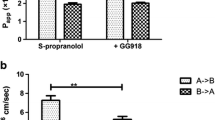
P-glycoprotein (P-gp) mediated efflux affects the pharmacokinetics of several drugs. By analogy to verapamil, 1,4-dihydropyridines (DHPs) have been widely studied as P-gp inhibitors. Previously, we have reported on two new DHPs: IA1(A) and IIA5(B) as inhibitors of human MRP1, an efflux protein closely related to P-gp. The aim of the present study was to investigate the inhibitory effects of these two compounds on intestinal P-gp using the method of in situ single-pass intestinal perfusion (SPIP) in rat. According to this, the intestinal absorption of zidovudine (a P-gp substrate) was studied in anaesthetized rat jejunum in the absence and presence of DHPs IA1(A) and IIA5(B) (2 mg/kg). Verapamil (0.8 mg/kg), a well-known P-gp inhibitor, was employed as a standard. Zidovudine solution (200 ig/mL) in phosphate buffer (pH 7.4) was perfused through the jejunal segment, the perfusate concentrations were quantified by HPLC, and the permeability coefficient (Peff) and fraction absorbed (Fabs) were calculated. Phenol red was used as a non-absorbable marker to correct water flux through the segment. In rats pretreated with compounds IA1 and IIA5, Peff and Fabs of zidovudine were found to be 0.1669 ± 0.12 cm/sec, 0.2035 ± 0.18 and 0.2798 ± 0.12 cm/sec, 0.3015 ± 0.14, respectively, and were comparable to those of the standard (Peff = 0.462713 ± 0.3 cm/sec, Fabs = 0.511835 ± 0.14). The differences between IA1, IIA5 and the standard were evaluated using ANOVA and found to be statistically significant (P < 0.05). Compounds IA1 and IIA5 have a modulating effect on intestinal P-gp. Compound IIA5 was relatively more potent P-gp inhibitor and, quite interestingly, the results were in agreement with our earlier in silico and in vitro studies.




Similar content being viewed by others
References
M. K. Arunasree and R. Arshad, Antimicrob. Agents Chemother., 55, 439 – 442 (2011).
E. Teodori, S. Dei, C. Martelli, et al., Curr. Drug Targets, 7, 893 – 909 (2006).
C. F. Higgins, Nature, 446, 749 – 757 (2007).
J. P. Gillet and M. M. Gottesman, Methods Mol. Biol., 596, 47 – 76 (2010).
T. Tsuruo, H. Iida, M. Nojiri, et al., Cancer Res., 43, 2905 – 2910 (1983).
P. A. Philip, S. Joel, S. C. Monkman, et al., Brit. J. Cancer, 65, 267 – 270 (1992).
A. Zarrin, A. R. Mehdipour, and R. Miri, Chem. Biol. Drug Des., 76, 369 – 381 (2010).
H. Engi, H. Sakagami, M. Kawase, et al., In Vivo, 20, 637 – 644 (2006).
X. F. Zhou, R. A. Coburn, and M. E. Morris, J. Pharm. Sci., 94, 2256 – 2265 (2005).
A. Shah, J. Bariwal, J. Molnar, et al., Top. Heterocycl. Chem., 15, 201 – 252 (2008).
A. T. Fojo, K. Ueda, D. J. Slamon, et al., Proc. Natl. Acad. Sci. USA, 84, 265 – 269 (1987).
M. V. S. Varma, M. Sarkar, et al., J. Chromatogr. B, 816, 243 – 249 (2005).
S. V. Ambudkar, S. Dey, C. A. Hrycyna, et al., Annu. Rev. Pharmacol. Toxicol., 39, 361 – 398 (1999).
R. L. Linardi and C. C. Natalini, Ciência Rural, Santa Maria, 36, 336 – 341 (2006).
N. Akhtar, A. Ahad, R. K. Khar, et al., Expert Opin. Ther. Pat., 21, 561 – 576 (2011).
T. Terao, E. Hisanaga, Y. Sai, et al., J. Pharm. Pharmacol., 48, 1083 – 1089 (1996).
B. Greiner, M. Eichelbaum, P. Fritz, et al., J. Clin. Invest., 104, 147 – 153 (1999).
N. Nagare, A. Damre, K. S. Singh, et al., Indian J. Pharm. Sci., 72, 625 – 629 (2010).
L. Barthe, J. Woodley, and G. Houin, Fund. Clin. Pharmacol., 13, 154 – 168 (1999).
N. Prasad, G. Dinesh, and B. Sathish Kumar, Eur. J. Pharm. Sci., 44, 27 – 31 (2011).
N. F. Ho, J. Y. Park, P. F. Ni, et al., in: Animal Models for Oral Drug Delivery in Man, W. Crouthamel and A. Sarapu (eds.), Amer. Pharm. Assoc., Washington DC (1983), pp. 27 – 106.
H. H. Lu, J. Thomas, and D. Fleisher, J. Pharm. Sci., 81, 21 – 25 (1992).
U. Fagerholm, M. Johansson, and H. Lennernas, Pharm. Res., 13, 1336 – 1342 (1996).
B. H. Stewart, O. H. Chan, R. H. Lu, et al., Pharm. Res. 12, 693 – 699 (1995).
K. Sirisha, M. Chandra Shekhar, K. Uma Sankar, et al., Bioorg. Med. Chem., 19, 3249 – 3254 (2011).
H. Saitoh and B. J. Aungst, Pharm. Res., 12, 1304 – 1310 (1995).
Guidelines for Acute Oral Toxicity of Chemicals, Test No. 425 (Adopted 3 October 2008), Organization for Economic Cooperation and Development (OECD).
D. Fleisher, K. C. Johnson, B. H. Stewart, et al., J. Pharm. Sci., 75, 934 – 939 (1986).
L. Salphati, K. Childers, L. Pan, et al., J. Pharm. Pharmacol., 53, 1007 – 1013 (2001).
P. Zakeri-Milania, V. Hadi, T. Hosnieh, et al., J. Pharm. Pharmaceut. Sci., 10, 368 – 379 (2007).
N. Sunil Thomas and P. Ramesh, Eur. J. Pharm. Sci., 18, 71 – 79 (2003).
R. Sandstrom and H. Lennernas, Drug Metab. Dispos., 27, 951 – 955 (1999).
Acknowledgements
The authors are grateful to UCPSc, Kakatiya University (Warangal, TS, India) for providing the facilities. One of the authors (K. S.) is thankful to the University Grants Commission (UGC, New Delhi, India) for the award of SRF and to the management of Vaagdevi College of Pharmacy (Warangal, TS, India) for their encouragement.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sirisha, K., Achaiah, G., Prasad, N. et al. Multidrug Resistance Reversal Activity of Some New Dihydropyridines Studied by IN SITU Single-Pass Intestinal Perfusion (SPIP) Method in Rat. Pharm Chem J 52, 8–14 (2018). https://doi.org/10.1007/s11094-018-1757-x
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-018-1757-x