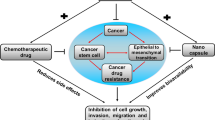
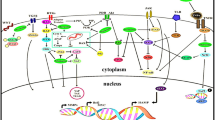
Abstract
The search of alternative therapeutic agents for the use of cancer patients has dramatically expanded. Natural products are especially in focus since their structures already function in nature and are more likely to be potent with fewer side effects. Phloretin is a natural product that has been studied for a wide variety of pharmacological actions against human malignancies. This systematic review aims to present up-to-date critical and comprehensive information on the anti-cancer ability of Phloretin with all associated molecular and cellular mechanisms in various forms of cancers. Data retrieved according to PRISMA guidelines from Science Direct, PubMed, and Scopus searching servers by using keywords including Phloretin, cancer name, synergistic, resistance and Pharmacokinetics property was analyzed via some in silico tools. This systematic review comprised 127 articles from different types of study, where Phloretin is hypothesized to be effective against 20 various forms of cancer. Phloretin has been found to inhibit cancer initiation and progression by modulating many imbalanced signalling pathways, including apoptosis, autophagy, necrosis, metastasis, angiogenesis, cell proliferation, glucose absorption, oxidative stress, inflammation, DNA damage, and many other pathways. This wide range of activity may be due to the structural targeting of numerous proteins including, Bcl-2, Bax, Bak, Bad, caspase, cyclins (B1, D1, E) and CDKs (4, 6,7) p18, p21, p27, p53, MMP-2, MMP- 8, MMP-9, Wnt/-catenin, PARP, TNF-α, NF-κB, IκB kinase, IL-1β, TNF-α, phospho-Akt, phosphor-p65, NF-κB, PI3K/Akt, MAPK/ERK, p-mTOR. The introduction of nano-technology-based strategies can improve the efficacy of Phloretin for cancer treatment. Existing evidence shows that Phloretin has synergistic effects with other natural compounds and conventional drugs, and this mechanism assists in reversing the resistance of anticancer drugs by regulating resistance-related proteins. However, Phloretin possesses favorable pharmacokinetic properties with low toxicity in the human body by in silico methods. Therefore, Phloretin could be a potential anti-cancer drug against numerous cancer treatment by mitigating it's toxic effect and enhancing efficacy using nano-technology-based strategies.










Similar content being viewed by others
Abbreviations
- Bcl-2:
-
B-cell leukemia 2
- Bax:
-
Bcl-2-Associated X protein
- PI3K:
-
Phosphoinositide 3-kinase
- Akt:
-
Ak strain transforming
- mTOR:
-
Mammalian target of rapamycin
- MMP9 :
-
Matrix metalloproteinase 9
- GLUT:
-
Glucose transporters
- ULK1 :
-
Unc-51 like autophagy activating kinase
- ERK1/2:
-
Extracellular signal-regulated kinase 1/2
- TPA :
-
Tissue plasminogen activator
- LC3B :
-
Light chain 3B
- BCL-XL :
-
B-cell lymphoma-extra large
- XIAP :
-
X-linked inhibitor of apoptosis protein
- VEGF :
-
Vascular endothelial growth factor
- MTT :
-
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide
- CAT :
-
Chloramphenicol acetyltransferase
- SOD :
-
Superoxide dismutase
- NF-B :
-
Nuclear factor-B
- Mac-1 :
-
Macrophage 1 antigen
- PEP :
-
Phosphoenolpyruvate
- GraB :
-
Granzyme B
- c-Myc :
-
Cellular Myc
- ROS :
-
Reactive oxygen species
- HCC :
-
Hepatocellular carcinoma
- SHP-1 :
-
Src homology region 2 domain-containing phosphatase-1
- STAT3 :
-
Signal transducer and activator of transcription 3
- pERK :
-
Protein kinase RNA-like endoplasmic reticulum kinase
- VEGFR :
-
Vascular endothelial growth factor receptor
- MAPK :
-
Mitogen-activated protein kinase
- CXCL12 :
-
C-X-C motif chemokine ligand 12
- CXCR4 :
-
C-X-C motif chemokine receptor 4
- TLR1 :
-
Toll-like receptor 1
- TLR2:
-
Toll-like receptor 2
- TNF-α :
-
Tumor necrosis factor-alpha
- NOD :
-
Nucleotide-binding oligomerization domain
- NLRP3:
-
Nucleotide-binding domain (NOD)-like receptor protein 3
- CDK4 :
-
Cyclin-dependent kinase 4
- CDK6 :
-
Cyclin-dependent kinase 6
- BAK :
-
Bcl-2 homologous antagonist/killer
- PARP :
-
Poly-ADP ribose polymerase
- HIF1 :
-
Hypoxia-inducible factor-1
- HKII :
-
Hexokinase 2
- PFKFB3 :
-
6-Phosphofructo-2-kinase/fructose-2,6-bisphosphatase isozyme 3
- PDHK1 :
-
Pyruvate dehydrogenase kinase 1
- LDH :
-
Lactate dehydrogenase
- Pink1 :
-
PTEN-induced kinase 1
- GSH:
-
Glutathione
- NSCLC :
-
Non-small cell lung cancer
- JNK :
-
C-Jun N-terminal kinase
- TRX:
-
Thioredoxin
- TXNIP :
-
Thioredoxin interacting protein
- 2-NBDLG :
-
2-[N-(7-nitrobenz-2-oxa-1,3-diazol-4-yl) amino]-2-deoxy-D-glucose
- PKC:
-
Protein kinase C
