A.V. Borhade, B.K. Uphade, A.G. Gadhave, Res. Chem. Intermed. 42, 2016 (2016)
A.V. Borhade, B.K. Uphade, A.G. Gadhave, Res. Chem. Intermed. 41, 1447 (2015)
G.D. Shirole, A.G. Gadhave, S. Bhalekar, S.N. Shelke, Indian J. Heterocycl. Chem. 27, 195 (2017)
A.G. Gadhave, R.B. Gaikar, S.R. Kuchekar, B.K. Karale, J. Heterocycl Chem. 51, 1849 (2014)
R.L. Siqueira, I.V.P. Yoshida, L.C. Pardini, M.A. Schiavon, Mater. Res. 10, 147 (2007)
J.S. Yeo, T.H. Park, S.H. Yoon, M.H. Seo, J. Miyawaki, I. Mochinda, Int. J. Electrochem. Sci. 8, 1308 (2013)
A. Khalafi-Nezhad, H. Ollah Foroughi, M.M. Doroodmand, F. Panahi, J. Mater. Chem. 21, 12842 (2011)
H.J. Jung, Y. Sohn, H.S. Hyun, W.G. Shin, Powder Technol. 269, 548 (2015)
C. Mandilas, E. Daskalos, G. Karagiannakis, A.G. Konstandopoulos, Mater. Sci. Eng. B 178, 22 (2013)
Acknowledgements
The authors are grateful to Principal (P.V.P. College, Pravaranagar & A.S.C. College, Satral) for providing necessary facilities and constant encouragement. We express our gratitude towards Principal Dr. B. K. Karale for valuable guidance. We also thank to SAIF, Punjab University, Chandigarh, for spectral analysis.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Aute, D., Kshirsagar, A., Uphade, B. et al. Aluminized polyborate-catalysed green and efficient synthesis of polyhydroquinolines under solvent-free conditions. Res Chem Intermed 46, 3491–3508 (2020). https://doi.org/10.1007/s11164-020-04158-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-020-04158-z