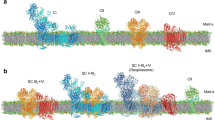
Abstract
Under aerobic conditions the production of Reactive Oxygen Species (ROS) by electron transport chains is unavoidable, and occurs in both autotrophic and heterotrophic organisms. In photosynthetic organisms both Photosystem II (PS II) and Photosystem I (PS I), in addition to the cytochrome b6/f complex, are demonstrated sources of ROS. All of these membrane protein complexes exhibit oxidative damage when isolated from field-grown plant material. An additional possible source of ROS in PS I and PS II is the distal, chlorophyll-containing light-harvesting array LHC II, which is present in both photosystems. These serve as possible sources of 1O2 produced by the interaction of 3O2 with 3chl* produced by intersystem crossing. We have hypothesized that amino acid residues close to the sites of ROS generation will be more susceptible to oxidative modification than distant residues. In this study, we have identified oxidized amino acid residues in a subset of the spinach LHC II proteins (Lhcb1 and Lhcb2) that were associated with either PS II membranes (i.e. BBYs) or PS I-LHC I-LHC II membranes, both of which were isolated from field-grown spinach. We identified oxidatively modified residues by high-resolution tandem mass spectrometry. Interestingly, two different patterns of oxidative modification were evident for the Lhcb1 and Lhcb2 proteins from these different sources. In the LHC II associated with PS II membranes, oxidized residues were identified to be located on the stromal surface of Lhcb1 and, to a much lesser extent, Lhcb2. Relatively few oxidized residues were identified as buried in the hydrophobic core of these proteins. The LHC II associated with PS I-LHC I-LHC II membranes, however, exhibited fewer surface-oxidized residues but, rather a large number of oxidative modifications buried in the hydrophobic core regions of both Lhcb1 and Lhcb2, adjacent to the chlorophyll prosthetic groups. These results appear to indicate that ROS, specifically 1O2, can modify the Lhcb proteins associated with both photosystems and that the LHC II associated with PS II membranes represent a different population from the LHC II associated with PS I-LHC I-LHC II membranes.





Similar content being viewed by others
Notes
Considering only one Lhcb1 and one Lhcb2 within the trimer.
For a very approximate estimate, consider an LHC II trimer which is not associated with a reaction center (for instance if it were moving between PS II and PS I during State 1 to State 2 transitions). Since each chl absorbs about 10 photons/sec (at 1800 μmoles photons/m2/sec) (Blankenship 2002) and each LHC II trimer contains 42 chl, each LHC II trimer absorbs about 420 photons/sec. If an LHC II trimer has a ½ life of 10 h, it would absorb 1.5 × 107 photons. Assuming the 3chl*-quenching mechanisms (carotenoids, NPQ and LHC II aggregation, etc.) were 99.99% effective at quenching 3chl* and that 10% of the unquenched 3chl* formed 1O2, then ~ 150 1O2 could be formed over the lifetime of the LHC II. LHC II trimers connected to reaction centers would be predicted to produce significantly less 1O2.
References
Alboresi A, Ballottari M, Hienerwadel R, Giacometti GM, Morosinotto T (2009) Antenna complexes protect photosystem I from photoinhibition. BMC Plant Biol 9:71
Anastassiou R, Argyroudi-Akoyunoglou JH (1995) Thylakoid-bound proteolytic activity against LHC II apoprotein in bean. Photosyn Res 43:241–250
Angelisová P, Ballek O, Sýkora J, Benada O, Čajka T, Pokorná J, Pinkas D, Hořejší V (2019) The use of styrene-maleic acid copolymer (SMA) for studies on T cell membrane rafts. Biochim Biophys Acta 1861:130–141
