Abstract
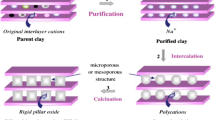
Al-pillared clays from local available bentonite were synthesized via two methods by the application of conventional and ultrasonic treatments during the intercalation stage. The results of XRD and N2 adsorption indicated an increase in the basal spacing, the specific BET surface area, and porosity subsequent to pillaring by both methods with a considerable shortening of the intercalation process for the ultrasonic method. Al-pillared bentonites were used as supports to prepare manganese oxide catalysts by the wet impregnation of 2 and 10 wt% of Mn. The presence of MnO2 and Mn2O3 were detected by X-ray diffraction and confirmed by temperature-programmed reduction for the higher loading catalyst with 10 wt% of Mn. The catalytic activity of our catalysts was evaluated in the CO oxidation reaction. According to our results, the concentration of the Mn seems to be the factor that decisively affects the catalytic activity at the expense of the preparation method.





Similar content being viewed by others
References
Royer S, Duprez D (2011) Catalytic oxidation of carbon monoxide over transition metal oxides. ChemCatChem 3:24–65
Gullotta F, Di Masi A, Ascenzi P (2012) Carbon monoxide: an unusual drug. IUBMB Life 64:378–386
Raub JA, Mathieu-Nolf M, Hampson NB, Thom SR (2000) Carbon monoxide poisoning—a public health perspective. Toxicology 145:1–14
Prockop LD, Chichkova RI (2007) Carbon monoxide intoxication: an updated review. J Neurol Sci 262:122–130
Raub JA, Benignus VA (2002) Carbon monoxide and the nervous system. Neurosci Biobehav Rev 26:925–940
Lawther PJ (1975) Carbon monoxide. Br Med Bull 31:256–260
Bateman DN (2012) Carbon monoxide. Medicine (Baltimore) 40:115–116
Biabani-Ravandi A, Rezaei M, Fattah Z (2013) Low-temperature CO oxidation over nanosized Fe–Co mixed oxide catalysts: effect of calcination temperature and operational conditions. Chem Eng Sci 94:237–244
Biabani-Ravandi A, Rezaei M (2012) Low temperature CO oxidation over Fe–Co mixed oxide nanocatalysts. Chem Eng J 184:141–146
Taylor SH, Rhodes C (2006) The oxidation of carbon monoxide at ambient temperature over mixed copper–silver oxide catalysts. Catal Today 114:357–361
Abdel Halim KS, Khedr MH, Nasr MI, El-Mansy AM (2007) Factors affecting CO oxidation over nanosized Fe2O3. Mater Res Bull 42:731–741
Gac W (2007) The influence of silver on the structural, redox and catalytic properties of the cryptomelane-type manganese oxides in the low-temperature CO oxidation reaction. Appl Catal B 75:107–117
Figueras F (1988) Pillared clays as catalysts. Catal Rev 30:457–499
Bergaya F, Aouad A, Mandalia T (2006) Chapter 7.5 pillared clays and clay minerals. Dev Clay Sci 1:393–421
Clearfield A (1996) Chapter 14—preparation of pillared clays and their catalytic properties. In: Moser WR (ed) Advances catalysis nanostructered material. Academic Press, San Diego, pp 345–394
Ding Z, Kloprogge JT, Frost RL (2001) Porous clays and pillared clays-based catalysts. Part 2: a review of the catalytic and molecular sieve applications. J Porous Mater 8:273–293
Gil A, Korili SA, Vicente MA (2008) Recent advances in the control and characterization of the porous structure of pillared clay catalysts. Catal Rev 50:153–221
Gil A, Gandía LM, Vicente MA (2000) Recent advances in the synthesis and catalytic applications of pillared clays. Catal Rev 42:145–212