- Myt1 :
-
Myelin transcription factor 1
- cdc2:
-
Cyclin-dependent kinase 2
- HSP70:
-
70 Kilodalton heat shock protein
- IL-6:
-
Interleukin-6
- MRP:
-
Multi-drug resistance associated protein 1
- BCRP:
-
Breast cancer resistance protein
- MDR:
-
Multi-drug resistance
References
Abkin SV, Ostroumova OS, Komarova EY, Meshalkina DA, Shevtsov MA, Margulis BA, Guzhova IV (2016) Phloretin increases the anti-tumor efficacy of intratumorally delivered heat-shock protein 70 KDa (HSP70) in a murine model of melanoma. Cancer Immunol Immunother. https://doi.org/10.1007/s00262-015-1778-1
Abu-Azzam O, Aldalaen S, Nasr M (2022) Potentiation of the cytotoxic activity of nutraceutical phloretin against cervical cancer by formulation into microemulsion. Pharm Chem J 55(11):1215–1218
Akhdar H, Legendre C, Aninat C, More F (2012) Anticancer drug metabolism: chemotherapy resistance and new therapeutic approaches. Top Drug Metabol 22:138–170
Alsanani A, Pate BB, Wondisford S, Johnson N, Banerjee D (2015) Stromal carcinoma associated fibroblasts promote drug resistance of human pancreatic cancer cells by modulation of ROS via CXCR4/CXCL12 signaling. Int J Transl Sci. https://doi.org/10.13052/ijts2246-8765.20151006
Anand M, Suresh K (2014) Biochemical Profiling and chemopreventive activity of phloretin on 7,12-dimethylbenz (A) anthracene induced oral carcinogenesis in male golden syrian hamsters. Toxicol Int. https://doi.org/10.4103/0971-6580.139805
Mariadoss A, Vijaya A, Dhanabalan AK, Munusamy H, Gunasekaran K, David E (2018) In silico studies towards enhancing the anticancer activity of phytochemical phloretin against cancer drug targets. Current Drug Therapy. https://doi.org/10.2174/1574885513666180402134054
Anand AV, Mariadoss VR, Baojun Xu, Karthikumar V, Vijayalakshmi S, Shalini V, Rani BS, Mohamed Sadiq A, Ernest D (2019) Phloretin loaded chitosan nanoparticles enhance the antioxidants and apoptotic mechanisms in DMBA induced experimental carcinogenesis. Chem Biol Interact 308:11–19. https://doi.org/10.1016/j.cbi.2019.05.008
Badwaik, Vivek D, Sarah Tockstein, David H Thompson (2018) Development of Dihydrochalcone-Functionalized Gold Nanoparticles for Augmented Antineoplastic Activity, 1917–26
Barreca D, Bellocco E, Laganà G, Ginestra G, Bisignano C (2014) Biochemical and antimicrobial activity of phloretin and its glycosilated derivatives present in apple and kumquat. Food Chem. https://doi.org/10.1016/j.foodchem.2014.03.118
Barreca D, Currò M, Bellocco E, Ficarra S, Laganà G, Tellone E, Giunta ML et al (2017) Neuroprotective effects of phloretin and its glycosylated derivative on rotenone-induced toxicity in human SH-SY5Y neuronal-like cells. BioFactors. https://doi.org/10.1002/biof.1358
Biswas P, Dey D, Biswas PK, Rahaman TI, Saha S, Parvez A, Khan DA, Lily NJ, Saha K, Sohel Md (2022) A comprehensive analysis and anti-cancer activities of quercetin in ROS-mediated cancer and cancer stem cells. Int J Mol Sci 23(19):11746
Cao X, Fang L, Gibbs S, Huang Y, Dai Z, Wen P, Zheng X, Sadee W, Sun D (2007) Glucose uptake inhibitor sensitizes cancer cells to daunorubicin and overcomes drug resistance in hypoxia. Cancer Chemother Pharmacol. https://doi.org/10.1007/s00280-006-0291-9
Casarini TP, Anunciato LA, Frank TB, Onzi G, Pohlmann AR, Guterres SS (2021) Innovative hydrogel containing polymeric nanocapsules loaded with phloretin: enhanced skin penetration and adhesion. Mater Sci Eng, C 120:111681
Casarini TPA, Frank LA, Pohlmann AR, Guterres SS (2020) Dermatological applications of the flavonoid phloretin. Eur J Pharmacol 889:173593
Chang WT, Huang WC, Liou CJ (2012) Evaluation of the anti-inflammatory effects of phloretin and phlorizin in lipopolysaccharide-stimulated mouse macrophages. Food Chem. https://doi.org/10.1016/j.foodchem.2012.03.002
Chen M, Gowd V, Wang M, Chen F, Cheng KW (2021) The apple dihydrochalcone phloretin suppresses growth and improves chemosensitivity of breast cancer cells: via inhibition of cytoprotective autophagy. Food Funct 12(1):177–190. https://doi.org/10.1039/d0fo02362k
Young CB (2019) Biochemical basis of anti-cancer-effects of phloretin—a natural dihydrochalcone. Molecules 24(2):278
Choi I, Kim J, Jeong HK, Kim B, Jou I, Park M, Chen L, Kang UJ, Zhuang X, Eun hye Joe. (2013) PINK1 deficiency attenuates astrocyte proliferation through mitochondrial dysfunction, reduced AKT and increased P38 MAPK activation, and downregulation of EGFR. Glia. https://doi.org/10.1002/glia.22475
Costea T, Vlad OC, Miclea LC, Ganea C, Szöllősi J, Mocanu MM (2020) Alleviation of multidrug resistance by flavonoid and non-flavonoid compounds in breast, lung, colorectal and prostate cancer. Int J Mol Sci. https://doi.org/10.3390/ijms21020401
