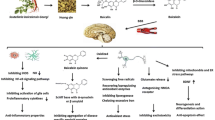
Abstract
Flavonoids have a longstanding tradition of being employed in traditional medicine to improve human health. Crocin has been suggested as a potential candidate for addressing a range of ailments, including neurodegenerative diseases (NDs). In the modern era, diverse pharmacological activities of crocin have been explored due to its bioactive constituents like kaempferol, picrocrocin, safranal, and crocetin. It demonstrates pharmacological and therapeutic efficacy against various diseases such as cancer, kidney diseases, NDs, and measles, attributable to several mechanisms. Various studies evidence that it exerts a neuroprotective effect in conditions like Alzheimer’s disease, Parkinson's disease, Huntington's disease, Amyotrophic lateral sclerosis, and others, by modulating relevant parameters. Despite harboring numerous pharmacological benefits, it exhibits low bioavailability due to poor solubility and limited ability to cross the blood–brain barrier (BBB). To address this issue, diverse approaches involving nanoformulations have been developed to enhance their bioavailability, aqueous solubility, and BBB permeability. Various types of studies have been conducted on crocin-loaded nanoparticles against NDs, which are discussed in this paper.




Similar content being viewed by others
References
Abeer A, Ahmed A (2019) Possible neuroprotective effects of crocin against motor and neurochemical changes in rotenone induced animal model of Parkinson’s disease. Med J Cairo Univ 87:2839–2847. https://doi.org/10.21608/mjcu.2019.59318
Abu-Izneid T, Rauf A, Khalil AA et al (2022) Nutritional and health beneficial properties of saffron (Crocus sativus L): a comprehensive review. Crit Rev Food Sci Nutr 62:2683–2706. https://doi.org/10.1080/10408398.2020.1857682
Al Badri YN, Chaw CS, Elkordy AA (2023) Insights into asymmetric liposomes as a potential intervention for drug delivery including pulmonary nanotherapeutics. Pharmaceutics. https://doi.org/10.3390/pharmaceutics15010294
Alexander GE (2022) Biology of Parkinson’s disease: pathogenesis and pathophysiology of a multisystem neurodegenerative disorder. Dialogues Clin Neurosci 6:259–280. https://doi.org/10.31887/DCNS.2004.6.3/GALEXANDER
Ali F (2023) Role of saffron and chamomile in the management of Parkinson’s disease (SAFCHEMRxPar). In: Clinicaltrial.gov.in https://www.clinicaltrials.gov/study/NCT05696665 Accessed date 19 Oct 2023
Antonio I, Inarejos M, Prodanov M, et al (2017) Saffron extract and its use for the prevention of mood disorders related to depression. US10933110B2
Asadi F, Jamshidi AH, Khodagholi F et al (2015) Reversal effects of crocin on amyloid β-induced memory deficit: modification of autophagy or apoptosis markers. Pharmacol Biochem Behav 139:47–58. https://doi.org/10.1016/J.PBB.2015.10.011
Awad R, Avital A, Sosnik A (2023) Polymeric nanocarriers for nose-to-brain drug delivery in neurodegenerative diseases and neurodevelopmental disorders. Acta Pharm Sin B 13:1866–1886. https://doi.org/10.1016/j.apsb.2022.07.003
Azarian F, Farsi S, Hosseini SA, Azarbayjani MA (2019) The effect of endurance training and crocin consumption on anxiety-like behaviors and aerobic power in rats with Alzheimer’s. Iran J Psychiatry Behav Sci. https://doi.org/10.5812/ijpbs.89011
Baluchnejadmojarad T (2019) Safranal, an active ingredient of saffron, attenuates cognitive deficits in amyloid β -induced rat model of Alzheimer’s disease: underlying mechanisms. Metab Brain Dis. https://doi.org/10.1007/s11011-019-00481-6
Bandiwadekar A, Jose J, Khayatkashani M et al (2022) Emerging novel approaches for the enhanced delivery of natural products for the management of neurodegenerative diseases. J Mol Neurosci 72:653–676. https://doi.org/10.1007/s12031-021-01922-7