Anisimova M, Gascuel O (2006) Approximate likelihood-ratio test for branches: a fast, accurate, and powerful alternative. Syst Biol 55:539–552
Austin JR, Staehelin LA (2011) Three-dimensional architecture of grana and stroma thylakoids of higher plants as determined by electron tomography. Plant Physiol 155:1601–1611
Ballottari M, Girardon J, Dall’osto L, Bassi R (2012) Evolution and functional properties of photosystem II light-harvesting complexes in eukaryotes. Biochim Biophys Acta 1817:143–157
Bell AJ, Frankel LK, Bricker TM (2015) High yield non-detergent Isolation of photosystem I-light-harvesting chlorophyll II membranes from spinach thylakoids: implications for the organization of the PS I antennae in higher plants. J Biol Chem 290:18429–18437
Benson SL, Maheswaran P, Ware MA, Hunter CN, Horton P, Jansson S, Ruban AV, Johnson MP (2015) An intact light-harvesting complex I antenna system is required for complete state transitions in Arabidopsis. Nat Plants 1:15176
Berthold DA, Babcock GT, Yocum CF (1981) A highly resolved, oxygen-evolving photosystem II preparation from spinach thylakoid membranes: EPR and electron-transport properties. FEBS Lett 134:231–234
Blankenship RE (2002) Molecular mechanisms of photosynthesis. Blackwell Science, Oxford
Boekema EJ, van Roon H, Calkoen F, Bassi R, Dekker JP (1999) Multiple types of association of photosystem II and its light-harvesting antenna in partially solubilized photosystem II membranes. Biochemistry 38:2233–2239
Bos I, Bland KM, Tian L, Croce R, Frankel LK, van Amerongen H, Bricker TM, Wientjes E (2017) Multiple LHCII antennae can transfer energy efficiently to a single photosystem I. Biochim Biophys Acta 1858:371–378
Bricker TM (1990) The structure and function of CPa-1 and CPa-2 in photosystem II. Photosynth Res 24:1–13
Bricker TM, Frankel LK (2002) The structure and function of CP47 and CP43 in photosystem II. Photosynth Res 72:131–146
Bricker TM, Mummadisetti MP, Frankel LK (2015) Recent advances in the use of mass spectrometry to examine structure/function relationships in photosystem II. J Photochem Photobiol B 152:227–246
Cao P, Su X, Pan X, Liu Z, Chang W, Li M (2018) Structure, assembly and energy transfer of plant photosystem II supercomplex. Biochim Biophys Acta 1859:633–644
Castresana J (2000) Selection of conserved blocks from multiple alignments for their use in phylogenetic analysis. J Mol Biol Evol 17:540–542
Chevenet F, Brun C, Bañuls A-L, Jacq B, Christen R (2006) TreeDyn: towards dynamic graphics and annotations for analyses of trees. BMC Bioinform 7:439
Choudhary FK, Rivero RM, Blumwald E, Mittler R (2017) Reactive oxygen species, abiotic stress and stress combination. Plant J 90:856–867
Chukhutsina VU, Liu X, Xu P, Croce R (2020) Light-harvesting complex II is an antenna of photosystem I in dark-adapted plants. Nat Plants 6:860–868
Crepin A, Caffarri S (2015) The specific localizations of phosphorylated Lhcb1 and Lhcb2 isoforms reveal the role of Lhcb2 in the formation of the PSI-LHCII supercomplex in Arabidopsis during state transitions. Biochim Biophys Acta 1847:1539–1548
Croce R (2020) Beyond ‘seeing is believing’: the antenna size of the photosystems in vivo. New Phytol 228:1214–1218
Croce R, van Amerongen H (2013) Light-harvesting in photosystem I. Photosyn Res 116:153–166
Danielsson R, Albertsson P-Å (2009) Fragmentation and separation analysis of the photosynthetic membrane from spinach. Biochim Biophys Acta 1787:25–36
Das K, Roychoudhury A (2014) Reactive oxygen species (ROS) and response of antioxidants as ROS-scavengers during environmental stress in plants. Front Environ Sci. https://doi.org/10.3389/fenvs.2014.00053
DeLano WL (2002) The PyMOL Molecular Graphics System, Version 1.2r3pre, Schrödinger, LLC.
