Abstract
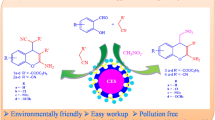
The calcinized eggshell as an efficient and green catalyst has been prepared from chicken eggshell waste and is characterized by different analytical techniques such as FT-IR, XRD, TGA, SEM, and EDAX. A series of substituted 2-arylbenzothiazoles have been synthesized by reaction of o-aminothiophenol and aromatic aldehydes under solvent free conditions using a grinding method at room temperature. The reaction proceeds smoothly in excellent yields (86–97 %), short reaction time (15–48 min), and with an easy work-up procedure. The reuse of catalyst and purification of products by non-chromatographic methods are some additional features of the present protocol. The calcinized eggshell is a safe, inexpensive, and a green catalyst with high catalytic efficiency obtained from renewable resources.






Similar content being viewed by others
References
I. Hutchinson, M.S. Chua, H.L. Browne, V. Trapani, T.D. Bradshaw, A.D. Westwell, M.F.G. Stevens, J. Med. Chem. 44(9), 1446 (2001)
H.L.K. Stanton, G. Roberto, C.H. Chung, C.W.Y. Marcus, L. Eva, S.M.W. Raymond, Y.L. Fung, Y.M.C. Gregory, S.L. Wing, H.C. Sau, H.L. Kim, H.C. Chor, B.S.L. Paul, W.Y.Y. Michael, C. Filly, C.O.T. Johnny, S.C.C. Albert, Bioorg. Med. Chem. 16(7), 3626 (2008)
M. Wang, M. Gao, B.H. Mock, K.D. Miller, G.W. Sledge, G.D. Hutchins, Q.H. Zheng, Bioorg. Med. Chem. 14(24), 8599 (2006)
H.M. Bryson, B. Fulton, P. Benfield, Drugs 52(4), 549 (1996)
S.R. Pattan, C. Suresh, V.D. Pujar, V.V.K. Reddy, V.P. Rasal, B.C. Koti, Indian J. Chem. 4B(11), 2404 (2005)
S. Tasler, O. Muller, T. Wieber, T. Herz, S. Pegoraro, W. Saeb, M. Lang, R. Krauss, F. Totzke, U. Zirrgiebel, J.E. Ehlert, M.H.G. Kubbutat, C. Schachtele, Bioorg. Med. Chem. 17(18), 6728 (2009)
B.C. Lee, J.S. Kim, B.S. Kim, J.Y. Son, S.K. Hong, H.S. Park, B.S. Moon, J.H. Jung, J.M. Jeong, S.E. Kim, Bioorg. Med. Chem. 19(9), 2980 (2011)
P. Naresh, P. Pattanaik, B. Rajeshwar, Int. J. Pharm. Sci. 3(2), 170 (2013)
A.K. Chakraborti, C. Selvam, G. Kaur, S. Bhagat, Synlett 5, 851 (2004)
A. Rostami, A. Yari, J. Iran. Chem. Soc. 9(4), 489 (2012)
N. Azizi, A.K. Amiri, R. Baghi, M. Bolourtchian, M.M. Hashemi, Monatsh. Chem. 140(12), 1471 (2009)
T.G. Deligeorgiev, S. Kaloyanova, A. Vasilev, J.J. Vaquero, Phosphorus Sulfur Silicon Relat. Elem. 185(11), 2292 (2010)
B. Maleki, H. Salehabadi, M.K. Moghaddam, Acta Chim. Slov. 57(3), 741 (2010)
C. Mukhopadhyay, A. Datta, J. Heterocycl. Chem. 46(1), 91 (2009)
A.K. Chakraborti, S. Rudrawar, K.B. Jadhav, G. Kaur, S.V. Chankeshwara, Green Chem. 9(12), 1335 (2007)
R.H. Shoar, M. Heidary, M. Farzaneh, R. Malakouti, Synth. Commun. 39(10), 1742 (2009)
H.L. Xiao, J.X. Chen, M.C. Liu, D.J. Zhu, J.C. Ding, H.Y. Wu, Chem. Lett. 38(2), 170 (2009)
H. Veisi, R. Ghorbani-Vaghei, A. Fraji, T. Ozturk, Chin. J. Chem. 28(11), 2249 (2010)
Y. Li, Y.L. Wang, J.Y. Wang, Chem. Lett. 35(4), 460 (2006)
U.R. Pratap, J.R. Mali, D.V. Jawale, R.A. Mane, Tetrahedron Lett. 50, 1352 (2009)
H. Eshghi, M. Rahimizadeh, A. Shiri, P. Sedaghat, Bull. Korean Chem. Soc. 33(2), 515 (2012)
K.U. Sadek, R.A. Mekheimer, A.M. Abdel Hameed, F. Elnahas, M.H. Elnagdi, Molecules 17, 6011 (2012)