Batarseh YS, Bharate SS, Kumar V et al (2017) Crocus sativus extract tightens the blood-brain barrier, reduces amyloid β load and related toxicity in 5XFAD mice. ACS Chem Neurosci 8:1756–1766. https://doi.org/10.1021/acschemneuro.7b00101
Behravanfar N, Abnous K, Razavi BM, Hosseinzadeh H (2017) Effects of crocin on spatial memory impairment induced by hyoscine and its effects on bdnf, creb, and p-creb protein and mrna levels in rat hippocampus. Jundishapur J Nat Pharm Prod. https://doi.org/10.5812/jjnpp.64315
Bhandari PR (2015) Crocus sativus L. (saffron) for cancer chemoprevention: a mini review. J Tradit Complement Med 5:81–87. https://doi.org/10.1016/J.JTCME.2014.10.009
Butnariu M, Quispe C, Herrera-Bravo J et al (2022) The pharmacological activities of Crocus sativus L.: a review based on the mechanisms and therapeutic opportunities of its phytoconstituents. Oxid Med Cell Longev. https://doi.org/10.1155/2022/8214821
Cacciatore I, Ciulla M, Fornasari E et al (2016) Solid lipid nanoparticles as a drug delivery system for the treatment of neurodegenerative diseases. Expert Opin Drug Deliv 13:1121–1131. https://doi.org/10.1080/17425247.2016.1178237
Cajanus A, Solje E, Koikkalainen J et al (2019) The association between distinct frontal brain volumes and behavioral symptoms in mild cognitive impairment, Alzheimer’s disease, and frontotemporal dementia. Front Neurol 10:1059. https://doi.org/10.3389/FNEUR.2019.01059/BIBTEX
Cano A, Sánchez-López E, Ettcheto M et al (2020) Current advances in the development of novel polymeric nanoparticles for the treatment of neurodegenerative diseases. Nanomedicine 15:1239–1261. https://doi.org/10.2217/nnm-2019-0443
Cardone L, Castronuovo D, Perniola M et al (2020) Saffron (Crocus sativus L.), the king of spices: an overview. Sci Hortic (amsterdam) 272:109560. https://doi.org/10.1016/J.SCIENTA.2020.109560
Cassidy L, Fernandez F, Johnson JB et al (2020) Oxidative stress in alzheimer’s disease: a review on emergent natural polyphenolic therapeutics. Complement Ther Med 49:102294. https://doi.org/10.1016/J.CTIM.2019.102294
Castillo-López IY, Govea-Camacho LH, Rodríguez-Torres IA et al (2020) Olfactory dysfunction in a Mexican population outside of COVID-19 pandemic: prevalence and associated factors (the Olfamex study). Curr Allergy Asthma Rep 20:1–12. https://doi.org/10.1007/S11882-020-00975-9/TABLES/3
Chalatsa I, Arvanitis DA, Koulakiotis NS et al (2019) The Crocus sativus compounds trans-crocin 4 and trans-crocetin modulate the amyloidogenic pathway and tau misprocessing in Alzheimer disease neuronal cell culture models. Front Neurosci 13:1–15. https://doi.org/10.3389/fnins.2019.00249
Chen Z, Li G, Liu J (2020) Autonomic dysfunction in Parkinson’s disease: implications for pathophysiology, diagnosis, and treatment. Neurobiol Dis 134:104700. https://doi.org/10.1016/J.NBD.2019.104700
Clark IA, Vissel B (2021) Broader insights into understanding tumor necrosis factor and neurodegenerative disease pathogenesis infer new therapeutic approaches. J Alzheimer’s Dis 79:931–948. https://doi.org/10.3233/JAD-201186
Clay E, Zhou J, Yi Z-M et al (2019) Economic burden for Alzheimer’s disease in China from 2010 to 2050: a modelling study. J Mark Access Health Policy 7:1667195. https://doi.org/10.1080/20016689.2019.1667195
Daneshvar A, Jouzdani AF, Firozian F, Asl SS (2022) Neuroprotective effects of crocin and crocin-loaded niosomes against the paraquat-induced oxidative brain damage in rats. Open Life Sci. https://doi.org/10.1515/biol-2022-0468
Desai HR, Sgunde UA (2022) Hybrid nanoparticles to cross the blood-brain barrier. J Nanopart Res. https://doi.org/10.1007/s11051-021-05175-8
