Abstract
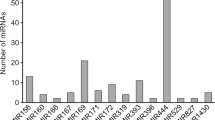
Banana has a high leaf area index and shallow root system, hence making it highly susceptible to water stress. One of the adaptive mechanisms is the deposition of cuticular wax on a leaf. Our previous analysis revealed the role of total cuticular wax, carbon chain length of the cuticular wax component, and the role of ester and alcohol wax compounds in maintaining the hydration of banana leaves which was further confirmed by gene expression analysis. To understand the regulatory mechanism of these wax genes in the present study, we have computationally mined and filtered miRNAs whose targets are involved in the wax biosynthetic process using M.balbisiana and M.acuminata transcriptome. Here, we predicted 96 and 62 conserved miRNA families and its targets in M.balbisiana and M.acuminata, respectively. Based on biochemical pathway, we filtered miRNAs and its targets which were involved in the wax biosynthetic process .Here, we report two miRNAs that are MbmiR531 whose target is KCS11 and MbmiR529 whose target is KCS10/FDH. The in silico characterization of MbmiR531 and MbmiR529 revealed that it had a strong, stable secondary structure with MFE of −23.4 and −26.5 kcal/mol, respectively, and MFEI value of −0.70 and −0.66 kcal/mol, respectively. Validation of the computational prediction was carried out on four high wax musa genotypes (990.6–1842.6 μg/dm2) and four low wax musa genotypes (424.1–780.2 μg/dm2) by qRT PCR which further revealed a negative relationship between the target gene and its miRNA thus indicating their role in wax biosynthesis regulation.






Similar content being viewed by others
References
Balcells I, Cirera S, Busk PK (2011) Specific and sensitive quantitative RT-PCR of miRNAs with DNA primers. BMC Biotechnol 11:70
Baldrich P, Campo S, Wu M-T, Liu T-T, Hsing Y-IC, Segundo BS (2015) MicroRNA-mediated regulation of gene expression in the response of rice plants to fungal elicitors. RNA Biol 12:847–863
Barakat A, Wall K, Leebens-Mack J, Wang YJ, Carlson JE, Depamphilis CW (2007) Large-scale identification of microRNAs from a basal eudicot (Eschscholzia californica) and conservation in flowering plants. Plant J 51:991–1003, PMID: 17635767
Barciszewska-Pacak M, Milanowska K, Knop K, Bielewicz D, Nuc P, Plewka P et al (2015) Arabidopsis microRNA expression regulation in a wide range of abiotic stress responses. Front Plant Sci 6:410
Bernard A, Joubes JE (2013) Arabidopsis cuticular waxes: advances in synthesis, export and regulation. Prog Lipid Res 52:110–129. doi:10.1016/j.plipres.2012.10.002
Bi F, Meng X, Ma C, Yi G (2015) Identification of miRNAs involved in fruit ripening in Cavendish bananas by deep sequencing. BMC Genomics 16:1
Busk PK (2014) A tool for design of primers for microRNA-specific quantitative RT-qPCR. BMC Bioinf 28(15):29. doi:10.1186/1471-2105-15-29
Bustin SA, Nolan T (2004) Data analysis and interpretation. In: Bustin SA (ed) A-Z of quantitative PCR. International University line, CA, USA, pp 439–481
Chai J, Feng R, Shi H, Ren M, Zhang Y, Wang J (2015) Bioinformatic identification and expression analysis of banana microRNAs and their targets. Plos One 10(4):e0123083
Chang S, Puryear J, Cairney J (1993) A simple and efficient method for isolating RNA from pine trees. Plant Mol Biol Report 11(2):113–116
Chen L, Zhong H et al (2011) Validation of reference genes for RT-qPCR studies of gene expression in banana fruit under different experimental conditions. Planta 234:377–390
D’Hont A, Denoeud F et al (2012) The banana (Musa acuminata) genome and the evolution of monocotyledonous plants. Nature 488(7410):213–217
