Abstract
Purpose
The developmental status of the male and female spike determines grain number (GN) per ear and yield. A better understanding of the effect of N on male and female spike differentiation and GN formation could help to gain insight into the mechanism of N regulation on yield.
Methods
A field experiment was conducted with four nitrogen rates: 0 (N0), 129 (N1), 185 (N2), and 300 kg N ha−1 (N3) with a single summer maize hybrid to determine tassel and ear development and the resulting GN.
Results
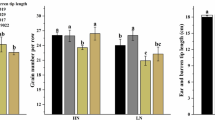
The expression of key proteins and genes related to fatty acid metabolism of N0 were down-regulated and the tapetum was abnormally degraded. This inhibited the synthesis of long-chain fatty acids (LCFAs) in the tassel and ear. Both pollen and silk development at the mononucleate stage were abnormal and presented as misshapen pollen, uneven silk tube, reduced silk, and poor pollination. Finally, anthesis-silking interval (ASI) was increased and the length of male and female spikes, fertilized florets, floret fertility percentage, and grain set rate were reduced in N0 treatment resulting in reduced GN. N application promoted fatty acid metabolism but N application greater than 185 kg N ha−1 did not increase LCFA of either male or female spike traits.
Conclusions
A nitrogen rate of 185 kg N ha−1 was sufficient to promote lipid metabolism, and increase the content of LCFAs. This resulted in normal pollen and silk development, shortened ASI and led to an increase in the number of fertile florets, fertilization percentage, and GN.





Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Baud S, Guyon V, Kronenberger J, Wuilléme S, Miquel M, Caboche M, Lepiniec L, Rochat C (2003) Multifunctional acetyl-CoA carboxylase 1 is essential for very long chain fatty acid elongation and embryo development in Arabidopsis. Plant J 33:75–86. https://doi.org/10.1046/j.1365-313X.2003.016010.x
Blackmore S (2007) Pollen and spores microscopic keys to understanding the earth’s biodiversity. Plant Syst Evol 263:3–12. https://doi.org/10.1007/s00606-006-0464-3
Bundy LG, Andraski TW, Ruark MD, Peterson AE (2011) Longterm continuous maize and nitrogen fertilizer effects on productivity and soil properties. Agron J 103:1346–1351. https://doi.org/10.2134/agronj2011.0094
Cai Q, Yuan Z, Chen MJ, Yin CS, Luo ZJ, Zhao XX, Liang WQ, Hu JP, Zhang DB (2014) Jasmonic acid regulates spikelet development in rice. Nat Commun 5:1–13. https://doi.org/10.1038/ncomms4476
Chen QF, Xiao S, Qi W, Mishra G, Ma J, Wang M, Chye ML (2010) The Arabidopsis acbp1acbp2 double mutant lacking acyl-CoA-binding proteins ACBP1 and ACBP2 is embryo lethal. New Phytol 186:843–855. https://doi.org/10.1111/j.1469-8137.2010.03231.x
Chen WW, Yu XH, Zhang KS, Shi JX, De Oliveira S, Schreiber L, Shanklin J, Zhang DB (2011) Male Sterile 2 encodes a plastid-localized fatty acyl carrier protein reductase required for pollen exine development in Arabidopsis. Plant Physiol 157:842–853. https://doi.org/10.1104/pp.111.181693
Chu FF, Cheng J, Zhang XD, Ye Q, Chen ST, Zhou JH, Cen KF (2019) Transcriptome and key gene expression related to carbon metabolism and fatty acid synthesis of Chlorella vulgaris under a nitrogen starvation and phosphorus repletion regime. J Applied Phycol 315:2881–2893. https://doi.org/10.1007/s10811-019-01811-y
Ciampitti IA, Murrell ST, Camberato JJ, Tuinstra M, Xia Y, Friedemann P, Vyn TJ (2013) Physiological dynamics of maize nitrogen uptake and partitioning in response to plant density and nitrogen stress factors: II Reproductive phase. Crop Sci 53:2588–2602. https://doi.org/10.2135/cropsci2013.01.0041
