Abstract
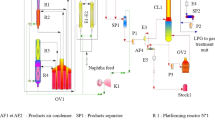
In this work, we consider the study of a heterogeneous model for a spherical fixed bed axial flow reactor. This study is applied to a naphtha reforming process. This process is used in the naphtha Magnaforming unit of the Skikda refinery in Algeria. This unit consists of four spherical-shaped reactors, of different sizes, garnished with bi-functional catalyst (Pt–Re/Al2O3) and placed in series. The aim is to enhance the performance challenge in this reforming process. We shall be interested in the development of a heterogeneous mathematical model that reproduces, as best possible, the process in its beginning cycle. The results obtained from the simulation of the model were compared with the industrial data and good agreement was found. A significant increase in the performance of the aromatics, hydrogen and light products (fuel gas and LPG) has been observed when using an egg-yolk distribution in the first reactor, an egg white in the second reactor and eggshell distribution for the metallic catalyst sites while the acidic site distribution was kept uniform. With this choice, it turns out that the use of the fourth reactor is not necessary. The same result was obtained when we treated the case of cylindrical reactors in a previous work.





Similar content being viewed by others
References
Ciapetta F, Wallace D (1972) Catalytic naphtha reforming. Catal Rev 5:158–167
Antos GJ (1976) US Patent 4,032,587
George JA, Abdullah MA (2004) Catalytic naphtha reforming. Marcel Dekker, New York
Antos GJ, Aitani AM, Parera JM (1995) Catalytic naphtha reforming. Marcel Dekker Inc, New York
Benitez VM, Pieck CL (2010) Influence of indium content on the properties of Pt–Re/ Al2O3 naphtha reforming catalysts. Catal Lett 136:45–51
Viviana Benitez MB, Mazzieri VA, Especel C, Epron F, Vera CR, Marécot P, Boutzeloit M, Pieck CL (2007) Preparation of trimetallic Pt–Re–Ge/Al2O3 and Pt–Ir–Ge/Al2O3 naphtha reforming catalysts by surface redox reaction. Appl Catal A 319:210–217
Pieck CL, Sad MR, Parera JM (1996) Chlorination of Pt–Re/A12O3 during naphtha reforming. J Chem Technol Biotechnol 67:61–67
Hartig F, Keil FJ (1993) Large-scale spherical fixed bed reactors: modeling and optimization. Ind Eng Chem Res 32:424–437
Hlavacek V, Kubicek M (1972) Modeling of chemical reactors—XXV cylindrical and spherical reaction with radial flow. Chem Eng Sci 27:177–186
Malkin AY, Ivanova AN, Ivanova SL, Andrianova ZS (1978) Non isothermal polymerization in a spherical reactor. Temperature distribution and reaction kinetics. J Eng Phys Thermophys 34:426–430
Streeter VL, Bedford KW (1998) Fluid mechanics. WCB McGraw-Hill, Inc., Boston
Rahimpour MR, Iranshahi D, Pourazadi E, Paymooni K (2011) Evaluation of optimum design parameters and operating conditions of axial- and radial-flow tubular naphtha reforming reactors, using the differential evolution method, considering catalyst deactivation. Energy Fuels 25:762–472
Boukezoula TF, Bencheikh L, Belkhiat DEC (2020) A heterogeneous model for a cylindrical fixed bed axial flow reactors applied to a naphtha reforming process with a non-uniform catalyst distribution in the pellet. Reac Kinet Mech Cat 131:335–351. https://doi.org/10.1007/s11144-020-01851-3
Smith R (1959) Kinetic analysis of naphtha reforming with platinum catalyst. Chem Eng Prog 55:76–80
McCallister KR, O’Neal TP (1971) Patent French, 2,078,056, UOP
Sinfelt JH (1976) Patent US, 3,953,368, Exxon
Morbidelli M, Gavriilidis A, Varma A (2011) Catalyst design: Optimal distribution of catalyst in pellets, reactors and membranes. Cambridge University Press, Cambridge
Boukezoula TF, Bencheikh L (2018) Theoretical investigation of non-uniform bifunctional catalyst for the aromatization of methyl cyclopentane. Reac Kinet Mech Cat 124:15–25
Scelza OA, Miguel SR, Baronetti GT, Castro AA (1987) Performance of Pt–Re/Al2O3 Catalysts different radial distribution profiles. React, Kinet, Catal, Lett 33(1):143–148
Iranshahi D, Pourazadi E, Paymooni K, Rahimpour MR (2011) A novel dynamic membrane reactor concept with radial-flow pattern for reacting material and axial-flow pattern for sweeping gas in catalytic naphtha reformers. AIChE 58:1230–1247
Iranshahi D, Pourazadi E, Paymooni K, Rahimpour MR (2011) Enhancement of aromatic production in naphtha reforming process by simultaneous operation of isothermal and adiabatic reactors. Int J Hydrogen Energy 36:2076–2085
Khosravanipour Mostafazadeh A, Rahimpour MR (2009) A membrane catalytic bed concept for naphtha reforming in the presence of catalyst deactivation. Chem Eng Process Intensif 48:683–694
Rahimpour MR (2009) Enhancement of hydrogen production in a novel fluidized-bed membrane reactor for naphtha reforming. Int J Hydrogen Energy 34:2235–2251
Iranshahi D, Pourazadi E, Bahmanpour AM, Rahimpour MR (2011) A comparison of two different flow types on performance of a thermally coupled recuperative reactor containing naphtha reforming process and hydrogenation of nitrobenzene. Int J Hydrogen Energy 36:3483–3495
