Abstract
Aims
Bradyrhizobium diazoefficiens is a soil bacterium capable of establishing nitrogen-fixing symbiosis with soybean plants, an important crop for food production worldwide. This interaction is under the control of several regulatory factors that affect the bacterial efficiency to nodulate legume plants and fix nitrogen within root nodules. To date, the function of the NtrYX two-component system in B. diazoefficiens remains unclear. In this work, we investigated the role of NtrY in the symbiotic interaction of B. diazoefficiens with soybean plants.
Methods
We constructed a non-polar mutant of the sensor protein NtrY of the system, and then analysed the symbiosis of this mutant with soybean plants.
Results
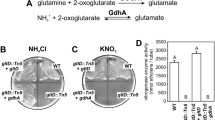
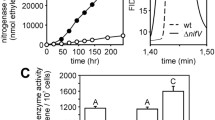
We found that the ntrY mutant was defective in plant dry weight, nitrogen content and nitrogenase activity. Haem c-staining of cbb3 oxidase components showed a clear reduction in the expression of this terminal oxidase in ntrY bacteroids with respect to wild type bacteroids. Such defect in cbb3 expression correlated with a decreased respiratory capacity of the bacteroids produced by the ntrY mutant.
Conclusions
These results suggest that the role of B. diazoefficiens NtrYX in symbiotic nitrogen fixation might be a consequence of its involvement in cbb3 oxidase expression in bacteroids.





Similar content being viewed by others
Abbreviations
- CFU:
-
Colony formation units
- HK:
-
Histidine kinase
- Lb:
-
leghaemoglobin
- LB:
-
Luria–Bertani medium
- NDWP:
-
Nodule dry weight per plant
- NNP:
-
Nodule number per plant
- OD500:
-
Optical density-500 nm
- PPF:
-
Photosynthetic Photon Flux
- PSY:
-
Peptone–salts–yeast extract
- RR:
-
Response regulator
- SDWP:
-
Shoot dry weight per plant
- TCS:
-
Two component system
- TMPD:
-
N,N,N′,N′-tetramethyl-p-phenylenediamine
- WT:
-
Wild-type
- YEM:
-
Yeast-extract-mannitol medium
References
Atack JM, Srikhanta YN, Djoko KY, Welch JP, Hasri NHM, Steichen CT, Vanden Hoven RN, Grimmond SM, Othman DSMP, Kappler U, Apicella MA, Jennings MP, Edwards JL, McEwan AG (2013) Characterization of an ntrX mutant of Neisseria gonorrhoeae reveals a response regulator that controls expression of respiratory enzymes in oxidase-positive Proteobacteria. J Bacteriol 195:2632–2641. https://doi.org/10.1128/jb.02062-12
Babst M, Hennecke H, Fischer HM (1996) Two different mechanisms are involved in the heat-shock regulation of chaperonin gene expression in Bradyrhizobium japonicum. Mol Microbiol 19:827–839
Bauer E, Kaspar T, Fischer H-M, Hennecke H (1998) Expression of the fixR-nifA operon in Bradyrhizobium japonicum depends on a new response regulator, RegR. J Bacteriol 180:3853–3863
Bonato P, Alves LR, Osaki JH, Rigo LU, Pedrosa FO, Souza EM, Zhang N, Schumacher J, Buck M, Wassem R, Chubatsu LS (2016) The NtrY–NtrX two-component system is involved in controlling nitrate assimilation in Herbaspirillum seropedicae strain SmR1. FEBS J 283:3919–3930. https://doi.org/10.1111/febs.13897
Bott M, Ritz D, Hennecke H (1991) The Bradyrhizobium japonicum cycM gene encodes a membrane-anchored homolog of mitochondrial cytochrome c. J Bacteriol 173:6766–6772
Bühler D, Rossmann R, Landolt S, Balsiger S, Fischer H-M, Hennecke H (2010) Disparate pathways for the biogenesis of cytochrome oxidases in Bradyrhizobium japonicum. J Biol Chem 285:15704–15713
Calatrava-Morales N, Nogales J, Ameztoy K, van Steenbergen B, Soto MJ (2017) The NtrY/NtrX system of Sinorhizobium meliloti GR4 regulates motility, EPS I production, and nitrogen metabolism but is dispensable for symbiotic nitrogen fixation. Mol Plant-Microbe Interact 30:566–577. https://doi.org/10.1094/mpmi-01-17-0021-r
