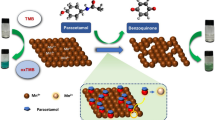
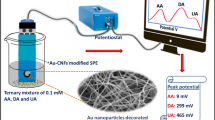
A carbon paste electrode (CPE) modified with multi-walled carbon nanotubes (MWCNTs) and natural deep eutectic solvent (NADES) was prepared and applied for the simultaneous voltammetric determination of acetaminophen (ACP) and thiosalicylic acid (TSA) in real samples. Electrochemical impedance spectroscopy (EIS) was applied for investigation of electron transfer rate of [Fe(CN)6]3-/4- as a redox couple probe on the MWCNTs/NADES/CPE surface. The modified electrode preserved and combined the properties of individual modifiers synergistically. Significant enhancement in the peak current responses of ACP and TSA were observed at the modified electrode compared to the bare electrode. Under the optimal conditions, a linear dynamic range of 5–2900 μM for ACP and 5–2250 μM for TSA was obtained. The limit of detection (LOD) for ACP and TSA were 4.71 μM and 4.35 μM, respectively. Finally, this method was successfully employed for the determination of ACP and TSA in real samples.







Similar content being viewed by others
References
A. P. Abbott, D. Boothby, G. Capper, et al., J. Am. Chem. Soc., 126(29), 9142 – 9147 (2004).
D. Carriazo, M. C. Serrano, M. C. Gutierrez, et al., Chem. Soc. Rev., 41, 4996 – 5014 (2012).
Y. H. Choi, J. Van Spronsen, Y. Dai, et al., Plant Physiol., 156, 1701 – 1705 (2011).
A. Paiva, R. Craveiro, I. Aroso, et al., ACS Sustain. Chem. Eng., 2(5), 1063 – 1071 (2014).
E. L. Smith, A. P. Abbott, and K. S. Ryder, Chem. Rev., 114(21), 11060 – 11082 (2014).
C. G. Gonzalez, N. R. Mustafa, E. G. Wilson, et al., Flavour Fragr. J., 33(1), 91 – 96 (2018).
L. G. Shaidarova, I. A. Chelnokova, G. F. Makhmutova, et al., Pharm. Chem. J., 48(8), 537 – 542 (2014).
M. Hadi and H. Mostaanzadeh, Russ. J. Electrochem., 54(12), 1045 – 1052 (2018).
B. D. Clayton and Y. N. Stock, Basic Pharmacology for Nurses, Mosby Inc., Harcourt Health Sciences Company, St. Louis (2001).