Dolatshahi M, Ranjbar Hameghavandi MH, Sabahi M, Rostamkhani S (2021) Nuclear factor-kappa B (NF-κB) in pathophysiology of Parkinson disease: diverse patterns and mechanisms contributing to neurodegeneration. Eur J Neurosci 54:4101–4123. https://doi.org/10.1111/EJN.15242
Duan Y, Kelley N, He Y (2020) Role of the NLRP3 inflammasome in neurodegenerative diseases and therapeutic implications. ncbi.nlm.nih.gov
Duchen MR (2000) Mitochondria and Ca2+in cell physiology and pathophysiology. Cell Calcium 28:339–348. https://doi.org/10.1054/CECA.2000.0170
Dugger BN, Dickson DW (2017) Pathology of neurodegenerative diseases. Cold Spring Harb Perspect Biol. https://doi.org/10.1101/CSHPERSPECT.A028035
Ebrahimzadeh A, Moghadam SY, Rahimi H et al (2019) Crocin acts as a neuroprotective mediator against methylphenidate-induced neurobehavioral and neurochemical sequelae: possible role of the CREB-BDNF signaling pathway. Acta Neurobiol Exp (wars) 79:352–366. https://doi.org/10.21307/ane-2019-033
Fifel K, Videnovic A (2020) Circadian alterations in patients with neurodegenerative diseases: neuropathological basis of underlying network mechanisms. Neurobiol Dis 144:105029. https://doi.org/10.1016/J.NBD.2020.105029
Fourcand F (2022) Neurologist formulated nocturnal nootropic founded on a novel theory of brain aging US20220233546A1. 48
Garcia AMI (2023) Japan patent.pdf https://www.jpo.go.jp/e/ Accessed date 19 Oct 2023
García-Sanz P, Aerts JMFG, Moratalla R (2021) The role of cholesterol in α-synuclein and lewy body pathology in GBA1 Parkinson’s disease. Mov Disord 36:1070–1085. https://doi.org/10.1002/MDS.28396
Garg V, Gupta R, Kapoor B et al (2016) Application of self-emulsifying delivery systems for effective delivery of nutraceuticals. Emulsions. Elsevier, Amsterdam, pp 479–518
Gauro R, Nandave M, Kumar V (2021) Advances in dendrimer-mediated targeted drug delivery to the brain. J Nanopart Res. https://doi.org/10.1007/s11051-021-05175-8
Guan PP, Cao LL, Wang P (2021) Elevating the levels of calcium ions exacerbate Alzheimer’s disease via inducing the production and aggregation of β-amyloid protein and phosphorylated tau. Int J Mol Sci 22:5900. https://doi.org/10.3390/IJMS22115900
Guillama Barroso G, Narayan M, Alvarado M et al (2020) Nanocarriers as potential drug delivery candidates for overcoming the blood-brain barrier: challenges and possibilities. ACS Omega 5:12583–12595. https://doi.org/10.1021/acsomega.0c01592
Hadipour M, Meftahi GH, Afarinesh MR et al (2020) Crocin attenuates the granular cells damages on the dentate gyrus and pyramidal neurons in the CA3 regions of the hippocampus and frontal cortex in the rat model of Alzheimer’s disease. J Chem Neuroanat. https://doi.org/10.1016/j.jchemneu.2020.101837
Haeri P, Mohammadipour A, Heidari Z, Ebrahimzadeh-bideskan A (2019) Neuroprotective effect of crocin on substantia nigra in MPTP-induced Parkinson’s disease model of mice. Anat Sci Int 94:119–127. https://doi.org/10.1007/s12565-018-0457-7
Haimhoffer Á, Rusznyák Á, Réti-Nagy K et al (2019) Cyclodextrins in drug delivery systems and their effects on biological barriers. Sci Pharm. https://doi.org/10.3390/scipharm87040033
Hajal C, Le Roi B, Kamm RD, Maoz BM (2021) Biology and models of the blood-brain barrier. Annu Rev Biomed Eng 23:359–384. https://doi.org/10.1146/annurev-bioeng-082120-042814
Heidari S, Mehri S, Hosseinzadeh H (2017) Memory enhancement and protective effects of crocin against D-galactose aging model in the hippocampus of wistar rats. Iran J Basic Med Sci 20:1250–1259. https://doi.org/10.22038/IJBMS.2017.9541
Hernandez C, Shukla S (2022) Liposome based drug delivery as a potential treatment option for Alzheimer’s disease. Neural Regen Res 17:1190–1198. https://doi.org/10.4103/1673-5374.327328
