Abstract
Aim
A principal goal of grassland management is to minimize the use of artificial fertilizers by maximising the productivity of nitrogen-fixing leguminous plants such as clovers. The objective of this study was to investigate whether a plant-growth-promoting strain of the free-living rhizosphere fungus Trichoderma hamatum (GD12) could be used as a natural and sustainable means of enhancing the competitiveness of white clover (Trifolium repens) while allowing increased productivity of both clover and ryegrass (Lolium perenne) in mixed species systems.
Methods
An assay was conducted in rhizotrons with white clover and ryegrass sown alone and in mixture and in soils inoculated and non-inoculated with GD12. Plant height, growing rate, phenological stage, number of Rhizobium nodules and biomass were assessed. A histological study of Rhizobium nodules and a stable isotopes analysis was conducted to determine the N fixation capacity of white clover.
Results
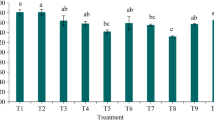
When introduced as a soil inoculant, the fungus increased biomass production of both plant species and shortened their phenological cycles. Furthermore, in clover, GD12 enhanced plant height and growth rate and stimulated Rhizobium nodulation, while 15 N stable isotope analysis demonstrated increased N2 fixation.
Conclusion
This shows that soil amendment with a beneficial strain of saprotrophic fungus bestows a competitive advantage to white clover in clover-ryegrass mixtures and provides a sustainable mechanism for improving the mixture productivity.








Similar content being viewed by others
References
Annicchiarico P, Proietti S (2010) White clover selected for enhanced competitive ability widens the compatibility with grasses and favours the optimization of legume content and forage yield in mown clover-grass mixture. Grass Forage Sci 65:318–324. doi:10.1111/j.1365-2494.2010.00749.x
Annicchiarico P, Tomasoni C (2010) Optimizing legume content and forage yield of mown white clover-Italian ryegrass mixtures through nitrogen fertilization and grass row spacing. Grass Forage Sci 65:220–226. doi:10.1111/j.1365-2494.2010.00740.x
Badar R, Qureshi SA (2012) Comparative effect of Trichoderma hamatum and host-specific Rhizobium species on growth of Vigna mungo. J Appl Pharm Sci 2(4):128–132. doi:10.7324/JAPS.2012.2409
Baker R, Elad Y, Chet I (1984) The controlled experiment in the scientific method with special emphasis on biological control. Phytopathology 74:1019–1021. doi:10.1094/Phyto-74-1019
Borrero C, Trillas MI, Delgado A, Avilés M (2012) Effect of ammonium/nitrate ratio in nutrients solution on control of Fusarium wilt of tomato by Trichoderma asperellum T34. Plant Pathol 61:132–139. doi:10.1111/j.1365-3059.2011.02490.x
Carlsson G, Palmborg C, Jummpponen A, Scherer-Lorenzen M, Högberg P, Huss-Danell K (2009) N2 fixation in three perennial Trifolium species in experimental grasslands of varied plant species richness and composition. Plant Ecol 205:87–104. doi:10.1007/s11258-009-9600-9
Chang YC, Chang YC, Baker R, Kleifeld O, Chet I (1986) Increased growth of plants in the presence of the biological-control agent Trichoderma harzianum. Plant Dis 70(2):145–148. doi:10.1094/PD-70-145
Contreras-Cornejo HA, Macías-Rodríguez L, Cortés-Penagos C, López-Bucio J (2009) Trichoderma virens, a plant beneficial fungus, enhances biomass production and promotes lateral root growth through an auxin-dependent mechanism in Arabidopsis. Plant Physiol 149:1579–1592. doi:10.1104/pp. 108.130369
Crotty FV, Adi SM, Blackshaw RP, Murray PJ (2012) Using stable isotopes to differentiate trophic feeding channels within soil food webs. J Eukaryot Microbiol 59(6):520–526. doi:10.1111/j.1550-7408.2011.00608.x
de Santiago A, Quintero JM, Avilés M, Delgado A (2009) Effect of Trichoderma asperellum strain T34 on iron nutrition in white lupin. Soil Biol Biochem 41:2453–2459. doi:10.1016/j.soilbio.2009.07.033
