Abstract
Aims
The accumulation of organic acids in soil can trigger autotoxicity, resulting in the continuous cropping obstacles (CCOs) for Panax notoginseng under conventional management systems. However, limited information is available about the threshold value of organic acids representing the CCOs in the soil of P. notoginseng from the forest understorey.
Methods
In this study, mixed organic acids at different concentrations were exogenously applied to simulate the P. notoginseng CCO. The plant growth, edaphic factors, soil extracellular enzyme activity, and bacterial/fungal abundances and diversity in the rhizospheric soil were assessed in the P. notoginseng from the forest understorey.
Results
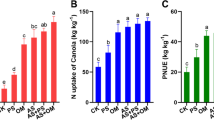
High concentrations (>150 mg/kg) rather than low concentrations (<150 mg/kg) of organic acid decreased the dry and fresh weight of plants (1.99%-1.98%), edaphic properties (2.75%-1.03%) and soil extracellular enzyme activities (3.38%-1.05%). Under high organic acid concentrations, the fungal abundance increased by 2.37%, whereas the bacterial abundance decreased by 4.56%. Moreover, the abundance of Pseudomonadales, Xanthomonadales, and Ilyonectria as pathogenic bacteria tended to increase. Compared to bacteria, the fungal community structure varied significantly, and the fungal core flora was more abundant. The SEM results showed that the direct and indirect factors affecting the α-diversity in the rhizosphere of P. notoginseng were increased under the high organic acid concentrations.
Conclusion
Organic acid concentrations greater than 150 mg/kg in P. notoginseng soils are detrimental to the relationships among plant growth, soil environment, and microbiomes, which implies the occurrence of the CCOs in P. notoginseng; and therefore, we recommend that P. notoginseng should not be planted again.








Similar content being viewed by others
Data availability
The datasets analysed during the current study are available from the corresponding author on reasonable request.
References
Bai YX, Wang G, Cheng YD, Shi PY, Yang CC, Yang HW, Xu ZL (2019) Soil acidification in continuously cropped tobacco alters bacterial community structure and diversity via the accumulation of phenolic acids. Sci Rep 9:12499. https://doi.org/10.1038/s41598-019-48611-5
Balke NE (1985) Effects of allelochemicals on mineral uptake and associated physiological process. ACS Symp Series 268:161–178. https://doi.org/10.1021/bk-1985-0268.ch011
Bao LM, Ding YF, Wei YL (2021) Analysis on the composition and diversity of fungi community in the continuous cropping and fallow soil of Panax notoginseng. J Chin Med Mater 44(01):7–12. https://doi.org/10.13863/j.issn1001-4454.2021.01.002
Bao LM, Liu YY, Ding YF, Shang JJ, Wei YL, Tan Y, Zi FT (2022) Interactions between phenolic acids and microorganisms in rhizospheric soil from continuous cropping of Panax notoginseng. Front Microbiol 13:791603–791603. https://doi.org/10.3389/fmicb.2022.791603
Bardhan S, Jose S, Jenkins MA, Webster CR, Udawatta RP, Stehn SE (2012) Microbial community diversity and composition across a gradient of soil acidity in spruce-fir forests of the southern Appalachian Mountains. Appl Soil Ecol. https://doi.org/10.1016/j.apsoil.2012.04.010
Chaudhary P, Chaudhary A, Parveen H, Rani A, Kumar G, Kumar R, Sharma A (2021) Impact of nanophos in agriculture to improve functional bacterial community and crop productivity. BMC Plant Biol 21:1–12. https://doi.org/10.1186/s12870-021-03298-7
