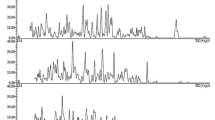
An HPLC-MS/MS method for determining dabigatran in human blood serum was developed. Samples were prepared by precipitation of proteins using MeOH followed by centrifugation and dilution with deionized H2O. The developed method had a simple sample-preparation procedure, short analysis time, and wide analytical range (from 1 to 1,000 ng/mL). This method was suitable for routine bioanalytical studies, in particular, therapeutic drug monitoring.










Similar content being viewed by others
References
WHO. Cardiovascular Diseases. Informational Bulletin No. 317, January, 2015; http: //www.who.int/mediacentre/factsheets/fs317/ru/[http: //www.who.int/mediacentre/factsheets/fs317/en/].
S. Stewart, C. L. Hart, D. J. Hole, and J. J. V. McMurray, Heart, 86(5), 516 – 521 (2001).
A. S. Go, E. M. Hylek, K. A. Phillips, et al., J. Am. Med. Assoc., 285(18), 2370 – 2375 (2001).
Atrial Fibrillation Investigators, “Risk factors for stroke and efficacy of antithrombotic therapy in atrial fibrillation: analysis of pooled data from five randomized clinical trials,” Arch. Intern. Med., 154, 1449 – 1457 (1994).
P. Kirchhof, S. Benussi, D. Kotecha, et al., Eur. Heart J., 37(38), 2893 – 2962 (2016).
L. Wallentin, S. Yusuf, M. D. Ezekowitz, et al., Lancet, 376(9745), 975 – 983 (2010).
Pradaxa Prescribing Information; http: //www.docs.boehringer-ingelheim. com/Prescribing%20Information/PIs/Pradaxa/Pradaxa.pdf (accessed Nov. 10, 2007).
S. Blech, T. Ebner, E. Ludwig-Schwellinger, et al., Drug Metab. Dispos., 36(2), 386 – 399 (2008).
W. Ageno, A. S. Gallus, A. Wittkowsky, et al., Chest, 141(2), e44S-88S (2012).
M. V. Huisman, G. Y. Lip, H. C. Diener, et al., Thromb. Haemostasis, 107(5), 838 (2012).
A. J. Camm, G. Y. Lip, R. De Caterina, et al., Eur. Heart J., 33(21), 2719 – 2747 (2012).
K. H. Liesenfeld, T. Lehr, C. Dansirikul, et al., J. Thromb. Haemostasis, 9(11), 2168 – 2175 (2011).
G. Pare, N. Eriksson, T. Lehr, et al., Circulation, 127(13), 1404 – 1412 (2013).
T. Ebner, K. Wagner, and W. Wienen, Drug Metab. Dispos., 38(9), 1567 – 1575 (2010).
F. Scaglione, Clin. Pharmacokinet., 52(2), 69 – 82 (2013).
T. Hellwig and M. Gulseth, Ann. Pharmacother., 47(11), 1478 – 1487 (2013).
Z. Y. Hu, R. B. Parker, and V. L. Herring, Anal. Bioanal. Chem., 405(5), 1695 – 1704 (2013).
P. A. Reilly, C. A. Conrad, R. A. Faaij, et al., Eur. Heart. J., 32(1), 6 (2011).
X. Delavenne, J. Moracchini, S. Laporte, et al., J. Pharm. Biomed. Anal., 58, 152 – 156 (2012).
M. Korostelev, K. Bihan, L. Ferreol, et al., J. Pharm. Biomed. Anal., 100, 230 – 235 (2014).
J. P. Antovic, M. Skeppholm, J. Eintrei, et al., Eur. J. Clin. Pharmacol., 69(11), 1875 – 1881 (2013).
J. Douxfils, L. Pochet, S. Lessire, et al., TrAC, Trends Anal. Chem., 84, 41 – 50 (2016).
J. Douxfils, J. M. Dogne, F. Mullier, et al., Thromb. Haemostasis, 110(3), 543 – 549 (2013).
J. Douxfils, H. Mani, V. Minet, et al., BioMed Res. Int., 2015, Article ID 345138 (2015).
A. N. Mironov (ed.), Guideline for Drug Review, Vol. 1 [in Russian], Grifi K, Moscow (2013).
Author information
Authors and Affiliations
Additional information
Translated from Khimiko-Farmatsevticheskii Zhurnal, Vol. 51, No. 12, pp. 57 – 64, December, 2017.
Rights and permissions
About this article
Cite this article
Rodina, T.A., Mel’nikov, E.S., Aksenov, A.A. et al. HPLC-MS/MS Method for Determining Dabigatran in Human Blood Serum. Pharm Chem J 51, 1129–1137 (2018). https://doi.org/10.1007/s11094-018-1753-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-018-1753-1