Hersh AM, Alomari S, Tyler BM (2022) Crossing the blood-brain barrier: advances in nanoparticle technology for drug delivery in neuro-oncology. Int J Mol Sci 23(8):4153. https://doi.org/10.3390/ijms23084153
Hill AE, Wade-Martins R, Burnet PWJ (2021) What is our understanding of the influence of gut microbiota on the pathophysiology of Parkinson’s disease? Front Neurosci. https://doi.org/10.3389/FNINS.2021.708587/FULL
Inoue E, Shimizu Y, Masui R et al (2018) Effects of saffron and its constituents, crocin-1, crocin-2, and crocetin on α-synuclein fibrils. J Nat Med 72:274–279. https://doi.org/10.1007/s11418-017-1150-1
Inoue E, Suzuki T, Shimizu Y et al (2021) Saffron ameliorated motor symptoms, short life span and retinal degeneration in Parkinson’s disease fly models. Gene 799:145811. https://doi.org/10.1016/j.gene.2021.145811
Jacob S, Nair AB (2018) Cyclodextrin complexes: perspective from drug delivery and formulation. Drug Dev Res 79:201–217. https://doi.org/10.1002/ddr.21452
Jain G, Pathan S, Zaidi S et al (2012) Mucoadhesive nanoemulsion of safranal for the treatment of Epilepsy. Planta Med 78:P_112. https://doi.org/10.1055/S-0032-1307620
Jurcau A (2021) Insights into the pathogenesis of neurodegenerative diseases: focus on mitochondrial dysfunction and oxidative stress. Int J Mol Sci 22:11847. https://doi.org/10.3390/IJMS22211184
Karami Z, Saghatchi Zanjani MR, Hamidi M (2019) Nanoemulsions in CNS drug delivery: recent developments, impacts and challenges. Drug Discov Today 24:1104–1115. https://doi.org/10.1016/j.drudis.2019.03.021
Karri V, Schuhmacher M, Kumar V (2020) A systems toxicology approach to compare the heavy metal mixtures (Pb, As, MeHg) impact in neurodegenerative diseases. Food Chem Toxicol 139:111257. https://doi.org/10.1016/J.FCT.2020.111257
Kaur R, Mehan S, Singh S (2019) Understanding multifactorial architecture of Parkinson’s disease: pathophysiology to management. Neurol Sci 40:13–23. https://doi.org/10.1007/s10072-018-3585-x
Kaur J, Gulati M, Kapoor B et al (2022) Advances in designing of polymeric micelles for biomedical application in brain related diseases. Chem Biol Interact 361:109960. https://doi.org/10.1016/j.cbi.2022.109960
Kaur A, Singh N, Kaur H et al (2023) Neurodegenerative disease and brain delivery of therapeutics: bridging the gap using dendrimers. J Drug Deliv Sci Technol. https://doi.org/10.1016/j.jddst.2023.104868
Khairnar SV, Pagare P, Thakre A et al (2022) Review on the scale-up methods for the preparation of solid lipid nanoparticles. Pharmceutics 14(9):1886
Khan MI, Jeong ES, Khan MZ et al (2023) Stem cells-derived exosomes alleviate neurodegeneration and Alzheimer’s pathogenesis by ameliorating neuroinflamation, and regulating the associated molecular pathways. Sci Reports 13:1–11. https://doi.org/10.1038/s41598-023-42485-4
Klajnert B, Cladera J, Bryszewska M (2006) Molecular interactions of dendrimers with amyloid peptides: pH dependence. Biomacromol 7:2186–2191. https://doi.org/10.1021/bm060229s
Koulakiotis NS, Purhonen P, Gikas E et al (2020) Crocus-derived compounds alter the aggregation pathway of Alzheimer’s disease—associated beta amyloid protein. Sci Reports 101(10):1–10. https://doi.org/10.1038/s41598-020-74770-x
Kumar R, Kumar R, Khurana N et al (2020) Enhanced oral bioavailability and neuroprotective effect of fisetin through its SNEDDS against rotenone-induced Parkinson’s disease rat model. Food Chem Toxicol 144:111590. https://doi.org/10.1016/J.FCT.2020.111590
Kumari Y, Kaur G, Kumar R et al (2019) Gold nanoparticles: new routes across old boundaries. Adv Colloid Interface Sci 274:102037. https://doi.org/10.1016/J.CIS.2019.102037
