Abstract
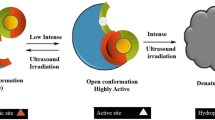
In this work, the impact of ultrasound on dextransucrase activity was investigated, and a biphasic model was introduced to investigate the inactivation kinetics of the dextransucrase catalytic reaction. In addition, a chemical kinetic model, the Michaelis–Menten equation, Eyring transition state theory and the Arrhenius equation were applied. Short periods of exposure to low-intensity ultrasound significantly increased the activity of dextransucrase. The maximum activity was 1.25 ± 0.01 u/mL at an ultrasound intensity of 1.8 W/cm3 for 3 min, which was 27.6% higher than the activity of the untreated enzyme (0.98 ± 0.038 U/mL). In addition, after the ultrasound treatment, the kinetic model of the dextransucrase catalytic reaction was fitted to a first-order kinetic model at low sucrose concentrations. At low sucrose concentrations, the Ea, ΔH, ΔS and ΔG of ultrasound-treated dextransucrase were reduced by 33.7, 35.7, 28.8 and 0.38%, respectively. Furthermore, compared with the parameters of the untreated sample, the value of Vmax the treated sample increased, and Km decreased. The thermal inactivation model of the dextransucrase catalytic reaction in a shaker was fitted to a biphasic model, and the percentage of the thermally stable fraction of ultrasound-treated dextransucrase was increased, while the rate of deactivation was decreased. Moreover, the degree of thermal inactivation was inversely proportional to the dextransucrase concentration. The fluorescence spectra showed a decrease in the amount of tryptophan on the surface of dextransucrase treated with ultrasound. In conclusion, short-term treatment with low ultrasound intensity increases the activity, thermal stability and reaction rate of dextransucrase.









Similar content being viewed by others
References
Lata K, Sharma M, Patel SN, Sangwan RS, Singh SP (2018) Bioprocess Biosyst Eng 41(8):1121–1131
Côté GL, Skory CD (2016) Carbohydr Res 428:57–61
Shi Q, Hou Y, Juvonen M, Tuomainen P, Kajala I, Shukla S, Goyal A, Maaheimo H, Katina K, Tenkanen M (2016) J Agric Food Chem 64(16):3276–3286
Sorndech W, Nakorn KN, Tongta S, Blennow A (2018) LWT 95:135–142
Zhang Y, Guo L, Xu D, Li D, Yang N, Chen F, Jin Z, Xu X (2018) Food Chem 256:373
Wang DL, Yan LF, Ma XB, Wang WJ, Zou MM, Zhong JJ, Ding T, Ye XQ, Liu DH (2018) Int J Biol Macromol 119:453–461
Li MQ, Zhang HB, Li Y, Hu XQ, Yang JW (2017) Int J Biol Macromol 107(Pt B):1641–1649
Chtourou M, Lahyani A, Trabelsi M (2018) Reac Kinet Mech Cat 126:237–247
Hou FR, Ma XB, Fan LH, Wang DL, Wang WJ, Ding T, Ye XQ, Liu DH (2019) Food Chem 285:355–362
Zhang ZH, Wang LH, Zeng XA, Han Z, Brennan CS (2019) Int J Food Sci Technol 54(1):1–13
Subhedar PB, Gogate PR (2014) J Mol Catal B 101:108–114
Dalagnol LMG, Silveira VCC, Silva HBD, Manfroi V, Rodrigues RC (2017) Process Biochem 61:S1359511317309480
Frampton JP, Lai D, Lounds M, Chung K, Kim J, Mansfield JF, Takayama S (2015) Adv Healthcare Mater 4(2):313–319
Hou DZ, Pu YY, Zou QS, Chen HL, Yu Y, Huang SX, Chen S (2018) Sugar Technol 20(1):60–68
Yamaner CI, Sezen IY, Tanriseven A (2010) Food Sci Biotechnol 19(1):175–184
Silvestri MG, Dills CE (1989) J Chem Educ 66(66):690
Knesebeck AM, Pacheco Ortiz RW, Cardozo-Filho L, Zanoelo EF (2018) Reac Kinet Mech Cat 125(2):521–534
Liing AC, Lund DB (2010) J Food Sci 43(4):1307–1310
Lente G (2015) Deterministic kinetics in chemistry and systems biology: the dynamics of complex reaction networks. Springer, New York
Szabó OE, Csiszár E (2013) Carbohydr Polym 98(2):1483–1489
Bashari M, Eibaid A, Wang J, Tian Y, Xu X, Jin Z (2013) Ultrason Sonochem 20(1):155–161