Davey MW, Gudimella R et al (2013) Draft Musa balbisiana genome sequence for molecular genetics in polyploid, inter-and intra-specific Musa hybrids. BMC Genomics 14:683
Dehury B, Panda D, Sahu J, Sahu M, Sarma K, Barooah M et al (2013) In silico identification and characterization of conserved miRNAs and their target genes in sweet potato (Ipomoea batatas L.) Expressed Sequence Tags (ESTs). Plant Signal Behav 8:e26543
Ebercon A, Blum A, Jordan WR (1977) Rapid colorimetric method for epicuticular wax contest of sorghum leaves. Crop Sci 17:179–180. doi:10.2135/cropsci1977.0011183X001700010047x
Food and Agriculture Organization of the United Nations (FAO 2012) [http://faostat.fao.org]
Gupta H, Tiwari T, Patel M, Mehta A, Ghosh A (2015) An approach to identify the novel miRNA encoded from H.Annuus EST sequences. Genom Data 6:139–144
Jones-Rhoades MW, Bartel DP, Bartel B (2006) MicroRNAs and their regulatory roles in plants. Annu Rev Plant Biol 57:19–53
Joubes J, Raffaele S, Bourdenx B, Garcia C, Laroche-Traineau J, Moreau P et al (2008) The VLCFA elongase gene family 30 in Arabidopsis thaliana: phylogenetic analysis, 3D modelling and expression profiling. Plant Mol Biol 67:547–566. doi:10.1007/s11103-008-9339-z
Khraiwesh B, Zhu J-K, Zhu J (2012) Role of miRNAs and siRNAs in biotic and abiotic stress responses of plants. Biochim Biophys Acta 1819:137–148
Kim J, Jung JH, Lee SB, Go YS, Kim HJ, Cahoon R et al (2013) Arabidopsis 3-ketoacyl-coenzyme A synthase9 is involved in the synthesis of tetracosanoic acids as precursors of cuticular waxes, suberins, sphingolipids, and phospholipids. Plant Physiol 162:567–580
Kozomara A, Griffiths-Jones S (2014) miRBase: annotating high confidence microRNAs using deep sequencing data. Nucleic Acids Res 42:68–73. doi:10.1093/nar/gkt1181, PMID: 24275495
Kruszka K, Pacak A, Swida-Barteczka A, Nuc P, Alaba S, Wroblewska Z et al (2014) Transcriptionally and post-transcriptionally regulated microRNAs in heat stress response in barley. J Exp Bot 65:6123, eru353
Kunst L, Samuels L (2009) Plant cuticles shine: advances in wax biosynthesis and export. Curr Opin Plant Biol 12(6):721–727
Lee SB, Kim H, Kim RJ, Suh MC (2014) Overexpression of Arabidopsis MYB96 confers drought resistance in Camelina sativa via cuticular wax accumulation. Plant Cell Rep 33:1535–1546
Levitt J (1972) Responses of plants to environmental stresses. Academic Press, New York
Lokesh U, Kiranmai K, Pandurangaiah M, Sudhakarbabu O, Nareshkumar A, Sudhakar C (2013) Role of plant fatty acid elongase (3 keto acyl-CoA synthase) gene in cuticular wax biosynthesis. J Agric Allied Sci 2:35–42
Merchuk L, Saranga Y (2013) Breeding approaches to increasing water-use efficiency. In: Rengel Z (ed) Improving water and nutrient-use efficiency in food production systems. John Wiley & Sons Inc, Hoboken, pp 145–160
Nobusawa T, Okushima Y, Nagata N, Kojima M, Sakakibara H, Umeda M (2013) Synthesis of very-long-chain fatty acids in the epidermis controls plant organ growth by restricting cell proliferation. Plos Biol 11:e1001531
Numnark S, Mhuantong W, Ingsriswang S, Wichadakul D (2012) C-mii: a tool for plant miRNA and target identification. BMC Genomics 13:S16. doi:10.1186/1471-2164-13-S7-S16
Prakash P, Rajakani R, Gupta V (2015) Transcriptome-wide identification of Rauvolfia serpentina microRNAs and prediction of their potential targets. Comput Biol Chem 61:62–74
Pruitt RE, Vielle-Calzada J-P, Ploense SE, Grossniklaus U et al (2000) FIDDLEHEAD, a gene required to suppress epidermal cell interactions in Arabidopsis, encodes a putative lipid biosynthetic enzyme. Proc Natl Acad Sci U S A 97:1311–1316
Ravishankar KV, Megha HS, Rekha A, Ganesh NK, Veerraju C (2015) Insights into Musa balbisiana and Musa acuminata species divergence and development of genic microsatellites by transcriptomics approach. Plant Genet 4:78–82