Herling MM, Breu J (2014) The largely unknown class of microporous hybrid materials: clays pillared by molecules. Zeitschrift für Anorg und Allg Chemie 640:547–560
Kloprogge JT (1998) Synthesis of smectites and porous pillared clay catalysts: a review. J Porous Mater 5:5–41
Poncelet G, Fripiat JJ (2001) In: Smith J (ed) Handbook of Heterogeneous Catalysis, 2nd edn. Wiley, New York
Pérez A, Centeno MA, Odriozola JA, Molina R, Moreno S (2008) The effect of ultrasound in the synthesis of clays used as catalysts in oxidation reactions. Catal Today 133–135:526–529
Olaya A, Blanco G, Bernal S, Moreno S, Molina R (2009) Synthesis of pillared clays with Al–Fe and Al–Fe–Ce starting from concentrated suspensions of clay using microwaves or ultrasound, and their catalytic activity in the phenol oxidation reaction. Appl Catal B 93:56–65
Tomul F (2011) Effect of ultrasound on the structural and textural properties of copper-impregnated cerium-modified zirconium-pillared bentonite. Appl Surf Sci 258:1836–1848
Carriazo JG, Martínez LM, Odriozola JA, Moreno S, Molina R, Centento MA (2007) Gold supported on Fe, Ce, and Al pillared bentonites for CO oxidation reaction. Appl Catal B 72:157–165
Álvarez A, Moreno S, Molina R, Ivanova S, Centeno MA, Odriozola JA (2012) Gold supported on pillared clays for CO oxidation reaction: effect of the clay aggregate size. Appl Clay Sci 69:22–29
Martínez TLM, Domínguez MI, Sanabria N, Hernández WY, Moreno S, Molina R, Odriozola JA, Centeno MA (2009) Deposition of Al–Fe pillared bentonites and gold supported Al–Fe pillared bentonites on metallic monoliths for catalytic oxidation reactions. Appl Catal A 364:166–173
Xu Z, Inumaru K, Yamanaka S (2001) Catalytic properties of metal loaded silica-pillared manganese titanate for CO oxidation. Appl Catal A 210:217–224
Vicente MA, Belver C, Trujillano R, Rives V, Álvarez AC, Lambert JF, Korili SA, Gandia LM, Gil A (2004) Preparation and characterisation of Mn- and Co-supported catalysts derived from Al-pillared clays and Mn- and Co-complexes. Appl Catal A 267:47–58
Gandia LM, Vicente MA, Gil A (2000) Preparation and characterization of manganese oxide catalysts supported on alumina and zirconia-pillared clays. Appl Catal A 196:281–292
Li J, Li L, Wu F, Zhang L, Liu X (2013) Dispersion–precipitation synthesis of nanorod Mn3O4 with high reducibility and the catalytic complete oxidation of air pollutants. Catal Commun 31:52–56
Tian H, He J, Liu L, Wang D, Hao Z, Ma C (2012) Highly active manganese oxide catalysts for low-temperature oxidation of formaldehyde. Microporous Mesoporous Mater 151:397–402
Liu Y, Luo M, Wei Z, Xin Q, Ying P, Li C (2001) Catalytic oxidation of chlorobenzene on supported manganese oxide catalysts. Appl Catal B 29:61–67
Morales MR, Barbero BP, Cadús LE (2007) Combustion of volatile organic compounds on manganese iron or nickel mixed oxide catalysts. Appl Catal B 74:1–10
Pozan GS (2012) Effect of support on the catalytic activity of manganese oxide catalyts for toluene combustion. J Hazard Mater 221–222:124–130
Reynolds RC (1985) Newmod, a computer program for the calculation of one dimensional X ray diffraction patterns of mixed-layered clays. RC Reynolds 8:57–62
Deboer J (1964) Studies on pore systems in catalysts IV. The two causes of reversible hysteresis. J Catal 3:268–273
Katdare SP, Ramaswamy V, Ramaswamy AV (1999) Ultrasonication: a competitive method of intercalation for the preparation of alumina pillared montmorillonite catalyst. Catal Today 49:313–320