Chaudhary P, Chaudhary A, Bhatt P, Kumar G, Khatoon H, Rani A, Kumar S, Sharma A (2022) Assessment of soil health indicators under the influence of nanocompounds and bacillus spp. in field condition. Front Environ Sci. https://doi.org/10.3389/fenvs.2021.769871
Chaudhry Q, Blom-Zandstra M, Gupta SK, Joner E (2004) Utilising the synergy between plants and rhizosphere microorganisms to enhance breakdown of organic pollutants in the environment. Environ Sci Pollut Res Int 12:34–48. https://doi.org/10.1065/espr2004.08.213
Chen SL, Zhou BL, Lin SS, Li X, Xi HJ, Yin YL, Ye XL (2011) Allelopathic effects of cinnamic acid and vanillin on soil microbes, soil enzymes activities and growth of grafted eggplants. Allelopathy J 28(01):29–40
Chen L, Zhang ML, Xin MY, Li JD (2015) Effects of exogenous phenolic acids on allelopathy of potted soybean seedlings. Agric Sci Tech 16:1151. https://doi.org/10.16175/j.cnki.1009-4229.2015.06.014
Chen LP, Dong KP, Yang ZX, Dong Y, Tang L, Zheng Y (2017) Allelopathy autotoxicity effect of successive cropping obstacle and its alleviate mechanism by intercropping. Chin Agric Sci Bull 33(08):91–98
Chen LC, Liao LP, Wang SL (2003) Effect of vanillin on nutrient absorbency in Chinese fir seedlings. Acta Phytoecological Sinica. 27(1):41–46. https://doi.org/10.17521/cjpe.2003.0006
Chen SF, Zhou YQ, Chen YR, Gu J (2018) Fastp: an ultra-fast all-in-one FASTQ preprocessor. Bioinformatics 34(17):i884–i890. https://doi.org/10.1093/bioinformatics/bty560
Chen P, Wang YZ, Liu QZ, Zhang YT, Li XY, Li HQ, Li WH (2020) Phase changes of continuous cropping obstacles in strawberry (Fragaria×ananassa Duch.) production. Appl Soil Ecol 155:103626. https://doi.org/10.1016/j.apsoil.2020.103626
Chen SY, Rui R, Wang S, He XH (2022) Comparative analysis of the floral fragrance compounds of Panax notoginseng flowers under the Panax notoginseng-pinus agroforestry system using SPME-GC-MS. Molecules 27:3565. https://doi.org/10.3390/molecules27113565
Cutino-Jimenez AM, Martins-Pinheiro M, Lima WC, Martín-Tornet A, Morales OG, Menck CFM (2010) Evolutionary placement of Xanthomonadales based on conserved protein signature sequences. Mol Phylogenet Evol 54(2):524–534. https://doi.org/10.1016/j.ympev.2009.09.026
de Menezes AB, Richardson AE, Thrall PH (2017) Linking fungal-bacterial co-occurrences to soil ecosystem function. Curr Opin Microbiol 37:135–141. https://doi.org/10.1016/j.mib.2017.06.006
Drenovsky RE, Vo D, Graham KJ, Scow KM (2004) Soil water content and organic carbon availability are major determinants of soil microbial community composition. Microb Ecol 48(3):424–430. https://doi.org/10.1007/s00248-003-1063-2
Einhellig FA (1996) Interactions involving allelopathy in cropping systems. Agron J 88:886–893. https://doi.org/10.2134/agronj1996.00021962003600060007x
German DP, Weintraub MN, Grandy AS, Lauber CL, Rinkes ZL, Allison SD (2011) Optimization of hydrolytic and oxidative enzyme methods for ecosystem studies. Soil Biol Biochem 43:1387–1397. https://doi.org/10.1016/j.soilbio.2011.03.017
Guan HL, Yang JZ, Chen YJ, Cui XM, Wang Y, Zhang YF (2010) Change of rhizospheric microbe colony in cultivated soil and its correlation to root rot disease in Panax notoginseng. Soils 42(03):378–384. https://doi.org/10.13758/j.cnki.tr.2010.03.019
Guo HB, Cui XM, An N, Cai GP (2010) Sanchi ginseng (Panax notoginseng (Burkill) F. H. Chen) in China: distribution, cultivation and variations. Genet Resour Crop Evol 57:453–460. https://doi.org/10.1007/s10722-010-9531-2
Jia WJ, Wang S, He XH, Zhao XY (2022) Different factors drive the assembly of pine and Panax notoginseng-associated microbiomes in Panax notoginseng-pine agroforestry systems. Front Microbiol 13:1018989. https://doi.org/10.3389/fmicb.2022.1018989