Lauretti E, Dincer O, Praticò D (2020) Glycogen synthase kinase-3 signaling in Alzheimer’s disease. Biochim Biophys Acta - Mol Cell Res 1867:118664. https://doi.org/10.1016/J.BBAMCR.2020.118664
Li GH, Li P, Lu L et al (2020) The outcome and burden of Chinese patients with neurodegenerative diseases: a 10-year clinical feature study. Int J Clin Pract 74:e13534. https://doi.org/10.1111/IJCP.13534
Li X, Naeem A, Xiao S et al (2022) Safety challenges and application strategies for the use of dendrimers in medicine. Pharmceutics. https://doi.org/10.3390/pharmaceutics14061292
Lin L, Liu G, Yang L (2019) Crocin improves cognitive behavior in rats with Alzheimer’s disease by regulating endoplasmic reticulum stress and apoptosis. Biomed Res Int. https://doi.org/10.1155/2019/9454913
Madkhali OA (2022) Perspectives and prospective on solid lipid nanoparticles as drug delivery systems. Molecules 27:1543. https://doi.org/10.1557/S0883769400056050
Menaceur C, Gosselet F, Fenart L, Saint-Pol J (2022) The blood–brain barrier, an evolving concept based on technological advances and cell–cell communications. Cells. https://doi.org/10.3390/cells11010133
Milowska K, Malachowska M, Gabryelak T (2011) PAMAM G4 dendrimers affect the aggregation of α-synuclein. Int J Biol Macromol 48:742–746. https://doi.org/10.1016/j.ijbiomac.2011.02.021
Milowska K, Szwed A, Mutrynowska M et al (2015) Carbosilane dendrimers inhibit α-synuclein fibrillation and prevent cells from rotenone-induced damage. Int J Pharm 484:268–275. https://doi.org/10.1016/j.ijpharm.2015.02.066
Moradi SZ, Momtaz S, Bayrami Z et al (2020) Nanoformulations of herbal extracts in treatment of neurodegenerative disorders. Front Bioeng Biotechnol 8:1–20. https://doi.org/10.3389/fbioe.2020.00238
Negarandeh Z, Salamat KM, Hosseini SA, Etemad Z (2019) The effect of endurance training with crocin consumption on IGF-1 and glycogen expression in rat hippocampus tissue of trimethyltin-treated model of Alzheimer’s disease. Asian J Sports Med 10(3):1–7. https://doi.org/10.5812/asjsm.1525.92246
Nejati K, Dadashpour M, Gharibi T et al (2022) Biomedical applications of functionalized gold nanoparticles: a review. J Clust Sci 33:1–16. https://doi.org/10.1007/s10876-020-01955-9
Nirale P, Paul A, Yadav KS (2020) Nanoemulsions for targeting the neurodegenerative diseases: Alzheimer’s, Parkinson’s and Prion’s. Life Sci 245:117394. https://doi.org/10.1016/j.lfs.2020.117394
Nouri H, Ghahghaei A, Lagzian M (2021) Impact of synthesized AuNPs from crocin against aggregation and conformational change in α-lactalbumin. Int J Pept Res Ther 27:2275–2289. https://doi.org/10.1007/s10989-021-10252-6
Pct W, Dubreuil S, Moras B, Bornerie M (2023) Composition based on crocus sativus and cannabis sativa WO2023012258A1. 48
Pct W (2023) Multicomponent composition comprising epigallocatechin gallate and saffron dry extract, and its use in the prevention and treatment of Parkinson’s disease. 48
Pérez-Carrión MD, Posadas I (2023) Dendrimers in neurodegenerative diseases. Processes 11:319. https://doi.org/10.3390/pr11020319
Perteghella S, Rassu G, Gavini E et al (2021) Crocetin as new cross-linker for bioactive sericin nanoparticles. Pharmceutics 13(5):680. https://doi.org/10.3390/pharmaceutics13050680
Pinto M, Silva V, Barreiro S et al (2022) Brain drug delivery and neurodegenerative diseases: polymeric PLGA-based nanoparticles as a forefront platform. Ageing Res Rev 79:101658. https://doi.org/10.1016/j.arr.2022.101658
Puglia C, Santonocito D, Musumeci T et al (2018) Nanotechnological approach to increase the antioxidant and cytotoxic efficacy of crocin and crocetin authors. Planta Med 85(3):258–265. https://doi.org/10.1055/a-0732-5757