Crespy V, Aprikian O, Morand C, Besson C, Manach C, Demigné C, Rémésy C (2001) Bioavailability of phloretin and phloridzin in rats. J Nutr 131(12):3227–3230. https://doi.org/10.1093/jn/131.12.3227
Cui D, Liu S, Tang M, Yongzhi Lu, Zhao M, Mao R, Wang C, Yuan Y, Li L, Chen Y (2020) Phloretin ameliorates hyperuricemia-induced chronic renal dysfunction through inhibiting NLRP3 inflammasome and uric acid reabsorption. Phytomedicine 66:153111
Davis ME (2016) Glioblastoma: overview of disease and treatment. Clin J Oncol Nurs 20(5):1–8. https://doi.org/10.1188/16.CJON.S1.2-8
Debela DT, Muzazu SGY, Heraro KD, Ndalama MT, Mesele BW, Haile DC, Kitui SK, Manyazewal T (2021) New approaches and procedures for cancer treatment: current perspectives. SAGE Open Medicine. https://doi.org/10.1177/20503121211034366
Dewanjee S, Sohel M, Hossain MS, Ansari F, Islam MT, Sultana F, Al Mamun A, Islam MM, Amin MN (2023) A Comprehensive Review on Clinically Proven Natural Products in the Management of Nerve Pain, with Mechanistic Insights. Heliyon
Duan H, Wang R, Yan X, Liu H, Zhang Y, Deguang Mu, Han J, Li X (2017) Phloretin induces apoptosis of human esophageal cancer via a mitochondria-dependent pathway. Oncol Lett 14(6):6763–6768
Fan C, Yilin Zhang Yu, Tian XZ, Teng J (2022) Phloretin enhances autophagy by impairing AKT activation and inducing JNK-beclin-1 pathway activation. Exp Mol Pathol 127(July):104814. https://doi.org/10.1016/j.yexmp.2022.104814
Frerichs H, Ball EG (1964) Studies on the metabolism of adipose tissue. XVI. Inhibition by phlorizin and phloretin of the insulin-stimulated uptake of glucose. Biochemistry 3(7):981–985
Friday E, Ledet J, Turturro F (2011) Response to dexamethasone is glucose-sensitive in multiple myeloma cell lines. J Exp Clin Cancer Res 30:1–7
Funari CS, Passalacqua TG, Rinaldo D, Napolitano A, Festa M, Capasso A, Piacente S, Pizza C, Young MCM, Durigan G (2011) Interconverting flavanone glucosides and other phenolic compounds in lippia salviaefolia Cham. Ethanol Extracts. Phytochemistry 72(16):2052–2061
FΑΟ. 2013. Food Wastage Footprint. Fao
Geohagen BC, Korsharskyy B, Vydyanatha A, Nordstroem L, LoPachin RM (2018) Phloretin cytoprotection and toxicity. Chem Biol Interact 296:117–123
Gonzalez-Menendez P, Hevia D, Rodriguez-Garcia A, Mayo JC, Sainz RM (2014) Regulation of GLUT transporters by flavonoids in androgen-sensitive and-insensitive prostate cancer cells. Endocrinology (united States). https://doi.org/10.1210/en.2014-1260
Grigoras CG, Destandau E, Fougère L, Elfakir C (2013) Evaluation of apple pomace extracts as a source of bioactive compounds. Ind Crops Prod. https://doi.org/10.1016/j.indcrop.2013.06.026
Hagl S, Deusser H, Soyalan B, Janzowski C, Will F, Dietrich H, Albert FW, Rohner S, Richling E (2011) Colonic availability of polyphenols and D-(-)-quinic acid after apple smoothie consumption. Mol Nutr Food Res. https://doi.org/10.1002/mnfr.201000252
Han L, Zhang Y, Li J, Xiao Y, Mei Lu, Li Y, Wang M (2021) Phloretin attenuation of hepatic steatosis: via an improvement of mitochondrial dysfunction by activating AMPK-dependent signaling pathways in C57BL/6J mice and HepG2 cells. Food Funct. https://doi.org/10.1039/d1fo02577e
He C, Wang J, Chakraborty T, Roy S (2020) A newfangled coordinated ruthenium phloretin complex reprogramming breast cancer microenvironment interceded by modulation of PI3K/Akt/Mtor/VEGF pathway and modifying the antioxidant status correlated with intensied apoptotic events. https://doi.org/10.21203/rs.3.rs-42460/v1
Hilt P, Schieber A, Yildirim C, Arnold G, Klaiber I, Conrad J, Beifuss U, Carle R (2003) Detection of phloridzin in strawberries (Fragaria x ananassa Duch) by HPLC− PDA− MS/MS and NMR spectroscopy. J Agric Food Chem 51(10):2896–2899. https://doi.org/10.1021/jf021115k
Hsiao Y-H, Hsieh M-J, Yang S-F, Chen S-P, Tsai W-C, Chen P-N (2019) Phloretin suppresses metastasis by targeting protease and inhibits cancer stemness and angiogenesis in human cervical cancer cells. Phytomedicine 62:152964
Huang WC, Fang LW, Liou CJ (2017) Phloretin attenuates allergic airway inflammation and oxidative stress in asthmatic mice. Front Immunol. https://doi.org/10.3389/fimmu.2017.00134
Hui J (2020) Down-regulation of aquaporin 9 by natural product phloretin mediates luminal bladder cancer migration in vitro. Johns Hopkins University
Wang H (2012) Phloretin induced apoptosis of human hepatoma cells SMMC-7721 and its correlative biological mechanisms. Afr J Pharm Pharmacol. https://doi.org/10.5897/ajpp11.869