Riederer M, Schreiber L (2001) Protecting against water loss: analysis of the barrier properties of plant cuticles. J Exp Bot 52:2023–2032. doi:10.1093/jexbot/52.363.2023
Robinson J.C (1996) Banana and Plantains. Wallingford, UK
Sampangi-Ramaiah MH, Ravishankar KV (2016) Current status of banana genome in the age of next generation sequencing. In: Mohandas S, Ravishankar KV (eds) Banana: genomics and transgenic approaches for genetic improvement. Springer, Singapore, pp 51–59
Sampangi-Ramaiah MH, Ravishankar KV, Seetharamaiah SK, Roy TK, Hunashikatti L, Rekha A, Shilpa P (2016) Barrier against water loss: relationship between epicuticular wax composition, gene expression and leaf water retention capacity in banana. Funct Plant Biol 43:492–501
Samuels L, Kunst L, Jetter R (2008) Sealing plant surfaces: cuticular wax formation by epidermal cells. Plant Biol 59:683
Seo PJ, Park C-M (2011) Cuticular wax biosynthesis as a way of inducing drought resistance. Plant Signal Behav 6:1043–1045
Seo PJ, Lee SB, Suh MC, Park M-J, Go YS, Park C-M (2011) The MYB96 transcription factor regulates cuticular wax biosynthesis under drought conditions in Arabidopsis. Plant Cell 23:1138–1152
Shepherd T, Wynne Griffiths D (2006) The effects of stress on plant cuticular waxes. New Phytol 171:469–499
Wang M, Wang Q, Wang B (2012) Identification and characterization of microRNAs in Asiatic cotton (Gossypium arboreum L.). Plos One 7:e33696
Wen J-Z, Liao J-Y, Zheng L-L, Xu H, Yang J-H, Guan D-G et al (2014) A contig-based strategy for the genome-wide discovery of MicroRNAs without complete genome resources. Plos One 9(2):e88179
Wienken CJ, Baaske P, Duhr S, Braun D (2011) Thermophoretic melting curves quantifies the conformation and stability of RNA and DNA. Nucleic Acids Res 39(8):e52–e52. doi:10.1093/nar/gkr035, PMC 3082908, PMID 21297115
Xia K, Ou X, Gao C, Tang H, Jia Y, Deng R, Xu X et al (2015) OsWS1 involved in cuticular wax biosynthesis is regulated by osa-miR1848. Plant Cell Environ 38:2662–2673
Yephremov A, Wisman E, Huijser P, Huijser C, Wellesen K, Saedler H (1999) Characterization of the FIDDLEHEAD gene of Arabidopsis reveals a link between adhesion response and cell differentiation in the epidermis. Plant Cell 11:2187–2201
Yuan S, Li Z, Li D, Yuan N, Hu Q, Luo H (2015) Constitutive expression of rice MicroRNA528 alters plant development and enhances tolerance to salinity stress and nitrogen starvation in creeping bentgrass. Plant Physiol 169(1):576–593
Zhang B, Pan X, Cox S, Cobb G, Anderson T (2006) Evidence that miRNAs are different from other RNAs. Cell Mol Life Sci 63:246–254
Zhang S-D, Ling L-Z, Zhang Q-F, Xu J-D, Cheng L (2015) Evolutionary Comparison of Two Combinatorial Regulators of SBP-Box Genes, MiR156 and MiR529, in Plants. Plos One 10(4):e0124621. doi:10.1371/journal.pone.0124621
Zhao B, Liang R, Ge L, Li W, Xiao H, Lin H et al (2007) Identification of drought-induced microRNAs in rice. Biochem Biophys Res Commun 354:585–590
Acknowledgements
We thank the Indian Council of Agricultural Research, New Delhi for financial assistance through the ICAR Network Project on Transgenics in Crops: Functional Genomics-Fusarium wilt and drought tolerance in Banana. M/s Genotypic Pvt. Ltd. Bangalore for sequencing and bioinformatic assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sampangi-Ramaiah, M.H., Ravishankar, K.V., Rekha, A. et al. A Contig-Based Computational Prediction of Conserved miRNAs and Their Probable Role in Regulation of Cuticular Wax Biosynthesis in Banana. Plant Mol Biol Rep 35, 203–214 (2017). https://doi.org/10.1007/s11105-016-1016-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11105-016-1016-9