G. Burgot, F. Auffret, and J. L. Burgot, Anal. Chim. Acta, 343(1 – 2), 125 – 128 (1997).
S. A. Mohamed and T. Elsaman, Pharm. Chem. J., 54(12), 1306 – 1310 (2021).
J. A. M. Pulgarnn, and L. F. G. Bermejo, Anal. Chim. Act, 333(1 – 2) 59 – 69 (1996).
D. Easwaramoorthy, Y. C. Yu, and H. J. Huang, Anal. Chim. Acta, 439(1), 95 – 100 (2001).
S. Ravisankar, M. Vasudevan, and M. Gandhimathi, Talanta, 46(6) 1577 – 1581 (1998).
A. Kunkel, S. Günter, and H. Wätzig, Electrophoresis, 8(10), 1882 – 1889 (1997).
P. M. Castellano, S. E. Vignaduzzo, and R. M. Maggio, Anal. Bioanal. Chem., 382(7), 1711 – 1714 (2005).
M. S. M. Quintino, K. Araki, H. E. Toma, et al., Electroanalysis, 14(23), 1629 – 1634 (2002).
H. H. Maurer, Anal. Bioanal. Chem., 388(7), 1315 – 1325 (2007).
R. Kachoosangi, G. Wildgoose, and R. Compton, Anal. Chim. Acta, 618(1), 54 – 60 (2008).
S. A. Kumar, C. F. Tang, and S. M. Chen, Talanta, 76(5), 997 – 1005 (2008).
S. Shahrokhian and E. Asadian, Electrochim. Acta, 55(3), 666 – 672 (2010).
S. Mehretie, S. Admassie, M. Tessema, et al., Anal. Bioanal. Electrochem., 3(1), 38 – 50 (2011).
M. Amare and W. Teklay, Cogent Chem., 5(1), 1 – 10 (2019).
Y. Wei, A. Wang, and Y. Liu, Rus. J. Electrochem., 54(12) 1141 – 1147 (2018).
J. McCaffrey, W. Henderson, B. K. Nicholson, et al., J. Chem. Soc. Dalton Trans., 36, 2577 – 2586 (1997).
M. J. Gismera, J. R. Procopio, M. T. Sevilla, et al., Electroanalysis, 15(2), 126 – 132 (2003).
A. K. Chhakkar and L. R. Kakkar, Fresenius J. Anal. Chem., 347(12), 483 – 485 (1993).
D. Shander, G. Ahluwalia, and D. Grosso, Method of Reducing Hair Growth Employing Sulfhydryl Active Compounds, US Patent 5,411,991 A (1992).
M. Aydin, N. Arsu, and Y. Yagci, Macromol. Rapid Commun., 24(12), 718 – 723 (2003).
H. Jacobelli, 3- or 4-Monosubstituted Phenol and Thiophenol Derivatives Useful as H3 Ligands, US Patent 20,050,267,095A1 (2005).
J. H. Wiener, Y. Kloog, V. Wacheck, et al., J. Invest. Dermat., 120(1), 109 – 115 (2003).
G. Elad, A. Paz, R. Haklai, et al., J. Biochem. Biophys. Acta, 1452(3), 228 – 242 (1999).
B. D. Clayton, Y. N. Stock, Basic Pharmacology for Nurses, Mosby Inc., Harcourt Health Sciences Company, St. Louis (2001).
J. L. N. de Aguiar, K. C. Leandro, S. M. P. Abrantes, et al., Braz. J. Pharm. Sci., 45(4), 723 – 727 (2009).
A. R. Medina, M. L. F. de Cordoba, and A. M. Dínaz, Fresenius J. Anal. Chem., 365(7), 619 – 624 (1996).
A. V. Pereira, C. Aniceto, and O. Fatibello-Filho, Analyst, 123(5), 1011 – 1015 (1998).
N. P. Shetti, D. S. Nayak, G. T. Kuchinad, et al., Electrochim. Acta, 269, 204 – 211 (2018).
A. A. J. Torriero, J. M. Luco, L. Sereno, et al., Talanta, 62(2), 247 – 254 (2004).
Y. Dai, J. van Spronsen, G.-J.Witkamp, et al., Anal. Chim. Acta, 766, 61 – 68 (2013).
J. Irudayaraj, and J. Tewari, J. Appl. Spectrosc., 57(12), 1599 – 1604 (2003).
H. Wang, Y. Jia, X. Wang, et al., J. Chil. Chem. Soc., 57(3), 1208 – 1212 (2012).
V. M. Parikh, Absorption Spectroscopy of Organic Molecules, Addison Wesley Publishing Company (1974).
Y. Li and S.-M. Chen, Int. J. Electrochem. Sci., 7(3), 2175 – 2187 (2012).
A. R. Kulkarni, N. P. Shetti, S. J. Malode, et al., Materials Today: Proceedings, 18(3), 723 – 730 (2019).
A. J. Bard, L. R. Faulkner, Electrochemical Methods: Fundamentals and Applications, Wiley, New York (2001).
R. S. Nicholson and I. Shain, Anal. Chem., 36(4), 706 – 723 (1964).
H. Nah, J. Yim, S.-G. Lee, et al., Ann. Lab. Med., 36(2), 188 – 190 (2016).
A. Yiðit, Y. Yardým, and Z. Þentürk, J. Anal. Chem., 75(5), 653 – 661 (2020).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zarei, E., Vafadar, M. & Asghari, A. Acetaminophen and Thiosalicylic Acid Sensor Based on Carbon Paste Electrode Modified with Multi-Walled Carbon Nanotubes and Natural Deep Eutectic Solvent. Pharm Chem J 57, 1862–1871 (2024). https://doi.org/10.1007/s11094-024-03090-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-024-03090-5