Jacob V, Hagai T, Soliman K (2011) Structure-activity relationships of flavonoids. Curr Org Chem. https://doi.org/10.2174/138527211796367309
Jin G, Zhao Z, Chakraborty T, Mandal A, Roy A, Roy S, Guo Z (2020) Decrypting the molecular mechanistic pathways delineating the chemotherapeutic potential of ruthenium-phloretin complex in colon carcinoma correlated with the oxidative status and increased apoptotic events. Oxidative Med Cell Longev. https://doi.org/10.1155/2020/7690845
Kang D, Zuo W, Wu Q, Zhu Q, Liu P (2020) Inhibition of specificity protein 1 is involved in phloretin-induced suppression of prostate cancer. BioMed Res Int. https://doi.org/10.1155/2020/1358674
Kapoor S, Padwad YS (2023) Phloretin induces G2/M arrest and apoptosis by suppressing the β-catenin signaling pathway in colorectal carcinoma cells. Apoptosis. https://doi.org/10.1007/s10495-023-01826-4
Karpuz M, Silindir-Gunay M, Ozer AY (2018) Current and future approaches for effective cancer imaging and treatment. Cancer Biother Radiopharm. https://doi.org/10.1089/cbr.2017.2378
Kim J, Durai P, Jeon D, Jung ID, Lee SJ, Park Y-M, Kim Y (2018) Phloretin as a potent natural TLR2/1 inhibitor suppresses TLR2-induced inflammation. Nutrients 10(7):868
Kim J-L, Lee D-H, Cheol-Ho Pan Su, Park J, Sang-Cheul Oh, Lee S-Y (2022a) Role of phloretin as a sensitizer to TRAIL-induced apoptosis in colon cancer. Oncol Lett 24(3):1–13
Kim JL, Lee DH, Pan CH, Park SJ, Oh SC, Lee SY (2022b) Role of phloretin as a sensitizer to TRAIL-induced apoptosis in colon cancer. Oncol Lett. https://doi.org/10.3892/ol.2022.13441
Kim M-S, Kwon JY, Kang NJ, Lee KW, Lee HJ (2009) Phloretin induces apoptosis in H-Ras MCF10A Human breast tumor cells through the activation of P53 via JNK and P38 mitogen-activated protein kinase signaling. Ann N Y Acad Sci 1171(1):479–483
KoBoRI M, Iwashita K, Shinmoto H, Tojiro TSUSHIDA (1999) Phloretin-induced apoptosis in B16 melanoma 4A5 cells and HL60 human leukemia cells. Biosci Biotechnol Biochem 63(4):719–725
Kuruppu AI, Paranagama P, Goonasekara CL (2019) Medicinal plants commonly used against cancer in traditional medicine formulae in Sri Lanka. Saudi Pharmaceut J. https://doi.org/10.1016/j.jsps.2019.02.004
Lee SY, Park JH, Ko SH, Shim JS, Kim DD, Cho HJ (2017) Mussel-inspired hyaluronic acid derivative nanostructures for improved tumor targeting and penetration. ACS Appl Mater Interfaces 9(27):22308–22320. https://doi.org/10.1021/acsami.7b06582
Lee YJ, Kim WI, Kim SY, Cho SW, Nam HS, Lee SH, Cho MK (2019) Flavonoid morin inhibits proliferation and induces apoptosis of melanoma cells by regulating reactive oxygen species, Sp1 and Mcl-1. Arch Pharmacal Res 42(6):531–542. https://doi.org/10.1007/s12272-019-01158-5
Lin J, Sahakian D, de Morais S, Jinghai Xu, Polzer R, Winter S (2005) The role of absorption, distribution, metabolism, excretion and toxicity in drug discovery. Curr Top Med Chem. https://doi.org/10.2174/1568026033452096
Lin ST, Tu SH, Yang PS, Hsu SP, Lee WH, Ho CT, Wu CH, Lai YH, Chen MY, Chen LC (2016) Apple polyphenol phloretin inhibits colorectal cancer cell growth via inhibition of the type 2 glucose transporter and activation of p53-mediated signaling. J Agric Food Chem 64(36):6826–6837. https://doi.org/10.1021/acs.jafc.6b02861
Liu Y, Fan C, Lv Pu, Wei C, Jin H, Teng Y, Zhao M et al (2016) Phloretin induces cell cycle arrest and apoptosis of human glioblastoma cells through the generation of reactive oxygen species. J Neurooncol 128(2):217–223. https://doi.org/10.1007/s11060-016-2107-z
Lu Y, Chen J, Ren D, Yang X, Zhao Y (2017) Hepatoprotective effects of phloretin against CCl4-induced liver injury in mice. Food Hydrocolloids. https://doi.org/10.1080/09540105.2016.1258546
Lynch TJ, Bell DW, Sordella R, Gurubhagavatula S, Okimoto RA, Brannigan BW, Harris PL et al (2004) Activating mutations in the epidermal growth factor receptor underlying responsiveness of non–small-cell lung cancer to Gefitinib. N Engl J Med 350(21):2129–2139. https://doi.org/10.1056/nejmoa040938
Ma L, Wang R, Nan Y, Li W, Wang Q, Jin F (2016) Phloretin exhibits an anticancer effect and enhances the anticancer ability of cisplatin on non-small cell lung cancer cell lines by regulating expression of apoptotic pathways and matrix metalloproteinases. Int J Oncol 48(2):843–853
Ma P, Wu Y, Tao L, Han J (2022) Effect of Gardenia tea on apoptosis of human thyroid cancer cell line SW579 through PI3K-Akt pathway. Food Sci Technol 42:e73122
“Manchurian Apricot Facts and Health Benefits.”. Accessed April 7, 2023. https://www.healthbenefitstimes.com/manchurian-apricot/.