de Santiago A, Quintero JM, Avilés M, Delgado A (2011) Effect of Trichoderma asperellum strain T34 on iron, copper, manganese, and zinc uptake by wheat grown on a calcareous medium. Plant Soil 342:97–104. doi:10.1007/s11104-010-0670-1
Frame J, Newbould P (1986) Agronomy of white clover. Adv Agron 40:1–88. doi:10.1016/S0065-2113(08)60280-1
Frankow-Lindberg BE, Frame J (1996) Developments and challenges in sustainable grassland production. Grassland Sci Eur 1:337–345
Frey B, Schüepp H (1992) Transfer of symbiotically fixed nitrogen from berseem (Trifolium alexandrinum L.) to maize via vesicular-arbuscularmycorrhizal hyphae. New Phytol 122:447–454. doi:10.1111/j.1469-8137.1992.tb00072.x
Gierus M, Kleen J, Loges R, Taube F (2012) Forage legume species determine the nutritional quality of binary mixture with perennial ryegrass in the first production year. An Feed Sci Technol 172:150–161. doi:10.1016/j.anifeedsci.2011.12.026
Harman GE (2000) Myths and dogmas of biocontrol. Changes in perceptions derived from research on Trichoderma harzianum T-22. Plant Dis 84(4):377–393. doi:10.1094/PDIS.2000.84.4.377
Harman GE, Howell CR, Viterbo A, Chat I, Lorito M (2004) Trichoderma species - opportunistic, avirulent plant symbionts. Nature Rev Microbiol 2:43–56
Harrod TR, Hogan DV (2008) The soils of North Wyke and Rowden. Soil survey of England and Wales. Rothamsted Research, Okehampton, UK http://www.rothamsted.ac.uk/sites/default/files/SoilsNWRowden.pdf Accessed 22 July 2014
Harvey A, Parsons AJ, Rook AJ, Penning PD, Orr RJ (2000) Dietary preference of sheep for perennial ryegrass and white clover at contrasting sward surface heights. Grass Forage Sci 55:242–252
Haynes RJ (1988) Competitive aspects of the grass-legume association. Adv Agron 33:227–262
Høgh-Jensen H, Schjoerring JK (2010) Interactions between nitrogen, phosphorus and potassium determine growth and N2-fixation in white clover and ryegrass leys. Nutr Cycl Agroecosyst 87:327–338. doi:10.1007/s10705-009-9341-0
Johansen A, Jensen ES (1996) Transfer of N and P from intact or decomposing roots of pea to barley interconnected by an arbuscular mycorrhizal fungus. Soil Biol Biochem 28:73–81. doi:10.1016/0038-0717(95)00117-4
Lancashire PD, Bleiholder H, Van Den Boom T, Langeluddeke P, Stauss R, Weber E, Witzenberger A (1991) An uniform decimal code for growth stage crops and weeds. Ann Appl Biol 119:561–601. doi:10.1111/j.1744-7348.1991.tb04895.x
Melino VJ, Drew EA, Ballard RA, Reeve WG, Thomson G, White RG, O’Hara GW (2012) Identifying abnormalities in symbiotic development between Trifolium spp. and Rhizobium leguminosarum v. trifolii leading to sub-optimal and ineffective nodule phenotypes. Ann Bot 110(1559–15):55
Murray PJ, Clements RO (1998) Transfer of nitrogen between clover and wheat: effect of root herbivory. Eur J Soil Biol 34:25–30. doi:10.1016/S1164-5563(99)80003-X
Murray PJ, Hatch DJ (1994) Sitona weevils (Coleoptera, Curculionidae) as agents for rapid transfer of nitrogen from white clover (Trifolium repens L) to perennial ryegrass (Lolium perenne L) Ann. Appl Biol 125:29–33. doi:10.1111/j.1744-7348.1994.tb04943.x
Murray PJ, Clegg CD, Crotty FV, de la Fuente-Martínez N, Williams JK, Blackshaw RP (2009) Dissipation of bacterially derived C and N through the meso and macrofauna of a grassland soil. Soil Biol Biochem 41:1146–1150. doi:10.1016/j.soilbio.2009.02.021
Naseby DC, Pascual JA, Lynch JM (2000) Effect of biocontrol strains of Trichoderma on plant growth, Pythium ultimum populations, soil microbial communities and soil enzyme activities. J Appl Microbiol 88:161–169. doi:10.1046/j.1365-2672.2000.00939.x
Nesheim L, Boller BC (1991) Nitrogen-fixation by white clover when competing with grasses at moderately low-temperatures. Plant Soil 133:47–56. doi:10.1007/BF00011898