Delepelaire P, Chua N-H (1979) Lithium dodecyl sulfate/polyacrylamide gel electrophoresis of thylakoid membranes at 4 C: characterizations of two additional chlorophyll a-protein complexes. Proc Natl Acad Sci U S A 76:111–115
Dereeper A, Guignon V, Blanc G, Audic S, Buffet S, Chevenet F, Dufayard JF, Guindon S, Lefort V, Lescot M, Claverie JM, Gascuel O (2008) Phylogeny.fr: robust phylogenetic analysis for the non-specialist. Nucleic Acid Res 36:W465–W469
Drop B, Webber-Birungi M, Yadav SK, Filipowicz-Szymanska A, Fusetti F, Boekema EJ, Croce R (2014) Light-harvesting complex II (LHCII) and its supramolecular organization in Chlamydomonas reinhardtii. Biochim Biophys Acta 1837:63–72
Duchene S, Siegenthaler PA (2000) Do glycerolipids display lateral heterogeneity in the thylakoid membrane? Lipids 35:739–744
Dunahay TG, Staehelin LA, Seibert M, Ogilvie PD, Berg SP (1984) Structural, biochemical and biophysical characterization of four oxygen-evolving Photosystem II preparations from spinach. Biochim Biophys Acta 764:179–193
Dupree EJ, Jayathirtha M, Yorkey H, Mihasan M, Petre BA, Darie CC (2020) A critical review of bottom-up proteomics: the good, the bad, and the future of this field. Proteomes. https://doi.org/10.3390/proteomes8030014
Foyer CH (2018) Reactive oxygen species, oxidative signaling and the regulation of photosynthesis. Environ Exp Bot 154:134–142
Frankel LK, Sallans L, Limbach PA, Bricker TM (2012) Identification of oxidized amino acid residues in the vicinity of the Mn(4)CaO(5) cluster of photosystem II: implications for the identification of oxygen channels within the photosystem. Biochemistry 51:6371–6377
Frankel LK, Sallans L, Bellamy H, Goettert JS, Limbach PA, Bricker TM (2013) Radiolytic mapping of solvent-contact surfaces in photosystem II of higher plants: experimental identification of putative water channels within the photosystem. J Biol Chem 288(32):23565–23572. https://doi.org/10.1074/jbc.M113.487033
Frankel LK, Sallans L, Limbach PA, Bricker TM (2013) Oxidized amino acid residues in the vicinity of Q(A) and Pheo(D1) of the photosystem II reaction center: putative generation sites of reducing-side reactive oxygen species. PLoS ONE 8(2):e58042. https://doi.org/10.1371/journal.pone.0058042
Galka P, Santabarbara S, Khuong TT, Degand H, Morsomme P, Jennings RC, Boekema EJ, Caffarri S (2012) Functional analyses of the plant photosystem I-light-harvesting complex II supercomplex reveal that light-harvesting complex II loosely bound to photosystem II is a very efficient antenna for photosystem I in state II. Plant Cell 24:2963–2978
Haferkamp S, Haase W, Pascal AA, van Amerongen H, Kirchhoff H (2010) Efficient light harvesting by photosystem II requires an optimized protein packing density in Grana thylakoids. J Biol Chem 285(22):17020–17028. https://doi.org/10.1074/jbc.M109.077750
Halliwell B (2006) Reactive species and antioxidants. Redox biology is a fundamental theme of aerobic life. Plant Physiol 141:312–322
Havaux M (2013) Carotenoid oxidation products as stress signals in plants. Plant J 79:597–606
Huang Z, Shen L, Wang W, Mao Z, Yi X, Kuang T, Shen J-R, Zhang X, Han G (2021) Structure of photosystem I-LHCI-LHCII from the green alga Chlamydomonas reinhardtii in state 2. Nature Commun 12:1100. https://doi.org/10.1038/s41467-021-21362-6
Hui Y, Jie W, Carpentier R (2000) Degradation of the photosystem I complex during photoinhibition. Photochem Photobiol 72:508–512
Jansson S (1994) The light-harvesting chlorophyll ab-binding proteins. Biochim Biophys Acta 1184:1–19
Johnson MP, Ruban AV (2009) Photoprotective energy dissipation in higher plants involves alteration of the excited state energy of the emitting chlorophyll(s) in the light harvesting antenna II (LHCII). J Biol Chem 284:23592–23601
Kale R, Hebert AE, Frankel LK, Sallans L, Bricker TM, Pospisil P (2017) Amino acid oxidation of the D1 and D2 proteins by oxygen radicals during photoinhibition of photosystem II. Proc Natl Acad Sci U S A 114:2988–2993
Kale R, Sallans L, Frankel LK, Bricker TM (2020) Natively oxidized amino acid residues in the spinach PS I-LHC I supercomplex. Photosynth Res 143:263–273
