Abstract
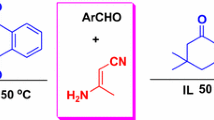
A facile and highly effective one-pot synthesis of polyhydroquinoline derivatives is reported via Hantzsch condensation of aldehydes, 1,3-dicarbonyl compounds, ethyl acetoacetate, and ammonium acetate in the presence of 1,3-disulfonic acid imidazolium hydrogen sulfate as an efficient and recyclable ionic liquid under solvent-free conditions. This methodology offers several advantages such as short reaction times, high yields, environmentally benign, mild reaction conditions, simple work-up procedure, and reusability of the ionic liquid.



Similar content being viewed by others
References
G. Guillena, D.J. Ramon, M. Yus, Tetrahedron Asymmetry 18, 693 (2007)
A. Domling, I. Ugi, Angew. Chem. Int. Ed. 39, 3168 (2000)
J. Zhu, H. Bienayme (eds.), Multicomponent Reactions (Wiley, Weinheim, 2005)
I. Ugi, Pure Appl. Chem. 73, 187 (2001)
C.C.A. Cariou, G.J. Clarkson, M. Shipman, J. Org. Chem. 73, 9762 (2008)
L. Yu, B. Chen, X. Huang, Tetrahedron Lett. 48, 925 (2007)
P.N. Kalaria, S.P. Satasia, D.K. Raval, Eur. J. Med. Chem. 78, 207 (2014)
S.C. Karad, V.B. Purohit, D.K. Raval, P.N. Kalaria, J.R. Avalani, P. Thakor, V.R. Thakkar, RSC Adv. 5, 16000 (2015)
P.P. Mager, R.A. Coburn, A.J. Solo, D.J. Triggle, H. Rothe, Drug Des. Discov. 8, 273 (1992)
A. Sausins, G. Duburs, Heterocycles 27, 269 (1988)
R. Manmhold, B. Jablonka, W. Voigdt, K. Schoenafinger, E. Schraven, J. Med. Chem. 27, 229 (1992)
A.C. Gaudio, A. Korokovas, Y. Takahata, J. Pharm. Sci. 83, 1110 (1994)
D.J. Triggle, in Comprehensive Medicinal Chemistry, vol. II, ed. by J.C. Emmet (Pergamon, Oxford, 1990)
V. Klusa, Drugs Future 20, 135 (1995)
R.G. Bretzel, C.C. Bollen, E. Maeser, K.F. Federlin, Am. J. Kidney Dis. 21, 53 (1993)
R.G. Bretzel, C.C. Bollen, E. Maeser, Drugs Future 17, 465 (1992)
R. Boer, V. Gekeler, Drugs Future 20, 499 (1995)
Z. Zarnegar, J. Safari, Z. Mansouri, Kafroudi. Catal. Commun. 59, 216 (2015)
V.K. Ahluwalia, B. Goyal, U. Das, J. Chem. Res. (s) 7, 266 (1997)
S. Margarita, O. Estael, V. Yamila, P. Beatriz, M. Lourdes, M. Nazario, Q. Margarita, S. Carlos, L.S. Jose, N. Hector, B. Norbert, M.P. Oswald, Tetrahedron 55, 875 (1999)
A. Hantzsch, Ann. Chem. 1, 215 (1882)
G. Sabitha, G.S.K.K. Reddy, C.S. Reddy, J.S. Yadav, Tetrahedron Lett. 44, 4129 (2003)
N.N. Karade, V.H. Budhewar, S.V. Shinde, W.N. Jadhav, Lett. Org. Chem. 4, 16 (2007)
A. Dondoni, A. Massi, E. Minghini, V. Bertolasi, Tetrahedron 60, 2311 (2004)
L.M. Wang, J. Sheng, L. Zhang, J.W. Han, Z. Fan, H. Tian, C.T. Qian, Tetrahedron 61, 1539 (2005)
M. Maheswara, V. Siddaiah, G.L. Damu, C.V. Rao, ARKIVOC ii, 201 (2006)
M.M. Heravi, K. Bakhtiri, N.M. Javadi, F.F. Bamoharram, M. Saeedi, H.A. Oskooi, J. Mol. Catal. A Chem. 264, 50 (2007)
S. Ko, C.F. Yao, Tetrahedron 62, 7293 (2006)
S.R. Cherkupally, R. Mekala, Chem. Pharm. Bull. 56, 1002 (2008)