Cui HY, Camberato JJ, Jin LB, Zhang JW (2015) Effects of shading on spike differentiation and grain yield formation of summer maize in the field. Int J Biometeorol 59:1189–1200. https://doi.org/10.1007/s00484-014-0930-5
DeBruin JL, Hemphill B, Schussler JR (2018) Silk development and kernel set in maize as related to nitrogen stress. Crop Sci 586:2581–2592. https://doi.org/10.2135/cropsci2018.03.0160
Djukanovic V, Smith J, Lowe K, Yang M, Gao H, Jones S, Nicholson MG, West A, Lape J, Bidney D, Falco SC, Jantz D, Alexander LA (2013) Male-sterile maize plants produced by targeted mutagenesis of the cytochrome P450-like gene MS26 using a re-designed I-Crel homing endonuclease. Plant J 765:888–899. https://doi.org/10.1111/tpj.12335
Domínguez E, Heredia-Guerrero JA, Heredia A (2015) Plant cutin genesis: unanswered questions. Trends Plant Sci 209:551–558. https://doi.org/10.1016/j.tplants.2015.05.009
Gaude N, Brehelin C, Tischendorf G, Kessler F, Dormann P (2007) Nitrogen deficiency in Arabidopsis affects galactolipid composition and gene expression and results in accumulation of fatty acid phytyl esters. Plant J 49(729–39):28. https://doi.org/10.1111/j.1365-313X.2006.02992.x
Gozlekci S, Uzun HI, Tepe S (2011) Determination of pollen viability, germination, and quantitiy in loquat cultivars. Acta Horticult 887:281–284. https://doi.org/10.17660/ActaHortic.2011.887.48
Haegele JW, Below FE (2013) Transgenic corn rootworm protection increases grain yield and nitrogen use of maize. Crop Sci 53(5):85–594. https://doi.org/10.2135/cropsci2012.06.0348
Haegele JW, Cook KA, Nichols DM, Below FE (2013) Changes in nitrogen use traits associated with genetic improvement for grain yield of maize hybrids released in different decades. Crop Sci 53:1256–1268. https://doi.org/10.2135/cropsci2012.07.0429
Haslam TM, Haslam R, Thoraval D, Pascal S, Delude C, Domergue F, Fernandez AM, Beaudoin F, Napier JA, Kunst L, Joubès J (2015) ECERIFERUM2-LIKE proteins have unique biochemical and physiological functions in very-long-chain fatty acid elongation. Plant Physiol 167:682–692. https://doi.org/10.1104/pp.114.253195
Hu J, Ren B, Dong ST, Liu P, Zhao B, Zhang JW (2020) Comparative proteomic analysis reveals that exogenous 6-benzyladenine (6-BA) improves the defense system activity of waterlogged summer maize. BMC Plant Biol 20:1–19. https://doi.org/10.1186/s12870-020-2261-5
Hülskamp M, Kopczak SD, Horejsi TF, Kihl BK, Pruitt RE (1995) Identification of genes required for pollen-stigma recognition in Arabidopsis thaliana. Plant J 8:703–714. https://doi.org/10.1046/j.1365-313X.1995.08050703.x
Iwatsuki K, Raven PH (2012) Evolution and diversification of land plants. Springer Science & Business Media pp, pp 99–119. https://doi.org/10.1007/978-4-431-65918-1_2
Jaynes DB, Colvin TS, Karlen DL, Cambardella CA, Meek DW (2001) Nitrate loss in subsurface drainage as affected by nitrogen fertilizer rate. J Environ Qual 30:1305–1314. https://doi.org/10.2134/jeq2001.3041305x
Kim HU, Huang AHC (2004) Plastid lysophosphatidyl acyltransferase is essential for embryo development in Arabidopsis. Plant Physiol 134:1206–1216. https://doi.org/10.1104/pp.103.035832
Kim HU, Li Y, Huang AH (2005) Ubiquitous and endoplasmic reticulum-located lysophosphatidyl acyltransferase, LPAT2, is essential for female but not male gametophyte development in Arabidopsis. Plant Cell 17:1073–1089. https://doi.org/10.1105/tpc.104.030403
Kim J, Lee SB, Suh MC (2021) Arabidopsis 3-Ketoacyl-CoA synthase 4 is essential for root and pollen tube growth. J Plant Biol 642:155–165. https://doi.org/10.1007/s12374-020-09288-w