Nyfeler D, Huguenin-Elie O, Suter M, Frossard E, Lüscher A (2011) Grass-legume mixture can yield more nitrogen than legume pure stands due to mutual stimulation of nitrogen uptake from symbiotic and non-symbiotic source. Agric Ecosyst Environ 140:155–163. doi:10.1016/j.agee.2010.11.022
Oke V, Long SR (1999) Bacteroid formation in the Rhizobium-legume symbiosis. Curr Opts Microbiol 2:641–646. doi:10.1016/S1369-5274(99)00035-1
Parsons AJ, Chapman DF (2000) The principles of pasture growth and utilization. In: Hopkins A (ed) Grass, its production & utilization, 3rd edn. British Grassland Society by Blackwell Science, Oxford, pp 31–89
Paynel F, Murray PJ, Cliquet JB (2001) Root exudates: a pathway for short-term N transfer from clover and ryegrass. Plant Soil 229(2):235–243. doi:10.1023/A:1004877214831
Peeters A (2009) Importance, evolution, environmental impact and future challenges of grasslands and grassland-based systems in Europe. Grassland Sci 55:113–125. doi:10.1111/j.1744-697X.2009.00154.x
Penning PD, Newman JA, Parsons AJ, Harvey A, Orr RJ (1997) Diet preferences of adult sheep and goats grazing ryegrass and white clover. Small Rumin Res 24:175–184. doi:10.1016/S0921-4488(96)00930-3
Pérez-de-Luque A, Rubiales D, Cubero JI, Press MC, Scholes J, Yoneyama K, Takeuchi Y, Plakhine D, Joel DM (2005) Interaction between Orobanche crenata and its host legumes: unsuccessful haustorial penetration and necrosis of the developing parasite. Ann Bot 95:935–942. doi:10.1093/aob/mci105
Rassmusen J, Eriksen J, Jensen ES, Esbensen KH, Høgh-Jensen H (2007) In situ carbon and nitrogen dynamics in ryegrass-clover mixture: transfer, deposition and leaching. Soil Biol Biochem 39:804–815. doi:10.1016/j.soilbio.2006.10.004
Rassmusen J, Søegaard K, Pirhofer-Walzi K, Eriksen J (2012) N2-fixation and residual N effect of four legume species and four companion grass species. Eur J Agron 36:66–74. doi:10.1016/j.eja.2011.09.003
Romão-Dumaresq AS, de Araújo WL, Talbot NJ, Talbot NJ (2012) RNA interference of endochitinases in the sugarcane endophyte Trichoderma virens 223 reduces its fitness as biocontrol agent of pineapple disease. PLoS ONE 7:e47888. doi:10.1371/journal.pone.0047888
Rook AJ, Harvey A, Parsons AJ, Penning PD, Orr RJ (2002) Effect of long-term changes in relative resource availability on dietary preference of grazing sheep for perennial ryegrass and white clover. Grass Forage Sci 57:54–60. doi:10.1046/j.1365-2494.2002.00301.x
Ruzin SE (1999) Plant microtechniques and microscopy. Oxford University Press, New York, USA
Ryder LS, Harris BD, Soanes DM, Kershaw MJ, Talbot NJ, Thornton CR (2012) Saprotrophic competitiveness and biocontrol fitness of a genetically modified strain of the plant-growth-promoting fungus Trichoderma hamatum GD12. Microbiology 158:84–97. doi:10.1099/mic.0.051854-0
Shaban WI, El-Bramawy MA (2011) Impact of dual inoculation with Rhizobium and Trichoderma on damping off, root rot diseases and plant growth parameters of some legumes field crop under greenhouse conditions. International Res. J Agric Sci Soil Sci 1(3):98–108
Shoresh M, Harman GE, Mastouri F (2010) Induced systemic resistance and plant responses to fungal biocontrol agents. Ann Rev Phytopathol 48:21–43
Sreenivasaprasad S, Sharada K, Brown A, Mills P (1996) PCR-based detection of Colletotrichum acutatum on strawberry. Plant Pathol 45:650–655
Studholme D, Harris B, Le Cocq K, Winsbury R, Perera V, Ryder L, Ward JL, Beale MH, Thornton CR, Grant M (2013) Investigating the beneficial traits of Trichoderma hamatum GD12 for sustainable agriculture – insights from genomics. Front Plant Sci 4:1–13. doi:10.3389/fpls.2013.00258
Talbot NJ, Salch YP, Ma M, Hamer JE (1993) Karyotypic variation within clonal lineages of the rice blast fungus, Magnaporthe grisea. Appl Environ Microbiol 59:585–593
Vance CP, Johnson LEB, Hardarson G (1980) Histological comparisons of plant and Rhizobium induced ineffective nodules in alfalfa. Physiol Plant Pathol 17:167–173. doi:10.1016/0048-4059(80)90049-1