Jian ZY, Wang WQ, You PJ (2009) Analysis of element contents in soil for continuous cropping Panax notoginseng. Mod. Chin Med 11:10–11. https://doi.org/10.13313/j.issn.1673-4890.2009.04.002
Jiang N, Qin LY, Ye YF (2011) Research advances in diseases of Panax notoginseng. J South Agric 42(09):1070–1074
Knief C, Delmotte N, Vorholt JA (2011) Bacterial adaptation to life in association with plants-a proteomic perspective from culture to in situ conditions. Proteomics 11(15):3086–3105. https://doi.org/10.1002/pmic.201000818
Lehman ME, Blum U, Gerig TM (1994) Simultaneous effects of ferulic and p-coumaric acids on cucumber leaf expansion in split-root experiments. J Chem Ecol 20:1773–1782. https://doi.org/10.1007/BF02059898
Li PD, Wang XX, Li YL, Wang HW, Liang FY, Dai CC (2010) The contents of phenolic acids in continuous cropping peanut and their allelopathy. Acta Ecol Sin 30(8):2128–2134
Li HQ, Zhang LL, Jiang XW, Liu QZ (2015) Allelopathic effects of phenolic acids on the growth and physiological characteristics of strawberry plants. Allelopathy J 35(1):61–75. https://www.researchgate.net/publication/279326538_Allelopathic_effects_of_phenolic_acids_on_the_growth_and_physiological_characteristics_of_strawberry_plants
Li R, Shen ZZ, Sun L, Zhang RF, Fu L, Deng XH, Shen QR (2016) Novel soil fumigation method for suppressing cucumber fusarium wilt disease associated with soil microflora alterations. Appl Soil Ecol 101:28–33. https://doi.org/10.1016/j.apsoil.2016.01.004
Li ZB, Fu JF, Zhou RJ, Wang D (2018) Effects of phenolic acids from ginseng rhizosphere on soil fungi structure, richness and diversity in consecutive monoculturing of ginseng. Saudi J Biol Sci 25(8):1788–1794. https://doi.org/10.1016/j.sjbs.2018.07.007
Li HC, Zuo YM, Yang SB, Yang TM, Li JC, Yang WZ, Zhang JY (2020) Ecological effect of root exudates of Panax notoginseng on continuous cropping obstacles and its alleviating methods. J Agric Sci Technol 22(08):159–167. https://doi.org/10.13304/j.nykjdb.2019.0594
Liu JJ, Yu ZH, Yao Q, Hu XY, Zhang W, Mi G, Chen XL, Wang GH (2017) Distinct soil bacterial communities in response to the cropping system in a Mollisol of Northeast China. Appl Soil Ecol 119:407–416. https://doi.org/10.1016/j.apsoil.2017.07.013
Liu YY, Bao LM, Wei YL, Zi FT, Tan Y (2022) The progress of interactions between continuous Panax notoginseng rhizosphere soil auto toxic substances and microorganism. J Chin Med Mate 45(01):242–247. https://doi.org/10.13863/j.issn1001-4454.2022.01.043
Magoč T, Salzberg SL (2011) FLASH: fast length adjustment of short reads to improve genome assemblies. Bioinformatics. https://doi.org/10.1093/bioinformatics/btr507
Mitter EK, de Freitas JR, Germida JJ (2017) Bacterial root microbiome of plants growing in oil sands reclamation covers. Front Microbiol 8:849. https://doi.org/10.3389/fmicb.2017.00849
Saiya-Cork KR, Sinsabaugh RL, Zak DR (2002) The effects of long-term nitrogen deposition on extracellular enzyme activity in an Acer saccharum forest soil. Soil Boil Biochem 34(9):1309–1315. https://doi.org/10.1016/S0038-0717(02)00074-3
Schloss PD, Westcott SL, Ryabin T, Hall JR, Hartmann M, Hollister EB, Lesniewski RA, Oakley BB, Parks DH, Robinson CJ, Sahl JW, Stres B, Thallinger GG, Van Horn DJ, Weber CF (2009) Introducing mothur: open–source, platform-independent, community-supported software for describing and comparing microbial communities. Appl Environ Microbiol 75(23):7537–7541. https://doi.org/10.1128/AEM.01541-09