Carrica MC, Fernandez I, Martí MA, Paris G, Goldbaum FA (2012) The NtrY/X two-component system of Brucella spp. acts as a redox sensor and regulates the expression of nitrogen respiration enzymes. Mol Microbiol 85:39–50. https://doi.org/10.1111/j.1365-2958.2012.08095.x
Carrica MC, Fernandez I, Sieira R, Paris G, Goldbaum FA (2013) The two-component systems PrrBA and NtrYX co-ordinately regulate the adaptation of Brucella abortus to an oxygen-limited environment. Mol Microbiol 88:222–233. https://doi.org/10.1111/mmi.12181
Cheng Z, Lin M, Rikihisa Y (2014) Ehrlichia chaffeensis proliferation begins with NtrY/NtrX and PutA/GlnA upregulation and CtrA degradation induced by proline and glutamine uptake. MBio 5:e02141. https://doi.org/10.1128/mBio.02141-14
Cogo C, Pérez-Giménez J, Rajeswari CB, Luna MF, Lodeiro AR (2018) Induction by Bradyrhizobium diazoefficiens of different pathways for growth in D-mannitol or L-arabinose leading to pronounced differences in CO2 fixation, O2 consumption, and lateral-flagellum production. Front Microbiol 5(9):1189. https://doi.org/10.3389/fmicb.2018.01189
Čuklina J, Hahn J, Imakaev M, Omasits U, Förstner KU, Ljubimov N, Goebel M, Pessi G, Fischer H-M, Ahrens CH, Gelfand MS, Evguenieva-Hackenberg E (2016) Genome-wide transcription start site mapping of Bradyrhizobium japonicum grown free-living or in symbiosis – a rich resource to identify new transcripts, proteins and to study gene regulation. BMC Genomics 17:302. https://doi.org/10.1186/s12864-016-2602-9
Dakora FD (1995) A functional relationship between Leghaemoglobin and Nitrogenase based on novel measurements of the two proteins in legume root nodules. Ann Bot 75:49–54. https://doi.org/10.1016/s0305-7364(05)80008-3
Delgado MJ, Bedmar EJ, Downie JA (1998) Genes involved in the formation and assembly of rhizobial cytochromes and their role in symbiotic nitrogen fixation. Adv Microb Physiol 40:191–231
Delgado MJ, Bonnard N, Tresierra-Ayala A, Bedmar EJ, Muller P (2003) The Bradyrhizobium japonicum napEDABC genes encoding the periplasmic nitrate reductase are essential for nitrate respiration. Microbiology 149:3395–3403
Dixon R, Kahn D (2004) Genetic regulation of biological nitrogen fixation. Nat Rev Microbiol 2:621–631
Downie JA (2005) Legume haemoglobins: symbiotic nitrogen fixation needs bloody nodules. Curr Biol 15:R196–R198. doi: S0960-9822(05)00268-X [pii]. https://doi.org/10.1016/j.cub.2005.03.007
Drepper T, Wiethaus J, Giaourakis D, Groß S, Schubert B, Vogt M, Wiencek Y, McEwan AG, Masepohl B (2006) Cross-talk towards the response regulator NtrC controlling nitrogen metabolism in Rhodobacter capsulatus. FEMS Microbiol Lett 258:250–256. https://doi.org/10.1111/j.1574-6968.2006.00228.x
Elsen S, Swem LR, Swem DL, Bauer CE (2004) RegB/RegA, a highly conserved redox-responding global two-component regulatory system. Microbiol Mol Biol Rev 68:263–279. https://doi.org/10.1128/mmbr.68.2.263-279.2004
Evans CGT, Herbert D, Tempest DW (1970) The continuous cultivation of microorganisms. 2. Construction of a chemostat. Methods Microbiol 2:275–327
Fedorova E, Thomson R, Whitehead LF, Maudoux O, Udvardi MK, Day DA (1999) Localization of H+-ATPases in soybean root nodules. Planta 209:25–32. https://doi.org/10.1007/s004250050603
Fernández I, Cornaciu I, Carrica MC, Uchikawa E, Hoffmann G, Sieira R, Márquez JA, Goldbaum FA (2017) Three-dimensional structure of full-length NtrX, an unusual member of the NtrC family of response regulators. J Mol Biol 429:1192–1212
Fernández I, Sycz G, Goldbaum FA, Carrica MDC (2018) Acidic pH triggers the phosphorylation of the response regulator NtrX in alphaproteobacteria. PLoS One 10 13(4):e0194486