Mansoori B, Mohammadi A, Davudian S, Shirjang S, Baradaran B (2017) The different mechanisms of cancer drug resistance: a brief review. Adv Pharmaceut Bull. https://doi.org/10.15171/apb.2017.041
Mariadoss AVA, Vinyagam R, Rajamanickam V, Sankaran V, Venkatesan S, David E (2019a) Pharmacological aspects and potential use of phloretin: a systemic review. Mini-Rev Med Chem. https://doi.org/10.2174/1389557519666190311154425
Mariadoss AV, Anand RV, Senthilkumar V, Paulpandi M, Murugan K, Baojun Xu, Gothandam KM, Kotakadi VS, David E (2019b) Phloretin loaded chitosan nanoparticles augments the ph-dependent mitochondrial-mediated intrinsic apoptosis in human oral cancer cells. Int J Biol Macromol 130(June):997–1008. https://doi.org/10.1016/J.IJBIOMAC.2019.03.031
Matsui C, Takatani-Nakase T, Maeda S, Nakase I, Takahashi K (2017) Potential roles of GLUT12 for glucose sensing and cellular migration in MCF-7 human breast cancer cells under high glucose conditions. Anticancer Res 37(12):6715–6722. https://doi.org/10.21873/anticanres.12130
McBrayer SK, Cheng JC, Singhal S, Krett NL, Rosen ST, Shanmugam M (2012) Multiple myeloma exhibits novel dependence on GLUT4, GLUT8, and GLUT11: implications for glucose transporter-directed therapy. Blood J Am Soc Hematol 119(20):4686–4697
Mehmood S, Haojie Yu, Li Wang Md, Uddin A, Amin BU, Haq F, Fahad S, Haroon M (2022) Cross-linked poly(cyclotriphosphazene-co-phloretin) microspheres and their application for controlled drug delivery. Macromol Res 30(9):623–630. https://doi.org/10.1007/S13233-022-0066-0/METRICS
Min J, Li Xu, Huang K, Tang H, Ding X, Qi C, Qin X, Zhifei Xu (2015) Phloretin induces apoptosis of non-small cell lung carcinoma A549 cells via JNK1/2 and P38 MAPK pathways. Oncol Rep 34(6):2871–2879
Minsat L, Peyrot C, Brunissen F, Renault JH, Allais F (2021) Synthesis of biobased phloretin analogues: an access to antioxidant and anti-tyrosinase compounds for cosmetic applications. Antioxidants. https://doi.org/10.3390/antiox10040512
Mintie CA, Singh CK, Ahmad N (2020) Whole fruit phytochemicals combating skin damage and carcinogenesis. Transl Oncol 13(2):146–156
Molnár J, Engi H, Hohmann J, Molnár P, Deli J, Wesolowska O, Michalak K, Wang Q (2010) Reversal of multidrug resistance by natural substances from plants. Curr Top Med Chem 10(17):1757–1768
Morshed AKM, Helal SP, Hossain A, Tuli Basak Md, Sanower Hossain Md, Mehedi Hasan Md, Hasibuzzaman Al, Rahaman TI, Mia MAR, Shing P (2023) Baicalein as promising anticancer agent: a comprehensive analysis on molecular mechanisms and therapeutic perspectives. Cancers 15(7):2128
Movius EG, Phyillaier MM, Robbins J (1989) Phloretin Inhibits cellular uptake and nuclear receptor binding of triiodothyronine in human hep G2 hepatocarcinoma cells. Endocrinology. https://doi.org/10.1210/endo-124-4-1988
Nahar Metu CL, Sutihar SK, Sohel M, Zohora F, Hasan A, Miah MT, Rani Kar T, Hossain MA, Rahman MH (2023) Unraveling the signaling mechanism behind astrocytoma and possible therapeutics strategies: a comprehensive review. Cancer Reports. 6(10):e1889
Nair SV, Ziaullah Rupasinghe HV (2014) Fatty acid esters of phloridzin induce apoptosis of human liver cancer cells through altered gene expression. PLoS ONE 9(9):e107149
Nakhate KT, Badwaik H, Choudhary R, Sakure K, Agrawal YO, Sharma C, Ojha S, Goyal SN (2022) Therapeutic potential and pharmaceutical development of a multitargeted flavonoid phloretin. Nutrients 14(17):3638
Nam S, Lee SY, Cho HJ (2017) Phloretin-loaded fast dissolving nanofibers for the locoregional therapy of oral squamous cell carcinoma. J Colloid Interface Sci. https://doi.org/10.1016/j.jcis.2017.08.030
Nguyen H, Zhang S, Morris ME (2003) Effect of flavonoids on MRP1-mediated transport in panc-1 cells. J Pharm Sci 92(2):250–257. https://doi.org/10.1002/jps.10283
Nobili S, Lippi D, Witort E, Donnini M, Bausi L, Mini E, Capaccioli S (2009) Natural compounds for cancer treatment and prevention. Pharmacol Res. https://doi.org/10.1016/j.phrs.2009.01.017
Ogawa T, Sasaki A, Ono K, Ohshika S, Ishibashi Y, Yamada K (2021) Uptake of fluorescent D-and L-glucose analogues, 2-NBDG and 2-NBDLG, into human osteosarcoma U2OS cells in a phloretin-inhibitable manner. Hum Cell 34:634–643