Vargas WA, Mandawe JC, Kenerley CM (2009) Plant-derived sucrose is a key element in the symbiotic association between Trichoderma virens and maize plants. Plant Physiol 151:792–808. doi:10.1104/pp. 109.141291
Vasse J, De Billy F, Camut S, Truchet G (1990) Correlation between ultrastructural differentiation of bacteroids and nitrogen fixation in alfalfa nodules. J Bacteriol 8:4295–4306
Viterbo A, Horwitz BA (2010) Mycoparasitism. In: Borkovich KA, Ebbole DJ (eds) Cellular and biology of filamentous fungi. American Society for Microbiology, Washington, pp 676–693
Walker JA, King JR (2010) Does relative time of emergence affect stand composition and yield in a grass-legume mixture? Kura clover (Trifolium ambiguum)-meadow bromegrass (Bromus biebersteinii) and Kura clover-orchardgrass (Dactylis glomerata) mixtures. Grass Forage Sci 65:237–347. doi:10.1111/j.1365-2494.2010.00742.x
White TJ, Bruns T, Lee S, Taylor J (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. PCR Protocols: Guide Methods Appl 18:315–322
Woledge J, Davidson K, Dennis WD (1992) Growth and photosynthesis of tall and short cultivars of white clover with tall and short grasses. Grass Forage Sci 47:230–238. doi:10.1111/j.1365-2494.1992.tb02267.x
Yedidia I, Srivastva AK, Kapulnik Y, Chet I (2001) Effect of Trichoderma harzianum on microelement concentrations and increased growth of cucumber plants. Plant Soil 235:235–242. doi:10.1023/A:1011990013955
Acknowledgments
The authors would like acknowledge to the Operative Program of European Social Fund (ESF) 2007–2013 “Andalucía se mueve con Europa” and to the project INIA-RTA2011-00031 that funded the contract and stay of C. Alcántara in North Wyke Rothamsted Research. Rothamsted Research is supported by the UK Biotechnology and Biological Sciences Research Council (BBSRC). The authors would to thank Dr. Domínguez for reviewing the statistical methods used in this study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Duncan D. Cameron.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Fig. 1
Cross sections of Rhizobium nodules on roots of white clover. a) General view of a nodule from clover growing in un-inoculated soil. b) Detail of a (square I) showing cells with bacteroids at the infection zone. c) Detail of a (square II) showing infected cells at the nitrogen-fixing zone. d) Detail of a (square III) showing infected cells at the senescence zone. e) Detailed picture of cells from the nitrogen-fixing zone showing bacteroids with rod-shaped form. f) General view of a nodule from clover growing in Trichoderma inoculated soil. g) Detail of f (square I) showing cells with bacteroids at the infection zone. h) Detail of f (square II) showing infected cells at the nitrogen-fixing zone. Notice the presence of cells containing starch and amyloplasts (arrows). i) Detail of f (square III) showing infected cells at the senescence zone. j) Detailed picture of cells from the nitrogen-fixing zone showing bacteroids with rod-shaped form and the presence of amyloplasts inside some cells (arrows) (TIFF 29214 kb)
Fig. 2
Rhizotrons from two replicates of the different species: clover (a, b), ryegrass (c, d) and mixture (e, f); and treatments (with or without T). Notice the differences in plant size between rhizotrons inoculated with GD12 and those un-inoculated (TIFF 567 kb)
Fig. 3
Root morphologies of clover and ryegrass plants grown in soil inoculated with T. hamatum GD12 (+T) or in un-inoculated soil (−T). Scale bar = 7,5 cm. (TIFF 188 kb)
Rights and permissions
About this article
Cite this article
Alcántara, C., Thornton, C.R., Pérez-de-Luque, A. et al. The free-living rhizosphere fungus Trichoderma hamatum GD12 enhances clover productivity in clover-ryegrass mixtures. Plant Soil 398, 165–180 (2016). https://doi.org/10.1007/s11104-015-2646-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-015-2646-7