Fischer HM (1994) Genetic regulation of nitrogen fixation in rhizobia. Microbiol Rev 58:352–386
Gage DJ (2004) Infection and invasion of roots by symbiotic, nitrogen-fixing rhizobia during nodulation of temperate legumes. Microbiol Mol Biol Rev 68:280–300. https://doi.org/10.1128/mmbr.68.2.280-300.2004
Graham PH, Vance CP (2003) Legumes: importance and constraints to greater use. Plant Physiol 131:872–877. https://doi.org/10.1104/pp.017004
Gregor J, Zeller T, Balzer A, Haberzettl K, Klug G (2007) Bacterial regulatory networks include direct contact of response regulator proteins: interaction of RegA and NtrX in Rhodobacter capsulatus. J Mol Microbiol Biotechnol 13:126–139
Hardy RWF, Holsten RD, Jackson EK, Burns RC (1968) The acetylene-ethylene assay for N2 fixation: laboratory and field evaluation. Plant Physiol 43:1185–1207
Hardy RWF, Burns RC, Holsten RD (1973) Application of the acetylene-ethylene assay for measurement of nitrogen fixation. Soil Biol Biochem 5:47–81
Hauser F, Lindemann A, Vuilleumier S, Patrignani A, Schlapbach R, Fischer HM, Hennecke H (2006) Design and validation of a partial-genome microarray for transcriptional profiling of the Bradyrhizobium japonicum symbiotic gene region. Mol Gen Genomics 275:55–67. https://doi.org/10.1007/s00438-005-0059-7
Hu Y, Ribbe MW (2016) Maturation of nitrogenase cofactor—the role of a class E radical SAM methyltransferase NifB. Curr Opin Chem Biol 31:188–194. https://doi.org/10.1016/j.cbpa.2016.02.016
Ishida ML, Assumpção MC, Machado HB, Benelli EM, Souza EM, Pedrosa FO (2002) Identification and characterization of the two-component NtrY/NtrX regulatory system in Azospirillum brasilense. Braz J Med Biol Res 35:651–661
LaRue TA, Child JJ (1979) Sensitive fluorometric assay for leghemoglobin. Anal Biochem 92:11–15. https://doi.org/10.1016/0003-2697(79)90618-3
Lee CC, Ribbe MW, Hu Y (2014) Cleaving the N,N triple bond: the transformation of dinitrogen to Ammonia by Nitrogenases. In: Kroneck PMH, Torres MES (eds) The metal-driven biogeochemistry of gaseous compounds in the environment. Springer Netherlands, Dordrecht
Lindemann A, Moser A, Pessi G, Hauser F, Friberg M, Hennecke H, Fischer H-M (2007) New target genes controlled by the Bradyrhizobium japonicum two-component regulatory system RegSR. J Bacteriol 189:8928–8943. https://doi.org/10.1128/jb.01088-07
Lodeiro AR, Lopez-Garcia SL, Vazquez TE, Favelukes G (2000) Stimulation of adhesiveness, infectivity, and competitiveness for nodulation of Bradyrhizobium japonicum by its pretreatment with soybean seed lectin. FEMS Microbiol Lett 188:177–184
López MF, Cabrera JJ, Salas A, Delgado MJ, López-García SL (2017) Dissecting the role of NtrC and RpoN in the expression of assimilatory nitrate and nitrite reductases in Bradyrhizobium diazoefficiens. Antonie Van Leeuwenhoek 110:531–542. https://doi.org/10.1007/s10482-016-0821-3
Mesa S, Alche Jd JD, Bedmar E, Delgado MJ (2004) Expression of nir, nor and nos denitrification genes from Bradyrhizobium japonicum in soybean root nodules. Physiol Plant 120:205–211. https://doi.org/10.1111/j.0031-9317.2004.0211.x
Mesa S, Ucurum Z, Hennecke H, Fischer H-M (2005) Transcription activation in vitro by the Bradyrhizobium japonicum regulatory protein FixK2. J Bacteriol 187:3329–3338. https://doi.org/10.1128/jb.187.10.3329-3338.2005
Mesa S, Hauser F, Friberg M, Malaguti E, Fischer H-M, Hennecke H (2008) Comprehensive assessment of the regulons controlled by the FixLJ-FixK2-FixK1 Cascade in Bradyrhizobium japonicum. J Bacteriol 190:6568–6579. https://doi.org/10.1128/jb.00748-08
Miller JH (1972) Experiments in molecular genetics. Cold Spring Harbor Laboratory, New York