Oliveira MR (2016) Phloretin-induced cytoprotective effects on mammalian cells: a mechanistic view and future directions. BioFactors 42(1):13–40
Park SY, Kim EJ, Shin HK, Kwon DY, Kim MS, Surh YJ, Park JHY (2007) Induction of apoptosis in HT-29 colon cancer cells by phloretin. J Med Food 10(4):581–586. https://doi.org/10.1089/jmf.2007.116
Patel BB, Alsanani A, Wondisford S, Johnson N, Banerjee D (2016) Stromal carcinoma associated fibroblasts promote drug resistance of human pancreatic cancer cells by modulation of ROS via CXCR4/CXCL12 signaling. Int J Transl Sci 2016(1):107–130
Patel BB, Ackerstaff E, Serganova IS, Kerrigan JE, Blasberg RG, Koutcher JA, Banerjee D (2017) Tumor stroma interaction is mediated by monocarboxylate metabolism. Exp Cell Res 352(1):20–33. https://doi.org/10.1016/j.yexcr.2017.01.013
Paul P, Biswas P, Dey D, Saikat ASM, Islam MA, Sohel Md, Hossain R et al (2021) Exhaustive plant profile of ‘dimocarpus longan lour’ with significant phytomedicinal properties: a literature based-review. Processes. https://doi.org/10.3390/pr9101803
Payne JN, Badwaik VD, Waghwani HK, Moolani HV, Tockstein S, Thompson DH, Dakshinamurthy R (2018) Development of dihydrochalcone-functionalized gold nanoparticles for augmented antineoplastic activity. Int J Nanomed. https://doi.org/10.2147/IJN.S143506
“Phloretin | C15H14O5 - PubChem.”.
“
“Phloretin CF | Topical Vitamin C | Vitamin C Gel | SkinCeuticals.”.
Picinelli A, Dapena E, Mangas JJ (1995) Polyphenolic pattern in apple tree leaves in relation to scab resistance a preliminary study. J Agric Food Chem. https://doi.org/10.1021/jf00056a057
Po, Ughachukwu, and Unekwe Pc. 2012. “Efflux Pump _ Mediated Resistance in Chemotherapy” 2 (2): 191–98. https://doi.org/10.4103/2141-9248.105671.
Pope ED, Kimbrough EO, Vemireddy LP, Surapaneni PK, Copland JA, Mody K (2019) Aberrant lipid metabolism as a therapeutic target in liver cancer. Expert Opin Ther Targets. https://doi.org/10.1080/14728222.2019.1615883
Prasanna S, Doerksen R (2008) Topological polar surface area: a useful descriptor in 2D-QSAR. Curr Med Chem 16(1):21–41. https://doi.org/10.2174/092986709787002817
Ranjanamala T, Vanmathiselvi K, Shreemaya R, Rajeswari SN, Sangeetha CC, Ghidan AY, Ghidan FY (2021) Virtual screening of anticancer efficacy of phloretin against apoptotic targets–An In Silico molecular docking study. J Phytol 13:152–160. https://doi.org/10.25081/JP.2021.V13.7268
Rawla P, Sunkara T, Gaduputi V (2019) epidemiology of pancreatic cancer: global trends, etiology and risk factors. World J Oncol 10(1):10–27. https://doi.org/10.14740/wjon1166
Rezk BM, Haenen GRMM, Van der Vijgh WJF, Bast A (2002) The antioxidant activity of phloretin: the disclosure of a new antioxidant pharmacophore in flavonoids. Biochem Biophys Res Commun. https://doi.org/10.1016/S0006-291X(02)00618-6
Rodriguez EB, Flavier ME, Rodriguez-Amaya DB, Amaya-Farfán J (2015) Phytochemicals and functional foods. Current situation and prospect for developing countries”. Segurança Alimentar e Nutricional. https://doi.org/10.20396/san.v13i1.1841
Roy S, Mondru AK, Chakraborty T, Das A, Dasgupta S (2022) Apple polyphenol phloretin complexed with ruthenium is capable of reprogramming the breast cancer microenvironment through modulation of PI3K/Akt/MTOR/VEGF pathways. Toxicol Appl Pharmacol. https://doi.org/10.1016/j.taap.2021.115822
Saraswati S, Alhaider A, Abdelgadir AM, Tanwer P, Korashy HM (2019) Phloretin attenuates STAT-3 activity and overcomes sorafenib resistance targeting SHP-1-mediated inhibition of STAT3 and Akt/VEGFR2 pathway in hepatocellular carcinoma. Cell Commun Signaling. https://doi.org/10.1186/s12964-019-0430-7
Schaefer S, Baum M, Eisenbrand G, Dietrich H, Will F, Janzowski C (2006) Polyphenolic apple juice extracts and their major constituents reduce oxidative damage in human colon cell lines. Mol Nutr Food Res. https://doi.org/10.1002/mnfr.200500136
Schiavano GF, De Santi M, Brandi G, Fanelli M, Bucchini A, Giamperi L, Giomaro G (2015) Inhibition of breast cancer cell proliferation and in vitro tumorigenesis by a new red apple cultivar. PLoS ONE. https://doi.org/10.1371/JOURNAL.PONE.0135840