Rafieipour F, Hadipour E, Emami SA et al (2019) Safranal protects against beta-amyloid peptide-induced cell toxicity in PC12 cells via MAPK and PI3 K pathways. Metab Brain Dis 34:165–172. https://doi.org/10.1007/s11011-018-0329-9
Rajaei Z, Hosseini M, Alaei H (2016) Effects of crocin on brain oxidative damage and aversive memory in a 6-OHDA model of Parkinson’s disease. Arq Neuropsiquiatr 74:723–729. https://doi.org/10.1590/0004-282X20160131
Rao SV, Muralidhara YSC, Rajini PS (2016) Evidence of neuroprotective effects of saffron and crocin in a Drosophila model of parkinsonism. Neurotoxicology 52:230–242. https://doi.org/10.1016/j.neuro.2015.12.010
Rashedinia M, Lari P, Abnous K, Hosseinzadeh H (2015) Protective effect of crocin on acrolein-induced tau phosphorylation in the rat brain. Acta Neurobiol Exp (wars) 75:208–219
Rather MA, Khan A, Alshahrani S et al (2021) Inflammation and Alzheimer’s disease: mechanisms and therapeutic implications by natural products. Mediators Inflamm. https://doi.org/10.1155/2021/9982954
Reddy S, Tatiparti K, Sau S, Iyer AK (2021) Recent advances in nano delivery systems for blood-brain barrier (BBB) penetration and targeting of brain tumors. Drug Discov Today 26:1944–1952. https://doi.org/10.1016/j.drudis.2021.04.008
Rezaei O, Nateghinia S, Estiar MA et al (2021) Assessment of the role of non-coding RNAs in the pathophysiology of Parkinson’s disease. Eur J Pharmacol 896:173914. https://doi.org/10.1016/J.EJPHAR.2021.173914
Riya S, Dua JS, Dn P (2022) An overview on Niosomes: Novel Pharmaceutical drug delivery system. J Drug Deliv Therapeutics 12:171–177. https://doi.org/10.22270/jddt.v12i2-s.5264
Sabjan KB, Munawar SM, Rajendiran D et al (2019) Nanoemulsion as oral drug delivery—a review. Curr Drug Res Rev 12:4–15. https://doi.org/10.2174/2589977511666191024173508
Sadoughi D (2019) The effect of crocin on apoptotic, inflammatory, BDNF, Pt, and A b 40 indicators and neuronal density of CA1, CA2, and CA3 regions of hippocampus in the model of Alzheimer suffering rats induced with trimethyltin chloride. Comp Clin Pathol 28:1403–1413. https://link.springer.com/article/10.1007/s00580-019-02981-4
Saeedi M, Rashidy-Pour A (2021) Association between chronic stress and Alzheimer’s disease: therapeutic effects of saffron. Biomed Pharmacother 133:110995. https://doi.org/10.1016/J.BIOPHA.2020.110995
Salama RM (2019) Neuroprotective effect of crocin against rotenone-induced Parkinson’s disease in rats: interplay between PI3K/Akt/mTOR signaling pathway and enhanced expression of miRNA-7 and miRNA-221. Neuropharmacology. https://doi.org/10.1016/j.neuropharm.2019.107900
Samarghandian S, Azimi-Nezhad M, Samini F (2015) Preventive effect of safranal against oxidative damage in aged male rat brain. Exp Anim 64:65–71
Sato Y, Minami K, Hirato T et al (2022) Drug delivery for neuronopathic lysosomal storage diseases: evolving roles of the blood brain barrier and cerebrospinal fluid. Metab Brain Dis 37:1745–1756. https://doi.org/10.1007/s11011-021-00893-3
Save SS, Rachineni K, Hosur RV, Choudhary S (2019) International journal of biological macromolecules natural compound safranal driven inhibition and dis-aggregation of α -synuclein fi brils. Int J Biol Macromol 141:585–595. https://doi.org/10.1016/j.ijbiomac.2019.09.053
Shaheen G, Akram M, Jabeen F et al (2019) Therapeutic potential of medicinal plants for the management of urinary tract infection: a systematic review. Clin Exp Pharmacol Physiol 46:613–624. https://doi.org/10.1111/1440-1681.13092
Shahidani S, Rajaei Z (2019) Pretreatment with crocin along with treadmill exercise ameliorates motor and memory deficits in hemiparkinsonian rats by anti-inflammatory and antioxidant mechanisms. Metab Brain Dis 13(5):680. https://doi.org/10.3390/pharmaceutics13050680