B.S. Londhe, U.R. Pratap, J.R. Mali, R.A. Mane, Bull. Korean Chem. Soc. 31(8), 2329 (2010)
F. Al Qalaf, R.A. Mekheimer, K.U. Sadek, Molecules 13(11), 2908 (2008)
P.S. Chandrachood, D.R. Garud, T.V. Gadakari, R.C. Torane, N.R. Deshpande, R.V. Kashalkar, Acta Chim. Slov. 58(2), 367 (2011)
C.H. Gill, M.D. Nikam, P.S. Mahajan, A.V. Chate, S.K. Dabhade, P.V. Badadhe, Res. Chem. Intermed. 41(10), 7509 (2015)
Z.Y. Yu, Q.S. Fang, J. Zhou, Z.B. Song, Res. Chem. Intermed. (2015). doi:10.1007/s11164-015-2133-z
M. Abdollahi-Alibeik, S. Poorirani, Phosphorus Sulfur Silicon Relat. Elem. 184(12), 3182 (2009)
K. Bahrami, M.M. Khodaei, F. Naali, J. Org. Chem. 73(17), 6835 (2008)
A. Amoozadeh, R.A. Azadeh, S. Rahmani, M. Salehi, M. Kubicki, G. Dutkiewicz, Phosphorus, Sulfur Silicon Relat. Elem. 190(11), 1874 (2015)
G.F. Chen, N. Xiao, J.S. Yang, H.Y. Li, B.H. Chen, L.F. Han, Res. Chem. Intermed. 41(8), 5159 (2015)
P.B. Gorepatil, Y.D. Mane, A.B. Gorepatil, M.V. Gaikwad, V.S. Ingle, Res. Chem. Intermed. 41(11), 8355 (2015)
M. Ghashang, Res. Chem. Intermed. 40(4), 1669 (2014)
M.F. Ding, C.P. Chen, S.T. Lin, J. Chin. Chem. Soc. 60(6), 645 (2013)
G.F. Chen, H.M. Jia, L.Y. Zhang, B.H. Chen, J.T. Li, Ultrason. Sonochem. 20(2), 627 (2013)
C. Praveen, A. Nandakumar, P. Dheenkumar, D. Muralidharan, P.T. Perumal, J. Chem. Sci. 124(3), 609 (2012)
A. Shokrolahi, A. Zali, M. Mahdavi, Phosphorus Sulfur Silicon Relat. Elem. 187(4), 535 (2012)
P. Bandyopadhyay, M. Sathe, G.K. Prasad, P. Sharma, M.P. Kaushik, J. Mol. Catal. A Chem. 341(1–2), 77 (2011)
Z. Wei, C. Xu, B. Li, Bioresour. Technol. 100(11), 2883 (2009)
P. Pongtonglor, E. Hoonnivathana, P. Limsuwan, S. Limsuwan, K. Naemchanthara, J. Appl. Sci. 11(21), 3659 (2011)
A. Montilla, M.D. del Castillo, M.L. Sanz, A. Olano, Food Chem. 90(4), 883 (2005)
A. Rajendran, C. Mansiya, Br. J. Environ. Clim. Change 1(2), 44 (2011)
S. Patil, S.D. Jadhav, M.B. Deshmukh, J. Chem. Sci. 125(4), 851 (2013)
E. Mosaddegh, A. Hassankhani, Chin. J. Catal. 35, 351 (2014)
A.V. Borhade, B.K. Uphade, D.R. Tope, J. Chem. Sci. 125(3), 583 (2013)
A.V. Borhade, B.K. Uphade, D.R. Tope, Res. Chem. Intermed. 40(1), 211 (2014)
A.V. Borhade, B.K. Uphade, A.G. Gadhave, Res. Chem. Intermed. 41(3), 1447 (2015)
A.V. Borhade, B.K. Uphade, J. Iran. Chem. Soc. 12(6), 1107 (2015)
A.V. Borhade, B.K. Uphade, Iran. J. Catal. 6(2), (2016)
P.K. Dash, Y. Balto, Res. J. Nanosci. Nanotechnol. 1(1), 25 (2011)
Acknowledgments
The authors are thankful to UGC, New Delhi for the financial assistance as well as use of the Sophisticated Analytical Instrument Facility (SAIF), Panjab University, Chandigarh and Savitribai Phule Pune University for providing spectral analysis facilities. The authors are thankful for Principal, Padmashri Vikhe Patil College, Pravaranagar providing necessary laboratory facilities.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Borhade, A.V., Uphade, B.K. & Gadhave, A.G. Calcinized eggshell: an environmentally benign green catalyst for synthesis of 2-arylbenzothiazole derivatives. Res Chem Intermed 42, 6301–6311 (2016). https://doi.org/10.1007/s11164-016-2463-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-016-2463-5