Minchin FR, James EK, Becana M (2008) Oxygen diffusion, production of reactive oxygen and nitrogen species, and antioxidants in legume nodules. In: MJ JED, Sprent JI, Newton WE (eds) Leguminous Nitrogen-Fixing Symbioses. Springer, Dordrecht
Mongiardini EJ, Quelas JI, Dardis C, Althabegoiti MJ, Lodeiro AR (2017) Transcriptional control of the lateral-flagellar genes of Bradyrhizobium diazoefficiens. J Bacteriol 199. https://doi.org/10.1128/jb.00253-17
Nogales J, Campos R, Ben Abdelkhalek H, Olivares J, Lluch C, Sanjuan J (2002) Rhizobium tropici genes involved in free-living salt tolerance are required for the establishment of efficient nitrogen-fixing Symbiosis with Phaseolus vulgaris. Mol Plant-Microbe Interact 15:225–232. https://doi.org/10.1094/mpmi.2002.15.3.225
Panzieri M, Marchettini N, Hallam TG (2000) Importance of the Bradhyrizobium japonicum symbiosis for the sustainability of a soybean cultivation. Ecol Model 135:301–310. https://doi.org/10.1016/S0304-3800(00)00383-5
Patriarca EJ, Tate R, Ferraioli S, Iaccarino M (2004) Organogenesis of legume root nodules. Int Rev Cytol 234:201–262. https://doi.org/10.1016/S0074-7696(04)34005-2
Pawlowski K, Klosse U, de Bruijn FJ (1991) Characterization of a novel Azorhizobium caulinodans ORS571 two-component regulatory system, NtrY/NtrX, involved in nitrogen fixation and metabolism. Mol Gen Genet MGG 231:124–138. https://doi.org/10.1007/bf00293830
Pérez-Giménez J, Mongiardini EJ, Althabegoiti MJ, Covelli J, Quelas JI, López-García SL, Lodeiro AR (2009) Soybean lectin enhances biofilm formation by Bradyrhizobium japonicum in the absence of plants. Int J Microbiol 2009:8–8. https://doi.org/10.1155/2009/719367
Pessi G, Ahrens CH, Rehrauer H, Lindemann A, Hauser F, Fischer HM, Hennecke H (2007) Genome-wide transcript analysis of Bradyrhizobium japonicum bacteroids in soybean root nodules. Mol Plant-Microbe Interact 20:1353–1363. https://doi.org/10.1094/MPMI-20-11-1353
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res. 1 29(9):e45
Preisig O, Anthamatten D, Hennecke H (1993) Genes for a microaerobically induced oxidase complex in Bradyrhizobium japonicum are essential for a nitrogen-fixing endosymbiosis. Proc Natl Acad Sci U S A 90:3309–3313
Preisig O, Zufferey R, Thöny-Meyer L, Appleby CA, Hennecke H (1996) A high-affinity cbb 3-type cytochrome oxidase terminates the symbiosis-specific respiratory chain of Bradyrhizobium japonicum. J Bacteriol 178:1532–1538. https://doi.org/10.1128/jb.178.6.1532-1538.1996
Quelas JI, Mongiardini EJ, Casabuono A, Lopez-Garcia SL, Althabegoiti MJ, Covelli JM, Perez-Gimenez J, Couto A, Lodeiro AR (2010) Lack of galactose or galacturonic acid in Bradyrhizobium japonicum USDA 110 exopolysaccharide leads to different symbiotic responses in soybean. Mol Plant-Microbe Interact 23:1592–1604. https://doi.org/10.1094/MPMI-05-10-0122
Quelas JI, Mongiardini EJ, Perez-Gimenez J, Parisi G, Lodeiro AR (2013) Analysis of two polyhydroxyalkanoate synthases in Bradyrhizobium japonicum USDA 110. J Bacteriol 195:3145–3155. https://doi.org/10.1128/JB.02203-12
Redondo-Nieto M, Lloret J, Larenas J, Barahona E, Navazo A, Martínez-Granero F, Capdevila S, Rivilla R, Martín M (2008) Transcriptional Organization of the Region Encoding the synthesis of the flagellar filament in Pseudomonas fluorescens. J Bacteriol 190:4106–4109. https://doi.org/10.1128/jb.00178-08
Reese MG (2001) Application of a time-delay neural network to promoter annotation in the Drosophila melanogaster genome. Comput Chem 26:51–56
Regensburger B, Hennecke H (1983) RNA polymerase from Rhizobium japonicum. Arch Microbiol 135:103–109