Siddique S, Chow JCL (2020) Applied sciences gold nanoparticles for drug delivery and cancer therapy. Appl Sci. https://doi.org/10.3390/app10113824
Simon DK, Tanner CM, Brundin P (2020) Parkinson disease epidemiology, pathology, genetics, and pathophysiology. Clin Geriatr Med 36:1–12. https://doi.org/10.1016/j.cger.2019.08.002
Song J, Lu C, Leszek J, Zhang J (2021) Design and development of nanomaterial-based drug carriers to overcome the blood–brain barrier by using different transport mechanisms. Int J Mol Sci. https://doi.org/10.3390/ijms221810118
Spuch C, Navarro C (2011) Liposomes for targeted delivery of active agents against neurodegenerative diseases (Alzheimer’s disease and Parkinson’s disease). J Drug Deliv 2011:1–12. https://doi.org/10.1155/2011/469679
(2023) Statistics. In: park. Found. https://www.hindustantimes.com/world-news/world-parkinson-s-day-2023-history-significance-facts-101681180342101.html#:~:text=The%20disease%20occurs%20with%20advancing,We're%20now%20on%20WhatsApp. Accessed date 21 Oct 2023
Tan MS, Yu JT, Jiang T et al (2013) The NLRP3 inflammasome in Alzheimer’s disease. Mol Neurobiol 48:875–882. https://doi.org/10.1007/S12035-013-8475-X/FIGURES/3
Tang J, Lu L, Wang Q et al (2020) Crocin reverses depression-like behavior in parkinson disease mice via VTA-mPFC pathway. Mol Neurobiol 57(7):3158–3170. https://doi.org/10.1007/s12035-020-01941-2
Tashima T (2020) Smart strategies for therapeutic agent delivery into brain across the blood–brain barrier using receptor-mediated transcytosis. Chem Pharm Bull 68:316–325. https://doi.org/10.1248/cpb.c19-00854
Tobore TO (2019) (2019) On the central role of mitochondria dysfunction and oxidative stress in Alzheimer’s disease. Neurol Sci 408(40):1527–1540. https://doi.org/10.1007/S10072-019-03863-X
Vishwas S, Singh SK, Gulati M et al (2022) Harnessing the therapeutic potential of fisetin and its nanoparticles: journey so far and road ahead. Chem Biol Interact 356:109869. https://doi.org/10.1016/j.cbi.2022.109869
Voet S, Srinivasan S, Lamkanfi M, van Loo G (2019) Inflammasomes in neuroinflammatory and neurodegenerative diseases. EMBO Mol Med 11:e10248. https://doi.org/10.15252/EMMM.201810248
Wang C, Cai X, Hu W et al (2019) Investigation of the neuroprotective effects of crocin via antioxidant activities in HT22 cells and in mice with Alzheimer’s disease. Int J Mol Med 43:956–966
Wong KH, Xie Y, Huang X et al (2020) Delivering crocetin across the blood-brain barrier by using γ-cyclodextrin to treat Alzheimer’s disease. Sci Rep 10:1–12. https://doi.org/10.1038/s41598-020-60293-y
Yang Meichun Z (2023) Traditional Chinese medicine preparation for preventing and treating senile dementia and preparation method thereof. CN116637164A
Yoshino Y, Ishisaka M, Umigai N et al (2014) Crocetin prevents amyloid <i>β</i><sub>1–42</sub>-induced cell death in murine hippocampal cells. Pharmacol Pharm 05:37–42. https://doi.org/10.4236/pp.2014.51007
Zhang J, Wang Y, Dong X, Liu J (2018) Crocetin attenuates inflammation and amyloid- β accumulation in APPsw transgenic mice. Immun Ageing 1–8. https://doi.org/10.1186/s12979-018-0132-9
Zhong QQ, Zhu F (2022) Trends in prevalence cases and disability-adjusted life-years of Parkinson’s disease: findings from the global burden of disease study 2019. Neuroepidemiology 56:261–270. https://doi.org/10.1159/000524208
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kaur, H., Bashir, B., Kaur, A. et al. Unravelling role of crocin for the treatment of Alzheimer’s and Parkinson’s disease: sojourn from food to nanomedicine. Phytochem Rev (2024). https://doi.org/10.1007/s11101-024-09936-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11101-024-09936-w