Segata N, Izard J, Waldron L, Gevers D, Miropolsky L, Garrett WS, Huttenhower C (2011) Metagenomic biomarker discovery and explanation. Genome Biol 12:1–18. https://doi.org/10.1186/gb-2011-12-6-r60
Segura A, Ramos JL (2013) Plant-bacteria interactions in the removal of pollutants. Curr Opin Biotechnol 24(3):467–473. https://doi.org/10.1016/j.copbio.2012.09.011
Shao WZ, Zhou XG, Wen YG, Wang L, Zhu HG, Chen QH, Zhang YN, You YM (2022) Effects of mixing Castanopsis Hystrix and Eucalyptus on soil hydrolytic enzyme activities and ecoenzymatic stoichiometry. Guihaia 42(04):543–555. https://doi.org/10.11931/guihaia.gxzw202012030
Shi R, Wang S, Xiong BJ, Gu HY, Wang HL, Ji C, Jia WJ, Abraham RH, Zhen WJ, Jiftah BA, He XH (2022) Application of bioorganic fertilizer on Panax notoginseng improves plant growth by altering the rhizosphere microbiome structure and metabolism. Microorganisms 10(2):275. https://doi.org/10.3390/microorganisms10020275
Stanek M, Zubek S, Stefanowicz AM (2021) Differences in phenolics produced by invasive Quercus rubra and native plant communities induced changes in soil microbial properties and enzymatic activity. Forest Ecol Manag 482:118901. https://doi.org/10.1016/j.foreco.2020.118901
Sun XY, Long GQ, Zhang GH, Meng ZG, Chen ZJ, Yang SC, Chen JW (2015) Properties of soil physical-chemistry and activities of soil enzymes in context of continuous cropping obstacles for Panax notoginseng. Ecol Environ Sci 24(3):409–417
Tan Y, Cui YS, Li HY, Kuang AX, Li XR, Wei YL, Ji XL (2017) Rhizospheric soil and root endogenous fungal diversity and composition in response to continuous Panax notoginseng cropping practices. Microbiol Res 194:10–19. https://doi.org/10.1016/j.micres.2016.09.009
Tu YT, Huang JC, Peng ZP, Wu XN, Liao WJ (2021) Effect of biochar on tomato growth and soil microecology under phenolic acid stress. Guangdong Agric Sci 48(01):94–103
Wang HJ, Wang KY, Li SL, Song X, Xu WN, Guan HL (2019) Effects of different soil factors on the survival rate of Panax notoginseng in continuous cropping land. Lishizhen Med Mate Med Res 30(12):2982–2984. https://doi.org/10.3969/j.issn.1008-0805
Wang P, Yang L, Sun J, Yang Y, Qu Y, Wang C, Wang C, Liu D, Huang L, Cui X, Liu Y (2022) Structure and function of rhizosphere soil and root endophytic microbial communities associated with root rot of Panax notoginseng. Front Plant Sci 12:3222. https://doi.org/10.3389/fpls.2021.752683
Wu HM, Wu LK, Zhu Q, Wang JY, Qin XJ, Xu JH, Kong LF, Chen J, Lin S, Umar Khan M, Amjad H, Lin WX (2017) The role of organic acids on microbial deterioration in the Radix pseudostellariae rhizosphere under continuous monoculture regimes. Sci Rep 7:3497. https://doi.org/10.1038/s41598-017-03793-8
Xiang YP, Xiao F (2022) Development suggestions of under-forest traditional Chinese herbal medicine industry in Yunnan province. For Invent Plan 47(02):166–171. https://doi.org/10.3969/j.issn.1671-3168
Xun LL, Zhao HG, Liang ZS, Wei MT, Liu FH, Han RL (2013) Study of microecology in root rot and healthy Panax notoginseng soil. Acta Agric Boreali-Occidentalis Sinica 22(11):146–151
Yang JZ, Guan HL, Liu DH, Su YQ, Wei ML, Wang Y (2016) Study on mechanism of sanqi (Panax notoginseng F.H. Chen) replant failure and its alleviation technology. North Hortic 14:160–163
Yang M, Chuan YC, Guo CW, Liao JJ, Xu YG, Mei XY, Liu YX, Huang HC, He XH, Zhu SS (2018) Panax notoginseng root cell death caused by the autotoxic ginsenoside Rg1 is due to over-accumulation of ROS, as revealed by transcriptomic and cellular approaches. Front Plant Sci 9:264. https://doi.org/10.3389/fpls.2018.00264