Kargul J, Turkina MV, Nield J, Benson S, Vener AV, Barber J (2005) Light-harvesting complex II protein CP29 binds to photosystem I of Chlamydomonas reinhardtii under state 2 conditions. FEBS J 272:4797–4806
Kumar A, Prasad A, Sedlářová M, Kale R, Frankel LK, Sallans L, Bricker TM, Pospíšil P (2021) Tocopherol controls D1 amino acid oxidation by oxygen radicals in photosystem II. Proc Natl Acad Sci 118(4):e2019246118. https://doi.org/10.1073/pnas.2019246118
Kyle DJ, Staehelin LA, Arntzen CJ (1983) Lateral mobility of the light-harvesting complex in chloroplast membranes controls excitation energy distribution in higher plants. Arch Biochem Biophys 222:527–541
Le Quiniou C, van Oort B, Drop B, van Stokkum HM, Croce R (2015) The high efficiency of Photosystem I in the green alga Chlamydomonas reinhardtii is maintained after the antenna size is substantially increased by the association of light-harvesting complexes II. J Biol Chem 290:30587–30595
Lindahl M, Yang D-H, Andersson B (1995) Regulatory proteolysis of the major light-harvesting chlorophyll a/ b protein of photosystem II by a light-induced membrane-associated enzymic system. Eur J Biochem 231:503–509
Lingvay M, Akhtar P, Sebők-Nagy K, Páli T, Lambrev PH (2020) Photobleaching of chlorophyll in light-harvesting complex II Increases in lipid environment. Front Plant Sci. https://doi.org/10.3389/fpls.2020.00849
Longoni P, Douchi D, Cariti F, Fucile G, Goldschmidt-Clermont M (2015) Phosphorylation of the light-harvesting complex II isoform Lhcb2 Is central to state transitions. Plant Physiol 169:2874–2883
Mazor Y, Borovikova A, Caspy I, Nelson N (2017) Structure of the plant photosystem I supercomplex at 2.6 angstrom resolution. Nat Plants 3:17014
Møller B, L., Høj PB, Henry LEA (1984) Electron mcroscopic characteristics of photosystem II preparations and their Iiactivation and reactivation with respect to oxygen evolution. In: Sybesma C (ed) Advances in photosynthesis research: proceedings of the VIth international congress on photosynthesis, Brussels, Belgium, August 1–6, 1983 Vol. 3. Springer Netherlands, Dordrecht, pp 219–222
Mozzo M, Dall’Osto L, Hienerwadel R, Bassi R, Croce R (2008) Photoprotection in the antenna complexes of photosystem II: role of individual xanthophylls in chlorophyll triplet quenching. J Biol Chem. https://doi.org/10.1074/jbc.M708961200
Munro D, Treberg JR (2017) A radical shift in perspective: mitochondria as regulators of reactive oxygen species. J Exp Biol 220:1170–1180
Natali A, Gruber JM, Dietzel L, Stuart MCA, van Grondelle R, Croce R (2016) Light-harvesting complexes (LHCs) cluster spontaneously in membrane environment leading to shortening of their excited state lifetimes. J Biol Chem 291:16730–16739
Nosek L, Semchonok D, Boekema EJ, Ilik P, Kouril R (2017) Structural variability of plant photosystem II megacomplexes in thylakoid membranes. Plant J 89:104–111
Pan X, Ma J, Su X, Cao P, Chang W, Liu Z, Zhang X, Li M (2018) Structure of the maize photosystem I supercomplex with light-harvesting complexes I and II. Science 360:1109–1113
Pietrzykowska M, Suorsa M, Semchonok DA, Tikkanen M, Boekema EJ, Aro EM, Jansson S (2014) The light-harvesting chlorophyll a/b binding proteins Lhcb1 and Lhcb2 play complementary roles during state transitions in Arabidopsis. Plant Cell 26(9):3646–3660. https://doi.org/10.1105/tpc.114.127373
Rabilloud T, Vincon M, Garin J (1995) Micropreparative one-and two-dimensional electrophoresis: improvement with new photopolymerization systems. Electrophoresis 16:1414–1422
Rinalducci S, Pedersen JZ, Zolla L (2004) Formation of radicals from singlet oxygen produced during photoinhibition of isolated light-harvesting proteins of photosystem II. Biochim Biophys Acta 1608:63–73
Schwarz EM, Tietz S, Froehlich JE (2018) Photosystem I-LHCII megacomplexes respond to high light and aging in plants. Photosynth Res 136:107–124
Sievers F, Higgins DG (2018) Clustal Omega for making accurate alignments of many protein sequences. Protein Sci 27:135–145
Slovin JP, Tobin EM (1982) Synthesis and turnover of the light-harvesting chlorophyll a/b-protein in Lemna gibba grown with intermittent red light: possible translational control. Planta 154:465–472