Li C, Haslam TM, Krüger A, Schneider LM, Mishina K, Samuels L, Yang H, Kunst L, Schaffrath U, Nawrath C, Chen G, Komatsuda T, von Wettstein-Knowles P (2018) The β-ketoacyl-CoA synthase HvKCS1, encoded by Cer-zh, plays a key role in synthesis of barley leaf wax and germination of barley powdery mildew. Plant Cell Physiol 594:811–827. https://doi.org/10.1093/pcp/pcy020
Li H, Pinot F, Sauveplane V, Werck-Reichhart D, Diehl P, Schreiber L, Franke R, Zhang P, Chen L, Gao YW, Liang WQ, Zhang DB (2010) Cytochrome P450 family member CYP704B2 catalyzes the omega-hydroxylation of fatty acids and is required for anther cutin biosynthesis and pollen exine formation in rice. Plant Cell 22:173–190. https://doi.org/10.1105/tpc.109.070326
Li H, Zhang D (2010) Biosynthesis of anther cuticle and pollen exine in rice. Plant Signal Behav 5:1121–1123. https://doi.org/10.4161/psb.5.9.12562
Li L (2015) The major metabolic pathways at maize developing young tassei in response to drought stress and identitication of drought-tolerant candidate SNAC genes. PhD dissertation. Xinjiang Agricultural University, Xinjiang, China
Li Q, Qi LY, Yin LN, Wang SW, Deng XP (2019) Relationship between lipid and flag leaf senescence induced by low nitrogen stress during grain filling of wheat. Acta Agron Sin 44(1221–8):30. https://doi.org/10.3724/SP.J.1006.2018.01221
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCT method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Lolle SJ, Hsu W, Pruitt RE (1998) Genetic analysis of organ fusion in Arabidopsis thaliana. Genet 149:607–619. https://doi.org/10.1093/genetics/149.2.607
Lolle SJ, Pruitt RE (1999) Epidermal cell interactions: a case for local talk. Trends Plant Sci 4:14–20. https://doi.org/10.1016/S1360-1385(98)01353-3
Magneschi L, Kudahettige RL, Alpi A, Perata P (2009) Expansin gene expression and anoxic coleoptiles elongation in rice cultivars. J Plant Physiol 166:1576–1580. https://doi.org/10.1016/j.jplph.2009.03.008
Narasimhan R, Wang GL, Li MY, Roth M, Welti R, Wang XM (2013) Differential changes in galactolipid and phospholipid species in soybean leaves and roots under nitrogen deficiency and after nodulation. Phytochemistry 96:81–91. https://doi.org/10.1016/j.phytochem.2013.09.026
Post-Beittenmiller D (1996) Biochemistry and molecular biology of wax production in plants. Annu Rev Plant Biol 47:405–430
Rossini MA, Maddonni GA, Otegui ME (2012) Interplant variability in maize crops grown under contrasting N × stand density combinations: links between development, growth and kernel set. Field Crops Res 133:90–100. https://doi.org/10.1016/j.fcr.2012.03.010
Rossini MA, Maddonni GA, Otegui ME (2016) Multiple abiotic stresses on maize grain yield determination: additive vs multiplicative effects. Field Crops Res 198:280–289. https://doi.org/10.1016/j.fcr.2016.07.004
Shi J, Tan H, Yu XH, Liu Y, Liang W, Ranathunge K, Franke RB, Schreiber L, Wang Y, Kai G, Shanklin J, Ma H, Zhang DB (2011) Defective pollenwall is required for anther and microspore development in rice andencodes a fatty acyl carrier protein reductase. Plant Cell 23:2225–2246. https://doi.org/10.1105/tpc.111.087528
van der Schoot C, Dietrich MA, Storms M, Verbeke JA, Lucas WJ (1995) Establishment of a cell-to-cell communication pathway between separate carpels during gynoecium development. Planta 195:450–455. https://doi.org/10.1002/1521-3773(20010716)40:143.0.CO;2-Q
Wallis JG, Browse J (2002) Mutants of Arabidopsis reveal many roles for membrane lipids. Prog Lipid Res 41:254–278. https://doi.org/10.1016/S0163-7827(01)00027-3