Katdare SP, Ramaswamy V, Ramaswamy AV (2000) Factors affecting the preparation of alumina pillared montmorillonite employing ultrasonics. Microporous Mesoporous Mater 37:329–336
Sanabria NR, Molina R, Moreno S (2009) Effect of ultrasound on the structural and textural properties of Al–Fe pillared clays in a concentrated medium. Catal Lett 130:664–671
Reed C, Lee YK, Oyama ST (2006) Structure and oxidation state of silica-supported manganese oxide catalysts and reactivity for acetone oxidation using ozone. J Phys Chem B 110:20–67
Zuo S, Huang Q, Li J, Zhou R (2009) Promoting effect of Ce added to metal oxide supported on Al pillared clays for deep benzene oxidation. Appl Catal B 91:204–209
Kazin PE, Tret YD (2008) Synthesis of magnetoresistive glass—ceramic composites in the SrO–MnOx–SiO2–La2O3 system. Catal Lett 2:34–36
Craciun R (1998) Structure/activity correlation for unpromoted and CeO2-promoted. Catal Commun 55:25–31
Galeano LA, Gil A, Vicente MA (2011) Strategies for immobilization of manganese on expanded natural clays: catalytic activity in the CWPO of methyl orange. Appl Catal B 104:252–260
Sing KSW, Everett DH, Haul RAW, Moscou L, Pierotti RA, Rouquéol J, Siemieniewska T (1985) International union of pure commission on colloid and surface chemistry including catalysis * reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity. Pure Appl Chem 57:603–619
Gregg SJ, Sing KS (1982) Adsorption, surface area and porosity. J Electrochem Soc 114(11):279C–279C
Lowell S, Shields JE, Thomas MA, Thommes M (2005) Characterization of porous solids and powders: surface area, pore size, and density. springer, Berlin
Schüth F, Sing KSW, Weitkamp J (2002) Handbook of porous solids. Wiley, Weinheim
Leofanti G, Padovan M, Tozzola G, Venturelli B (1998) Surface area and pore texture of catalysts. Catal Today 41:207–219. doi:10.1016/S0920-5861(98)00050-9
Jozwiak WK, Kaczmarek E, Maniecki TP et al (2007) Reduction behavior of iron oxides in hydrogen and carbon monoxide atmospheres. Appl Catal A 326:17–27
Li J, Li L, Cheng W, Wu F, Lu X, Li Z (2014) Controlled synthesis of diverse manganese oxide-based catalysts for complete oxidation of toluene and carbon monoxide. Chem Eng J 244:59–67
Tomul F, Balci S (2009) Characterization of Al, Cr-pillared clays and CO oxidation. Appl Clay Sci 43:13–20
Sambeth JE, Thomas HJ (2013) Volatile organic compound removal over bentonite-supported Pt, Mn and Pt/Mn monolithic catalysts. Reac Kinet Mech Cat 108:443–458
Zaki MI, Hasan MA, Pasupulety L, Fouad NE (1999) CO and CH total oxidation over manganese oxide supported on ZrO, TiO, TiO–AlO and SiO–AlO catalysts. New J Chem 23:1197–1202
Venkataswamy P, Jampaiah D, Lin F, Alxneit I, Reddy BM (2015) Applied surface science structural properties of alumina supported Ce–Mn solid solutions and their markedly enhanced catalytic activity for CO oxidation. Appl Surf Sci 349:299–309
Acknowledgments
The authors thank the project MINECO/FEDER/Ref: MAT2013-40823-R for financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dhahri, M., Muñoz, M.A., Yeste, M.P. et al. Preparation of manganese-impregnated alumina-pillared bentonite, characterization and catalytic oxidation of CO. Reac Kinet Mech Cat 118, 655–668 (2016). https://doi.org/10.1007/s11144-016-1017-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11144-016-1017-6