Abstract
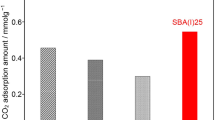
Three different aminosilanes ((3-aminopropyl)trimethoxysilane (1NS), N-[3-(trimethoxysilyl) propyl]ethylenediamine (2NS), N1-(3-trimethoxysilylpropyl)diethylenetriamine (3NS)) were grafted covalently inside nanoporous silica (NPS-1) with a large surface area to prepare CO2 adsorbents. The prepared CO2 sorbents were evaluated for their CO2 sorption capacity, kinetic behavior, temperature programmed desorption (TPD) and textural properties. Grafting efficiency of 1NS was better due to the smaller molecular size compared to 2NS and 3NS, which are difficult to react with the hydroxyl group of the silica surface due to steric hindrance. The highest adsorption capacity of 7.0 wt% was observed for the 2NS/NPS-1 adsorbent, followed by 5.2 wt% for 1NS/NPS-1, then 5.0 wt% for 3NS/NPS-1. The adsorption capacity of 2NS/NPS-1 was highest at 30 °C, and it gradually decreased as the adsorption temperature increased. TPD analysis showed that the reaction of primary amine of 2NS with CO2 inside the nanoporous silica could form less thermally stable carbamic acid and carbamate compared to 1NS and 3NS.







Similar content being viewed by others
References
J.C. Chow, J.G. Watson, A. Herzog, S.M. Benson, G.M. Hidy, W.D. Gunter, S.J. Penkala, C.M. White, J. Air Waste Manag. Assoc. 53, 645 (2003)
M.L. Gray, Y. Soong, K.J. Champagne, H. Pennline, J.P. Baltrus, R.W. Stevens Jr., R. Khatri, S.S.C. Chuang, T. Filburn, Fuel Process. Technol. 86, 1449 (2005)
H. Yang, Z. Xu, M. Fan, R. Gupta, R.B. Slimane, A.E. Bland, I. Wright, J. Environ. Sci. 20, 14 (2008)
J.C. Hicks, J.H. Drese, D.J. Fauth, M.L. Gray, G. Qi, C.W. Jones, J. Am. Chem. Soc. 130, 2902 (2008)
R. Veneman, Z.S. Li, J.A. Hogendoorn, S.R.A. Kersten, D.W.F. Brilman, Chem. Eng. J. 207, 18 (2012)
Y.C. Park, S.H. Jo, S.Y. Lee, J.H. Moon, C.K. Ryu, J.B. Lee, C.K. Yi, Korean J. Chem. Eng. 33, 73 (2016)
G.P. Knowles, J.V. Graham, S.W. Delaney, A.L. Chaffee, Fuel Process. Technol. 86, 1435 (2005)
D. H. Kim, J. M. Celedonio, Y. S. Ko, Top. Catal. 60, 706 (2017)
J.H. Park, J.M. Celedonio, H. Seo, Y.K. Park, Y.S. Ko, Catal. Today 265, 68 (2016)
J.M. Celedonio, J.H. Park, Y.S. Ko, Res. Chem. Intermed. 42, 141 (2016)
P.J.E. Harlick, A. Sayari, Ind. Eng. Chem. Res. 46, 446 (2007)
R.S. Franchi, P.J.E. Harlick, A. Sayari, Ind. Eng. Chem. Res. 44, 8007 (2005)
Y. Belmabkhout, A. Sayari, Adsorpt. J. Int. Adsorpt. Soc. 15, 318 (2009)
A. Danon, P.C. Stair, E. Weitz, J. Phys. Chem. C 115, 11540 (2011)
E.D. Canck, L. Ascoop, A. Sayari, P.V.D. Voort, Phys. Chem. Chem. Phys. 15, 9792 (2013)
I. Kim, H.F. Svendsen, Ind. Eng. Chem. Res. 46, 5803 (2007)
N. McCann, M. Maeder, M. Attalla, Ind. Eng. Chem. Res. 47, 2002 (2008)
M.B. Yue, L.B. Sun, Y. Cao, Z.J. Wang, Y. Wang, Q. Yu, J.H. Zhu, Microporous Mesoporous Mater. 114, 74 (2008)
M.W. Hahn, J. Jelic, E. Berger, K. Reuter, A. Jentys, J.A. Lercher, J. Phys. Chem. B 120, 1988 (2016)
Acknowledgements
This work was supported by a Korea CCS R&D Center (KCRC) grant funded by the Korea government (Ministry of Science, ICT, and Future Planning) (No. 2014049259).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, D.H., Ko, Y.S. Effect of various aminosilanes functionalized inside nanoporous silica on CO2 adsorption performance. Res Chem Intermed 44, 3661–3672 (2018). https://doi.org/10.1007/s11164-018-3373-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-018-3373-5