Wan XY, Wu SW (2020) Molecular cloning of genic male-sterility genes and their applications for plant heterosis via biotechnology-based male-sterility systems. Synthetic Biology-New Interdisciplinary Sci 75–102. https://doi.org/10.5772/intechopen.86976
Wan XY, Wu SW, Li ZW, An XL, Tian YH (2020) Lipid metabolism: critical roles in male fertility and other aspects of reproductive development in plants. Mol Plant 137:955–983. https://doi.org/10.1016/j.molp.2020.05.009
Wang F, Yuan ZJ, Zhao ZW, Li CX, Zhang X, Liang HF, Liu YW, Xu Q, Liu HT (2020) Tasselseed5 encodes a cytochrome C oxidase that functions in sex determination by affecting jasmonate catabolism in maize. J Int Plant Biol 62:247–255. https://doi.org/10.1111/jipb.12826
Wang WJ, Liu XW, Gai XS, Ren JJ, Liu XF, Cai YL, Wang Q, Ren HZ (2015) Cucumis sativus L. WAX2 plays a pivotal role in wax biosynthesis, influencing pollen fertility and plant biotic and abiotic stress responses. Plant Cell Physiol 567:1339–1354. https://doi.org/10.1093/pcp/pcv052
Wolters-Arts M, Lush WM, Mariani C (1998) Lipids are required for directional pollen-tube growth. Nature 392:818–821. https://doi.org/10.1038/33929
Xiao YG, Chen Y, Charnikhova T, Mulder PP, Heijmans J, Hoogenboom A, Agalou A, Michel C, Morel JB, Dreni L, Kater MM, Bouwmeester H, Wang M, Zhu Z, Ouwerkerk PBF (2014) OsJAR1 is required for JA-regulated floret opening and anther dehiscence in rice. Plant Mol Biol 86:19–33. https://doi.org/10.1007/s11103-014-0212-y
Zhang DB, Shi JX, Yang XJ (2016) Role of lipid metabolism in plant pollen exine development. In: YN Akamura, Y Li-Beisson (eds) Lipids in plant and algae development. Springer, Cham, pp 315–337. https://doi.org/10.1007/978-3-319-25979-6_13
Zhang S, Wu S, Niu C, Liu D, Yan T, Tian Y, Liu SS, Xie K, Li ZW, Wang YB, Zhao W, Dong ZY, Zhu TT, Hou Q, Ma B, An XL, Li JP, Wan X (2021) ZmMs25 encoding a plastid–localized fatty acyl reductase is critical for anther and pollen development in maize. J Exp Bot 7212:4298–4318. https://doi.org/10.1093/jxb/erab142
Zhu S, Feng P, Feng J, Feng J, Xu J, Wang ZM, Xu JL, Yuan ZH (2017) The roles of starch and lipid in Chlorella sp. during cell recovery from nitrogen starvation. Bioresource Technol 247:58–65. https://doi.org/10.1016/j.biortech.2017.08.120
Funding
This research was funded by the National Key R&D Projects (NK202218080314), China Agriculture System of MOF and MARA (CARS-02), and Shandong Central Guiding the Local Science and Technology Development (YDZX20203700002548).
Author information
Authors and Affiliations
Contributions
J.W.Z. conceived the original screening and research plans and supervised the experiments; N.N.Y. performed most of the experiments, analyzed the data, and wrote the article with contributions of all the authors; J.W.Z. and B.Z.R. supervised and complemented the writing. S.T.D., P.L,. and B.Z. helped with the discussion and revised the manuscript. All authors approved the final manuscript.
Corresponding author
Ethics declarations
Consent to participate and for publication
All authors consent to participate and for publication of the manuscript.
Conflicts of interest
The authors declare that there are no conflicts of interest or competing interests.
Additional information
Responsible Editor: Ad C. Borstlap.
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yu, N., Ren, B., Zhao, B. et al. Nitrogen rate effects reproductive development and grain set of summer maize by influencing fatty acid metabolism. Plant Soil 487, 341–354 (2023). https://doi.org/10.1007/s11104-023-05930-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-023-05930-x