Iranshahi D, Bahmanpour AM, Pourazadi E, Rahimpour MR (2010) Mathematical modeling of a multi-stage naphtha reforming process using novel thermally coupled recuperative reactors to enhance aromatic production. Int J Hydrogen Energy 35:10984–10993
Meidanshahi V, Bahmanpour AM, Iranshahi D, Rahimpour MR (2011) Theoretical investigation of aromatics production enhancement in thermal coupling of naphtha reforming and hydrodealkylation of toluene. Chem Eng Process Intensif 50:893–903
Rahimpour MR, Vakili R, Pourazadi E, Bahmanpour AM, Iranshahi D (2011) Enhancement of hydrogen production via coupling of MCH dehydrogenation reaction and methanol synthesis process by using thermally coupled heat exchanger reactor. Int J Hydrogen Energy 36:3371–3383
Iranshahi D, Pourazadi E, Paymooni K, Rahimpour MR (2012) Utilizing DE optimization approach to boost hydrogen and octane number in a novel radial-flow assisted membrane naphtha reactor. Chem Eng Sci 68:236–249
Rahimpour MR, Abbasloo A, Sayyad Amin J (2008) A novel radial-flow, spherical-bed reactor concept for methanol synthesis in the presence of catalyst deactivation. Chem Eng Technol 31:1615–1629
Rahimpour MR, Iranshahi D, Bahmanpour AM (2010) Dynamic optimization of a multi-stage spherical, radial flow reactor for the naphtha reforming process in the presence of catalyst deactivation using differential evolution (DE) method. Int J Hydrogen Energy 35:7498–7511
Iranshahi D, Rahimpour MR, Asgari A (2010) A novel dynamic radial-flow, spherical-bed reactor concept for naphtha reforming in the presence of catalyst deactivation. Int J Hydrogen Energy 35:6261–6275
Iranshahi D, Pourazadi E, Paymooni K, Bahmanpour AM, Rahimpour MR, Shariati A (2010) Modeling of an axial flow, spherical packed-bed reactor for naphtha reforming process in the presence of the catalyst deactivation. Int J Hydrogen Energy 35:12784–12799
Iranshahi D, Paymooni K, Pourazadi EK, Rahimpour MR (2012) Enhancement in research octane number and hydrogen production via dynamic optimization of a novel spherical axial-flow membrane naphtha reformer. Ind Eng Chem Res 51:398–409
Pourazadi E, Vakili R, Iranshahi D, Jahanmiri A, Rahimpour MR (2013) Optimal design of a thermally coupled fluidized bed heat exchanger reactor for hydrogen production and octane improvement in the catalytic naphtha reformers. Can J Chem Eng 91:54–65
Fathi J, Rahimpour MR (1992) Sensitivity of catalytic naphtha reformers to different parameters. Iran J Chem Technol 16:57–67
Rahimpour MR, Iranshahi D, Pourazadi E, Bahmanpour AM (2011) A comparative study on a novel combination of spherical and membrane tubular reactors of the catalytic naphtha reforming process. Int J Hydrogen Energy 36:505–551
Boukezoula TF, Bencheikh L (2018) Industrial analysis of catalytic reforming reactor. Rev Roum Chim 63(3):181–187
Liang KM, Guo HY, Pan SW (2005) A study on naphtha catalytic reforming reactor simulation and analysis. J Zhejiang Univ Sci 6B:590–596
Pourazadia E, Iranshahia D, Rahimpour MR, Jahanmiri A (2012) Incorporating multi membrane tubes for simultaneous management of H2/HC and hydrogenation of nitrobenzene to aniline in naphtha heat exchanger reactor. Chem Eng J 184:286–297
Lid T, Skogestad S (2008) Data reconciliation and optimal operation of a catalytic naphtha reformer. J Process Control 18:320–331
Askari A, Karimi H, Rahimi MR, Ghanbari M (2012) Simulation and modeling of catalytic reforming process. Pet Coal 54:76–84
Talaghat MR, Roosta A, Khosrozadeh I (2017) A novel study of upgrading catalytic reforming unit by improving catalyst regeneration process to enhance aromatic compounds, hydrogen production and hydrogen purity. Journal of Chemical and Petroleum Engineering 51(2):81–94
Rase HF (1977) Chemical reactor design for process plants, vol 2. Wiley, New York
Zahedi S, Farsi M, Rahimpour MR (2021) Modeling and optimization of heavy naphtha reforming on bifunctional Pt–Re/Al2O3 catalyst. Top Catal. https://doi.org/10.1007/s11244-021-01510-4
Zaidoon MS, Adnan AA, Khalid AS (2020) A detailed reaction kinetic model of heavy naphtha reforming. Arab J Sci Eng 45:7361–7370
Ivanchina E, Chernyakova E, Pchelintseva I, Poluboyartsev D (2021) Mathematical modeling and optimization of semi-regenerative catalytic reforming of naphtha. Oil Gas Sci Technol Rev IFP Energies nouvelles 76:64
Ebrahimian S, Iranshahi D (2019) Modeling and optimization of thermally coupled reactors of naphtha reforming and propane ammoxidation with different feed distributions. Reac Kinet Mech Cat 129(4):315–335
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Boukezoula, T.F., Bencheikh, L. & Belkhiat, D.E.C. A heterogeneous mathematical model for a spherical fixed bed axial flow reactor applied to a naphtha reforming process: enhancing performance challenge using a non-uniform catalyst distribution in the pellet. Reac Kinet Mech Cat 135, 2323–2340 (2022). https://doi.org/10.1007/s11144-022-02257-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11144-022-02257-z