Terefe NS, Buckow R, Versteeg C (2015) Crit Rev Food Sci Nutr 55(2):147–158
Ma XB, Wang WJ, Zou MM, Ding T, Ye XQ, Liu DH (2015) RSC Adv 5(130):107591–107600
Jadhav SH, Gogate PR (2014) Ind Eng Chem Res 53(4):1377–1385
Pu Y, Zou Q, Hou D, Zhang Y, Chen S (2017) Carbohydr Polym 156:71–76
Lente G (2018) Curr Opin Chem Eng 21:76–83
Zhu LJ, Zhu LH, Murtaza A, Liu Y, Liu SY, Li JJ, Iqbal A, Xu XY, Pan SY, Hu WF (2019) Molecules 24(10):1922
de Souza SA, Augusto PED (2019) Júnior BRDCL, Nogueira CA, Vieira ÉNR, de Barros FAR, Stringheta PC, Ramos AM. LWT 107:164–170
Ma H, Huang L, Jia J, He R, Zhu W (2011) Ultrason Sonochem 18(1):419–424
Jian J, Haile M, Wenjuan Q, Kai W, Cunshan Z, Ronghai H, Lin L, John O (2015) Ultrason Sonochem 27:46–53
Wenjuan Q, Haile M, Bin L, Ronghai H, Zhongli P, Ernest Ekow A (2013) Ultrason Sonochem 20(6):1408–1413
Marguerite D, Magali RS, Rene MW, Michel V, Pierre FM (1997) Appl Biochem Biotechnol 62(1):47–59
Neely BW (1959) J Am Chem Soc 81(16):4416–4418
Imai M, Ikari K, Suzuki I (2004) Biochem Eng J 17(2):79–83
Li H, Li S, Peng J, Srinivasakannan C, Zhang L, Yin S (2018) Ultrason Sonochem 40(Pt A):1021–1030
Song W, De La Cruz AA, Rein K, O'Shea KE (2006) Environ Sci Technol 40(12):3941–3946
Mawson R, Gamage M, Terefe NS, Kai K (2011) Ultrasound in enzyme activation and inactivation. Springer, New York
Ebert KH, Patat F (1963) Zeitschrift für Naturforschung B 17(11):738–748
Cybulska J, Pakula R (1963) Exp Med Microbiol 15:285–296
Mooser G, Shur D, Lyou M, Watanabe C (1985) J Biol Chem 260(11):6907–6915
Shalini GR, Shivhare US, Basu S (2008) J Food Eng 85:147–153
Ladero M, Santos A, García-Ochoa F (2006) Enzyme Microb Technol 38(1):1–9
Lencki RW, Arul J, Neufeld RJ (2010) Biotechnol Bioeng 40(11):1427–1434
Bhagia S, Wyman CE, Kumar R (2019) Biotechnol Biofuels 12(1):1–15
Donaldson TL, Boonstra EF, Hammond JM (1980) J Colloid Interface Sci 74(2):441–450
Miao J, Liao W, Kang M, Jia Y, Wang Q, Duan S, Xiao S, Cao Y, Ji H (2018) Food Funct 9(12):6578–6586
Sluzky V, Klibanov AM, Langer R (2010) Biotechnol Bioeng 40(8):895–903
Lencki RW, Tecante A, Choplin L (2010) Biotechnol Bioeng 42(9):1061–1067
Ortega N, Diego SD, Perez-Mateos M, Busto MD (2004) Food Chem 88(2):209–217
Lecker DN, Khan A (1996) Biotechnol Progr 12(5):713–717
Casey EJ, Laidler KJ (1951) J Am Chem Soc 73(4):1455–1457
Miller WA (1985) Purification, surfactant stabilization, molecular weight studies, and divalent metal ion kinetics of dextransucrase from Leuconostoc mesenteroides NRRL B-512F. Retrospective Theses and Dissertations. https://doi.org/10.31274/rtd-180813-8707
Yi J, Jiang B, Zhang Z, Liao X, Zhang Y, Hu X (2012) J Agric Food Chem 60(2):593–599
Zou Y, Yang H, Zhang M, Zhang X, Xu W, Wang D (2019) Asian-Australas J Anim Sci 32:1611
Acknowledgements
The authors express their thanks to the Engineering Center for Sugar and Comprehensive Utilization. This work was supported by financial aid from the National Natural Science Foundation of China (Grant No. 21768001).
Funding
This study was funded by the National Natural Science Foundation of China (Grant No. 21768001).
Author information
Authors and Affiliations
Contributions
Conceptualization, formal analysis, methodology, software and writing—original draft were performed by Weibing Lan. Data curation, project administration, resources, writing—review & editing were performed by Shan Chen. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lan, W., Chen, S. Chemical kinetics, thermodynamics and inactivation kinetics of dextransucrase activity by ultrasound treatment. Reac Kinet Mech Cat 129, 843–864 (2020). https://doi.org/10.1007/s11144-020-01728-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11144-020-01728-5