Shang Ao, Liu H-Y, Min Luo Yu, Xia XY, Li H-Y, Ding-Tao Wu, Sun Q, Geng F, Gan R-Y (2022) Sweet Tea (Lithocarpus Polystachyus Rehd.) as a new natural source of bioactive dihydrochalcones with multiple health benefits. Crit Rev Food Sci Nutr 62(4):917–934
Shrihastini V, Muthuramalingam P, Adarshan S, Sujitha M, Chen JT, Shin H, Ramesh M (2021) Plant derived bioactive compounds, their anti-cancer effects and in silico approaches as an alternative target treatment strategy for breast cancer: an updated overview. Cancers. https://doi.org/10.3390/cancers13246222
Si W, Shen J, Zheng H, Fan W (2019) The role and mechanisms of action of MicroRNAs in cancer drug resistance. Clin Epigenetics. https://doi.org/10.1186/s13148-018-0587-8
Siegel RL, Miller KD, Wagle NS, Jemal A (2023) Cancer Statistics, 2023. CA Cancer J Clin. https://doi.org/10.3322/caac.21763
Slimestad R, Fossen T, Verheul MJ (2008) The flavonoids of tomatoes. J Agric Food Chem 56(7):2436–2441
So, Felicia V, Najla Guthrie, Ann F Chambers, Madeleine Moussa, Kenneth K Carroll, Felicia V So, Najla Guthrie, et al. 2009. “Inhibition of Human Breast Cancer Cell Proliferation and Delay of Mammary Tumorigenesis by Flavonoids and Citrus Juices Inhibition of Human Breast Cancer Cell Proliferation and Delay of Mammary Tumorigenesis by Flavonoids and Citrus Juices” 5581 (November 2017). https://doi.org/10.1080/01635589609514473.
Sohel M, Aktar S, Biswas P, Amin MA, Hossain MA, Ahmed N, Mim MI, Islam F, Mamun AA (2023) Exploring the anti-cancer potential of dietary phytochemicals for the patients with breast cancer: a comprehensive review. Cancer Med 12(13):14556–14583
Sohel Md, Partha Biswas Md, Al Amin Md, Hossain A, Sultana H, Dey D, Aktar S et al (2022a) Genistein, a potential phytochemical against breast cancer treatment-insight into the molecular mechanisms. Processes. https://doi.org/10.3390/pr10020415
Sohel Md, Islam MN, Hossain MA, Sultana T, Amit Dutta Md, Rahman S, Aktar S, Islam K, Mamun AA (2022b) Pharmacological Properties to Pharmacological Insight of Sesamin in Breast Cancer Treatment: A Literature-Based Review Study. International Journal of Breast Cancer. https://doi.org/10.1155/2022/2599689
Sohel M, Sultana H, Sultana T, Al Amin M, Aktar S, Ali MC, Rahim ZB, Hossain MA, Al Mamun A, Amin MN, Dash R (2022c) Chemotherapeutic potential of hesperetin for cancer treatment, with mechanistic insights: a comprehensive review. Heliyon. 8(1):e08815
Sohel Md, Sultana H, Sultana T, Mamun AA, Amin MN, Md Hossain Md, Ali SA, Sultana A, Rahim ZB (2022cd) Chemotherapeutic activities of dietary phytoestrogens against prostate cancer: from observational to clinical studies. Curr Pharm Des 28(19):1561–1580
Suhail S, Gupta S, Kumar V, Gupta OP (2016) Role of curcumin in oral cancer prevention. Fract Geomet Nonlinear Anal Med Biol. https://doi.org/10.15761/fgnamb.1000119
Sun YS, Zhao Z, Yang ZN, Fang Xu, Hang Jing Lu, Zhu ZY, Shi W, Jiang J, Yao PP, Zhu HP (2017) Risk factors and preventions of breast cancer. Int J Biol Sci. https://doi.org/10.7150/ijbs.21635
Szabo K, Mitrea L, Călinoiu LF, Teleky BE, Martău GA, Plamada D, Pascuta MS, Nemeş SA, Varvara RA, Vodnar DC (2022) Natural polyphenol recovery from apple-, cereal-, and tomato-processing by-products and related health-promoting properties. Molecules. https://doi.org/10.3390/molecules27227977
Szliszka E, Czuba ZP, Mazur B, Paradysz A, Krol W (2010) Chalcones and Dihydrochalcones Augment TRAIL-Mediated Apoptosis in Prostate Cancer Cells. Molecules 15(8):5336–5353
Tang J, Gong Y (2021) Synergistic effect of phloretin combined with radiotherapy on lung cancer. Int J Radiat Oncol Biol Phys. https://doi.org/10.1016/j.ijrobp.2021.07.809
Tuli HS, Rath P, Chauhan A, Ramniwas S, Vashishth K, Varol M, Jaswal VS, Haque S, Sak K (2022a) Phloretin, as a potent anticancer compound: from chemistry to cellular interactions. Molecules 27(24):8819
Tuli HS, Rath P, Chauhan A, Ramniwas S, Vashishth K, Varol M, Jaswal VS, Haque S, Sak K (2022b) Phloretin, as a potent anticancer compound: from chemistry to cellular interactions. Molecules. https://doi.org/10.3390/molecules27248819