G. Babu, P.T. Perumal, Aldrichimica Acta 33, 16 (2000)
S. Ko, M.N.V. Sastry, C. Lin, C.F. Yao, Tetrahedron Lett. 46, 5771 (2005)
F. Shirini, S.V. Atghia, M.A. Khoshdel, Iran. J. Catal. 1, 93 (2011)
A. Kumar, R.A. Maurya, Tetrahedron Lett. 48, 3887 (2007)
S. Kumar, P. Sharma, K.K. Kapoor, M.S. Hundal, Tetrahedron 64, 536 (2008)
J.B. Sainani, A.C. Shah, V.P. Aray, Indian J. Chem. 33B, 526 (1994)
N.K. Ladani, D.C. Mungra, M.P. Patel, R.G. Patel, Chin. Chem. Lett. 22, 1407 (2011)
A. Kumar, R.A. Maurya, Synlett 6, 883 (2008)
P.T. Anastas, J.C. Warner, Green Chemistry: Theory and Practice (Oxford University Press, Oxford, 1998)
M. Poliakoff, M.J. Fitzpatric, T.R. Farren, P.T. Anastas, Green Chemistry: Science and Politics of change. Science 297, 807 (2002)
I.T. Horváth, P.T. Anastas, Innovations and green chemistry. Chem. Rev. 107, 2169 (2007)
Z. Chen, Q. Zhu, W. Su, Tetrahedron Lett. 52, 2601 (2011)
H.G.O. Alvim, G.A. Bataglion, L.M. Ramos, A.L. de Oliveira, H.C.B. de Oliveira, M.N. Eberlin, J.L. de Macedo, W.A. da Silva, B.A.D. Neto, Tetrahedron 70, 3306 (2014)
H.G.O. Alvim, T.B. de Lima, H.C.B. de Oliveira, F.C. Gozzo, J.L. de Macedo, P.V. Abdelnur, W.A. Silva, B.A.D. Neto, ACS Catal. 3, 1420 (2013)
R. Fu, Y. Yang, W. Lai, Y. Ma, Z. Chen, J. Zhou, W. Chai, Q. Wang, R. Yuan, Synth. Commun. 45, 467 (2015)
J. Akbari, A. Heydari, H.R. Kalhor, S.A. Kohan, J. Comb. Chem. 12, 137 (2010)
F. Shirini, N.G. Khaligh, S. Akbari-Dadamahaleh, J. Mol. Catal. A Chem. 365, 15 (2012)
F. Shirini, N.G. Khaligh, J. Mol. Liq. 177, 386 (2013)
F. Shirini, A. Yahyazadeh, K. Mohammadi, Chin. Chem. Lett. 25, 341 (2014)
M. Tajbakhsh, H. Alinezhad, M. Norouzi, S. Baghery, M. Akbari, J. Mol. Liq. 177, 44 (2013)
M. Nasr-Esfahani, T. Abdizadeh, Orient. J. Chem. 28, 1249 (2012)
X.L. Zhang, S.R. Sheng, X.L. Liu, X.L. Liu, ARKIVOC xiii, 79 (2007)
M. Hong, C. Cai, W.B. Yi, J. Fluorine Chem. 131, 111 (2010)
A. Khojastehnezhad, F. Moeinpour, A. Davoodnia, Chin. Chem. Lett. 22, 807 (2011)
A. Davoodnia, M. Khashi, N. Tavakoli-Hoseini, Chin. J. Catal. 34, 1173 (2013)
C.S. Reddy, M. Raghu, Chin. Chem. Lett. 19, 775 (2008)
Acknowledgments
The authors acknowledge financial support for this work from the Iran National Science Foundation (INSF) (Grant Number 93014856) and partial support of this work by the Research Council of the University of Guilan. F. Shirini and K. Mohammadi also wish to thank the Sobhandarou Company for the partial facilities.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Mohammadi, K., Shirini, F. & Yahyazadeh, A. 1, 3-Disulfonic acid imidazolium hydrogen sulphate as an efficient and reusable ionic liquid for the multicomponent synthesis of polyhydroquinoline derivatives under solvent-free conditions. Res Chem Intermed 42, 2047–2054 (2016). https://doi.org/10.1007/s11164-015-2134-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-015-2134-y