Regensburger B, Meyer L, Filser M, Weber J, Studer D, Lamb JW, Fischer H-M, Hahn M, Hennecke H (1986) Bradyrhizobium japonicum mutants defective in root-nodule bacteroid development and nitrogen fixation. Arch Microbiol 144:355–366. https://doi.org/10.1007/bf00409885
Sambrook J, Russell D (2001) Molecular cloning: a laboratory manual. Cold Spring Harbor Laboratory Press, New York
Schäfer A, Tauch A, Jäger W, Kalinowski J, Thierbach G, Pühler A (1994) Small mobilizable multi-purpose cloning vectors derived from the Escherichia coli plasmids pK18 and pK19: selection of defined deletions in the chromosome of Corynebacterium glutamicum. Gene 145:69–73. https://doi.org/10.1016/0378-1119(94)90324-7
Sciotti M-A, Chanfon A, Hennecke H, Fischer H-M (2003) Disparate oxygen responsiveness of two regulatory cascades that control expression of symbiotic genes in Bradyrhizobium japonicum. J Bacteriol 185:5639–5642. https://doi.org/10.1128/jb.185.18.5639-5642.2003
Shah VK, Brill WJ (1977) Isolation of an iron-molybdenum cofactor from nitrogenase. Proc Natl Acad Sci U S A 74:3249–3253
Simon R, Priefer U, Pühler A (1983) Vector plasmids for in vivo and in vitro manipulation of gram-negative bacteria. In: Pühler A (ed) Molecular genetics of the Bacteria-plant interaction. Springer, Heidelberg
Sukdeo N, Charles TC (2003) Application of crossover-PCR-mediated deletion-insertion mutagenesis to analysis of the bdhA-xdhA2-xdhB2 mixed-function operon of Sinorhizobium meliloti. Arch Microbiol 179:301–304. https://doi.org/10.1007/s00203-003-0532-9
Swem DL, Bauer CE (2002) Coordination of Ubiquinol oxidase and cytochrome cbb 3 oxidase expression by multiple regulators in Rhodobacter capsulatus. J Bacteriol 184:2815–2820. https://doi.org/10.1128/jb.184.10.2815-2820.2002
Talbi C, Sánchez C, Hidalgo-Garcia A, González EM, Arrese-Igor C, Girard L, Bedmar EJ, Delgado MJ (2012) Enhanced expression of Rhizobium etli cbb 3 oxidase improves drought tolerance of common bean symbiotic nitrogen fixation. J Exp Bot 63:5035–5043. https://doi.org/10.1093/jxb/ers101
Thöny-Meyer L, Stax D, Hennecke H (1989) An unusual gene cluster for the cytochrome bc 1 complex in Bradyrhizobium japonicum and its requirement for effective root nodule symbiosis. Cell 57:683–697. https://doi.org/10.1016/0092-8674(89)90137-2
Torres MJ, Hidalgo-Garcia A, Bedmar EJ, Delgado MJ (2013) Functional analysis of the copy 1 of the fixNOQP operon of Ensifer meliloti under free-living micro-oxic and symbiotic conditions. J Appl Microbiol 114:1772–1781. https://doi.org/10.1111/jam.12168
Torres MJ, Argandona M, Vargas C, Bedmar EJ, Fischer HM, Mesa S (2014) The global response regulator RegR controls expression of denitrification genes in Bradyrhizobium japonicum. PLoS One 9:e99011. https://doi.org/10.1371/journal.pone.0099011
Torres-Quesada O, Oruezabal RI, Peregrina A, Jofré E, Lloret J, Rivilla R, Toro N, Jiménez-Zurdo JI (2010) The Sinorhizobium meliloti RNA chaperone Hfq influences central carbon metabolism and the symbiotic interaction with alfalfa. BMC Microbiol 6(10):71. https://doi.org/10.1186/1471-2180-10-71
Udvardi M, Poole PS (2013) Transport and metabolism in legume-rhizobia Symbioses. Annu Rev Plant Biol 64:781–805. https://doi.org/10.1146/annurev-arplant-050312-120235
Vargas C, McEwan AG, Downie JA (1993) Detection of c-type cytochromes using enhanced Chemiluminescence. Anal Biochem 209:323–326. https://doi.org/10.1006/abio.1993.1127
Vincent JM (1974) Root-nodule symbioses with Rhizobium. In: Quispel A (ed) The biology of nitrogen fixation. American Elsevier Publishing Co., New York
Wang D, Xue H, Wang Y, Yin R, Xie F, Luo L (2013) The Sinorhizobium meliloti ntrX gene is involved in succinoglycan production, motility, and symbiotic nodulation on alfalfa. Appl Environ Microbiol 79:7150–7159. https://doi.org/10.1128/AEM.02225-13