Ulaszewska M, Vázquez-Manjarrez N, Garcia-Aloy M, Llorach R, Mattivi F, Dragsted LO, Praticò G, Manach C (2018) Food intake biomarkers for apple, pear, and stone fruit lars dragsted. Genes Nutr. https://doi.org/10.1186/s12263-018-0620-8
Wu C-H, Ho Y-S, Tsai C-Y, Wang Y-J, Tseng H, Wei P-L, Lee C-H, Liu R-S, Lin S-Y (2009a) In vitro and in vivo study of phloretin-induced apoptosis in human liver cancer cells involving inhibition of type ii glucose transporter. Int J Cancer 124(9):2210–2219
Wu CH, Ho YS, Tsai CY, Wang YJ, Tseng H, Wei PL, Lee CH, Liu RS, Lin SY (2009b) In vitro and in vivo study of phloretin-induced apoptosis in human liver cancer cells involving inhibition of type II glucose transporter. Int J Cancer. https://doi.org/10.1002/ijc.24189
Wu F, Zhou Y, Li L, Shen X, Chen G, Wang X, Liang X, Tan M, Huang Z (2020) Computational approaches in preclinical studies on drug discovery and development. Front Chem. https://doi.org/10.3389/fchem.2020.00726
Wu KH, Ho CT, Chen ZF, Chen LC, Whang-Peng J, Lin TN, Ho YS (2018) The apple polyphenol phloretin inhibits breast cancer cell migration and proliferation via inhibition of signals by type 2 glucose transporter. J Food Drug Anal 26(1):221–231. https://doi.org/10.1016/j.jfda.2017.03.009
Xintaropoulou C, Ward C, Wise A, Marston H, Turnbull A, Langdon SP (2015) A comparative analysis of inhibitors of the glycolysis pathway in breast and ovarian cancer cell line models. Oncotarget 6(28):25677
Xue X, Liang XJ (2012) Overcoming drug efflux-based multidrug resistance in cancer with nanotechnology. Chin J Cancer. https://doi.org/10.5732/cjc.011.10326
Yang C-H, Yen-Chuan Ou, Lin C-C, Lin Y-S, Tung M-C, Chia-Cheng Yu, Lin J-T, Wen C-Y (2022) Phloretin in benign prostate hyperplasia and prostate cancer: a contemporary systematic review. Life 12(7):1029
Yang G, Yin X, Ma D, Su Z (2020) Anticancer Activity of Phloretin against the Human Oral Cancer Cells Is Due to G0/G1 Cell Cycle Arrest and ROS Mediated Cell Death. J BUON 25:344–349
Yang KC, Tsai CY, Wang YJ, Wei PL, Lee CH, Chen JH, Chih Hsiung Wu, Ho YS (2009) Apple polyphenol phloretin potentiates the anticancer actions of paclitaxel through induction of apoptosis in human hep G2 cells. Mol Carcinog. https://doi.org/10.1002/mc.20480
You Q, Xu JP, Zhu ZX, Hu Z, Cai QP (2020) Phloretin flavonoid exhibits selective antiproliferative activity in doxorubicin-resistant gastric cancer cells by inducing autophagy, inhibiting cell migration and invasion, cell cycle arrest and targeting ERK1/2 MAP pathway. J BUON 25:308–313
Zhang S, Morris ME (2003) Effects of the flavonoids biochanin a, morin, phloretin, and silymarin on p-glycoprotein-mediated transport. J Pharmacol Exp Ther. https://doi.org/10.1124/jpet.102.044412
Zhao W, Bai B, Hong Z, Zhang X, Zhou B (2020) Berbamine (BBM), a natural STAT3 inhibitor, synergistically enhances the antigrowth and proapoptotic effects of sorafenib on hepatocellular carcinoma cells. ACS Omega. https://doi.org/10.1021/acsomega.0c03527
Zhao Y, Fan Y, Wang M, Wang J, Cheng J, Zou J, Zhang X, Shi Y, Guo D (2020) Studies on pharmacokinetic properties and absorption mechanism of phloretin: in vivo and in vitro. Biomed Pharmacother 132:110809
Zhou Mo, Zheng J, Bi J, Xinye Wu, Lyu J, Gao K (2018) Synergistic inhibition of colon cancer cell growth by a combination of atorvastatin and Phloretin. Oncol Lett 15(2):1985–1992. https://doi.org/10.3892/ol.2017.7480
Zhu S-P, Liu G, Xiao-Ting Wu, Chen F-X, Liu J-Q, Zhou Z-H, Zhang J-F, Fei S-J (2013a) The effect of Phloretin on human Γδ T cells killing colon cancer SW-1116 cells. Int Immunopharmacol 15(1):6–14
Zhu SP, Liu G, Xiao Ting Wu, Chen FX, Liu JQ, Zhou ZH, Zhang JF, Fei SJ (2013b) The effect of phloretin on human Γδ t cells killing colon cancer SW-1116 cells. Int Immunopharmacol 15(1):6–14. https://doi.org/10.1016/j.intimp.2012.11.001
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sohel, M., Nity, N.U., Sarker, M.R. et al. Exploring the chemotherapeutic potential and therapeutic insight of phloretin against human malignancies: a systematic review. Phytochem Rev (2024). https://doi.org/10.1007/s11101-024-09938-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11101-024-09938-8