Ye C, Liu YX, Zhang JX, Li TY, Zhang YJ, Guo CW, Yang M, He XH, Zhu YY, Huang HC, Zhu SS (2021) α-Terpineol fumigation alleviates negative plant-soil feedbacks of Panax notoginseng via suppressing Ascomycota and enriching antagonistic bacteria. Phytopathol Res 3(1):1–17. https://doi.org/10.1186/s42483-021-00090-1
Yele DJ, Ralph J (2008) Evidence for cleavage of lignin by a brown rot basidiomycete. Environ Microbiol 10(7):1844–1849. https://doi.org/10.1111/j.1462-2920.2008.01605.x
Yin S, Wang CK, Jin Y, Zhou ZH (2019) Changes in soil-microbe-exoenzyme C:N:P stoichiometry along an altitudinal gradient in Mt. Datudingzi, Northeast China. Chin J Plant Ecol 43(11):999–1009. https://doi.org/10.17521/cjpe.2019.0141
Yu P, He X, Baer M, Beirinckx S, Tian T, Moya YA, Hochholdinger F (2021) Plant flavones enrich rhizosphere Oxalobacteraceae to improve maize performance under nitrogen deprivation. Nat plants 7(4):481–499. https://doi.org/10.1038/s41477-021-00897-y
Yuan N, Zheng RH, Gu XP, Wu XL, Yue JJ (2010) Allelopathic effect of phenolic acids on Phyllostachys edulis seedlings. J Nanjing Forestry Univ 34(1):29–33. https://doi.org/10.3969/j.jssn.1000-2006.2010.01.007
Zhang ZY, Lin WX (2009) Continuous cropping obstacle and allelopathic autotoxicity of medicinal plants. Chin J Ecol Agric 17(1):189–196. https://doi.org/10.3724/SP.J.1011.2009.00189
Zhang Y, Zheng Y, Xia P, Xun L, Liang Z (2019) Impact of continuous Panax notoginseng plantation on soil microbial and biochemical properties. Sci Rep 9(1):1–8. https://doi.org/10.1038/s41598-019-49625-9
Zhang YJ, Li TY, Ye C, Lu RC, Liu YX, Huang HC, He XH, Yang M, Zhu SS (2021) Leaching alleviates phenol-mediated root rot in Panax notoginseng by modifying the soil microbiota. Plant Soil 468(1):491–507. https://doi.org/10.1007/s11104-021-05136-z
Zheng YB, Li M (2018) Autotoxicity of phenolic acids in root exudates of Andrographis paniculata (Burm. F.) nees. Allelopathy J 45(2):153–162. https://doi.org/10.26651/allelo.j/2018-45-2-1183
Zhou G, Gong FW, Peng J, Lou XQ (2016) Panax notoginseng F.H. Chen: cultivation techniques under different forests. J Agric For 6:41–44. https://doi.org/10.16175/j.cnki.1009-4229.2016.12.033
Acknowledgments
We are very grateful for the improvements of editor and reviewers about the manuscript, and the Yunnan Ten Thousand People Plan Youth Top Talent Project (YNWR-QNBJ-2019-028).
Funding
This work was supported by Major Science and Technology Project of Yunnan Province (202102AE090042, 2019ZG0901, 2021Y250), China Agriculture Research System (CARS-21) and Kunming Science and Technology Bureau (2021JH002).
Author information
Authors and Affiliations
Contributions
JH, SW, and XH designed the experiments and analysed the data. JH and SW draw the figures and wrote down the manuscript. SW and XH revised and edited the manuscript. All authors contributed to the article and approved the submitted version.
Corresponding authors
Ethics declarations
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Responsible Editor: Eric Blanchart.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 2969 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hei, J., Wang, S. & He, X. Effects of exogenous organic acids on the growth, edaphic factors, soil extracellular enzymes, and microbiomes predict continuous cropping obstacles of Panax notoginseng from the forest understorey. Plant Soil (2023). https://doi.org/10.1007/s11104-023-06044-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11104-023-06044-0