Werner D, Mörschel E, Kort R, Mellor RB, Bassarab S (1984) Lysis of bacteroids in the vicinity of the host cell nucleus in an ineffective (fix −) root nodule of soybean (Glycine max). Planta 162:8–16
Wu J, Bauer CE (2008) RegB/RegA, a global redox-responding two-component system. In: Utsumi R (ed) Bacterial signal transduction: networks and drug targets. Springer New York, New York, NY
Ye J, Coulouris G, Zaretskaya I, Cutcutache I, Rozen S, Madden T (2012) Primer- BLAST: a tool to design target-specific primers for polymerase chain reaction. BMC Bioinformatics 13(134). https://doi.org/10.1186/1471-2105-13-134
Acknowledgements
We are grateful to Paula Giménez, Silvana Tongiani and Abel Bortolameotti (members of CPA CONICET at IBBM) and Rubén Bustos (UNLP). We also thank Susana Jurado and Roxana Peralta (Servicio Central de Microscopía, Facultad de Ciencias Veterinarias, UNLP, La Plata, Argentina) for excellent assistance with electron microscopy. We are also grateful to Germán Tortosa (EEZ, CSIC, Granada, Spain) for excellent technical assistance and Cristina Sanchez for fixN primers design. Thanks are also due to A Di Maggio for careful copyediting of the manuscript and Gabriel Robles-Luna for his help in image editing.
Funding
This work was supported by the Agencia Nacional de Promoción de la Investigación Científica y Tecnológica (ANPCyT; project PICT 2013–2864), Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET) and Secyt-UNLP, Argentina. MJD received financial support from the European Regional Development Fund (ERDF), cofinanced grants AGL2013–45087-R and AGL2017–85676-R from the Ministerio de Economía y Competitividad (Spain) and PE2012-AGR1968 from the Junta de Andalucía. MFL was supported by fellowships from CONICET and by a travel grant from Secyt-UNLP. VAH was supported by a fellowship from CONICET. SLG is researcher at CONICET.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Responsible Editor: Ulrike Mathesius.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 21.3 kb)
ESM 2
(DOCX 20.4 kb)
Fig. S1
Construction of a B. diazoefficiens ntrY deletion mutant. (A) Scheme of the strategy followed to obtain the ntrY deletion mutant (LP4489) without alteration of the reading frame. (B) Verification of the deletional mutation by PCR using specific primers on genomic DNA of wild-type (USDA 110) or ntrY mutant (LP 4489) strains. Primers 4489check FW and RV hybridize to genomic regions outside the mutation site generating products of 2678 pb on the wild-type strain and 413 pb if the deletion was successful. 1, PCR product using USDA110 DNA as template; 2, PCR product using LP4489 DNA as template. A DNA ladder (M) was used to verify the size of the PCR fragments obtained. Arrows depict positions and orientation of the PCR primers used for mutagenesis and checking (see Table S1 and Material and methods). (PNG 318 kb)
Fig. S2
Haem-staining relative band intensity analysis. Image analysis of free-living samples was performed using the ImageJ gel plot tool. Results show the relative intensity of FixP-FixO/c1 (A) or CycM (B) bands in the ntrY mutant compared with the USDA110 wild-type strain as the mean and standard error of three independent gels. (PNG 678 kb)
Rights and permissions
About this article
Cite this article
López, M.F., Hegel, V.A., Torres, M.J. et al. The Bradyrhizobium diazoefficiens two-component system NtrYX has a key role in symbiotic nitrogen fixation of soybean plants and cbb3 oxidase expression in bacteroids. Plant Soil 440, 167–183 (2019). https://doi.org/10.1007/s11104-019-04067-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-019-04067-0