Staehelin LA, Paolillo DJ (2020) A brief history of how microscopic studies led to the elucidation of the 3D architecture and macromolecular organization of higher plant thylakoids. Photosynth Res 145:237–258
Standfuss J, Terwisscha van Scheltinga AC, Lamborghini M, Kuhlbrandt W (2005) Mechanisms of photoprotection and nonphotochemical quenching in pea light-harvesting complex at 2.5 A resolution. EMBO J 24:919–928
Su X, Ma J, Wei X, Cao P, Zhu D, Chang W, Liu Z, Zhang X, Li M (2017) Structure and assembly mechanism of plant C(2)S(2)M(2)-type PSII-LHCII supercomplex. Science 357:815–820
Takahashi H, Iwai M, Takahashi Y, Minagawa J (2006) Identification of the mobile light-harvesting complex II polypeptides for state transitions in Chlamydomonas reinhardtii. Proc Natl Acad Sci U S A 103:477–482
Taylor RM, Sallans L, Frankel LK, Bricker TM (2018) Natively oxidized amino acid residues in the spinach cytochrome b6f complex. Photosynth Res 137:141–151
Triantaphylidès C, Havaux M (2009) Singlet oxygen in plants: production, detoxification and signaling. Trends iPlant Sci 14:219–228
Tripathy BC, Oelmüller R (2012) Reactive oxygen species generation and signaling in plants. Plant Signal Behav 7:1621–1633
Urban A, Rogowski P, Wasilewska-Dębowska W, Romanowska E (2020) Effect of light on the rearrangements of PSI super-and megacomplexes in the non-appressed thylakoid domains of maize mesophyll chloroplasts. Plant Science 301:110655
van Bezouwen LS, Caffarri S, Kale RS, Kouril R, Thunnissen AWH, Oostergetel GT, Boekema EJ (2017) Subunit and chlorophyll organization of the plant photosystem II supercomplex. Nat Plants 3:17080
Veerman J, McCommell MD, Vasil’ev S, Mamedov F, Styring S, Bruce D (2007) Functional heterogeneity of photosystem II in domain specific regions of the thylakoid membrane of spinach (Spinacia oleracea L.). Biochemistry 46:3443–3453
Wei X, Su X, Cao P, Liu X, Chang W, Li M, Zhang X, Liu Z (2016) Structure of spinach photosystem II-LHCII supercomplex at 3.2 A resolution. Nature 534:69–74
Wientjes E, Drop B, Kouril R, Boekema EJ, Croce R (2013) During state 1 to state 2 transition in Arabidopsis thaliana, the photosystem II supercomplex gets phosphorylated but does not disassemble. J Biol Chem 288:32821–32826
Wientjes E, van Amerongen H, Croce R (2013) LHCII is an antenna of both photosystems after long-term acclimation. Biochim Biophys Acta 1827:420–426
Xu H, Freitas MA (2009) MassMatrix: a database search program for rapid characterization of proteins and peptides from tandem mass spectrometry data. Proteomics 9(6):1548–1555
Yadav KNS, Semchonok DA, Nosek L, Kouřil R, Fucile G, Boekema EJ, Eichacker LA (2017) Supercomplexes of plant photosystem I with cytochrome b6f, light-harvesting complex II and NDH. Biochim Biophys Acta 1858:12–20
Yang D-H, Webster J, Adam Z, Lindahl M, Andersson B (1998) Induction of acclimative proteolysis of the light-harvesting chlorophyll a/b protein of photosystem II in response to elevated light intensities. Plant Physiol 118:827–834
Yang D-H, Paulsen H, Andersson B (2000) The N- terminal domain of the light-harvesting chlorophyll a/b-binding protein complex (LHCII) is essential for its acclimative proteolysis. FEBS Lett 466:385–388
Zhang R, Sahu ID, Bali AP, Dabney-Smith C, Lorigan GA (2017) Characterization of the structure of lipodisq nanoparticles in the presence of KCNE1 by dynamic light scattering and transmission electron microscopy. Chem Phys Lipids 203:19–23
Zolla L, Rinalducci S (2002) Involvement of active oxygen species in degradation of light-harvesting proteins under light stresses. Biochemistry 41:14391–14402
Acknowledgements
This work was solely supported by the United States Department of Energy, Office of Basic Energy Sciences grant DE-FG02-09ER20310 to TMB and LKF.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kale, R.S., Seep, J.L., Sallans, L. et al. Oxidative modification of LHC II associated with photosystem II and PS I-LHC I-LHC II membranes. Photosynth Res 152, 261–274 (2022). https://doi.org/10.1007/s11120-022-00902-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-022-00902-1