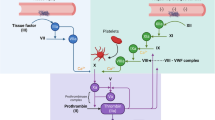
Two different literature methods were used to compare the experimental efficacy and accuracy of dabigatran assays in blood of 30 patients with knee replacements. Blood plasma was collected from patients who underwent anticoagulant therapy and were administered the medicine at a dose of 220 mg. Residual and peak dabigatran concentrations were determined by HPLC-MS and HPLC-MS/MS.
Similar content being viewed by others
References
V. T. Vavilova, Med. Sovet, No. 12, 44 – 47 (2015).
I. N. D’yakov, Remedium, No. 11, 42 – 43 (2014).
“Russian clinical recommendations for diagnosis, treatment, and prevention of complications from venous thromboembolism,” Flebologiya, 9(4), 1 – 52 (2015).
N. G. Khorev, A. P. Momot, and D. A. Zaloznyi, Tromb., Gemostaz Reol., No. 4(44), 31 – 47 (2010).
A. Dabi and A. P. Koutrouvelis, “Reversal strategies for intracranial hemorrhage related to direct oral anticoagulant medications,” Crit. Care Res. Pract., URL: https://doi.org/10.1155/2018/4907164.
A. Bromlcy and A. Plitt, J. Cardiol. Ther., 7(1), 1 – 13(2018).
Mosaad Almegren, Vasc. Health Risk Manage., 13, 287 – 292 (2017).
G. Lippi and E. Favaloro, Clin. Chem. Lab. Med., 53(2), 1 – 13 (2014).
V. Taune, M. Skeppholm, A. Agren, et al., J. Thromb. Haemostasis, 16(12), 2462 – 2470 (2018).
J. Stangier, K. Rathgen, H. Stahle, et al., Br. J. Clin. Pharmacol. B, 64(3), 292 – 303 (2007).
M. Korostelev, K. Bihan, L. Ferreol, et al., J. Pharm. Biomed. Anal., 100, 230 – 235 (2014).
J. Kuhn, T. Gripp, T. Flieder, et al., PLoS One, 10(12), 1 – 19 (2015).
E. M. H. Schmitz, K. Boonen, D. J. A. van den Heuvel, et al., J. Thromb. Haemostasis., 12, 1636 – 1646 (2014).
D. A. Sychev, A. N. Levanov, T. N. Shelekhova, et al., Pharmacogenomics Pers. Med., 11, 127 – 137 (2018).
J. Harenberg, S. Kraemer, S. Du, et al., Semin. Thromb. Hemostasis, 40(1), 129 – 134 (2014).
X. Delavenne, J. Moracchini, S. Laporte, et al., J. Pharm. Biomed. Anal., 58, 152 – 156 (2012).
Handbook for Drug Review [in Russian], Vol. 1, Grif i K, Moscow, 2014.
Guideline on Validation of Bioanalytical Methods (Draft), European Medicines Agency, Committee for Medicinal Products for Human Use, London, 2009.
Bioanalytical Method Validation, Guidance for Industry, U. S. Department of Health and Human Services, Food and Drug Administration, Center for Drug Evolution and Research (CDER). U. S. Government Printing Office,Washington, 2001.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiko-Farmatsevticheskii Zhurnal, Vol. 53, No. 8, pp. 59 – 63, August, 2019.
Rights and permissions
About this article
Cite this article
Kozlov, A.V., Smirnov, V.V., Sychev, D.A. et al. Comparison of Quantitative Analytical Techniques for Dabigatran in Blood Plasma of Humans with Knee Replacements. Pharm Chem J 53, 771–774 (2019). https://doi.org/10.1007/s11094-019-02077-x
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-019-02077-x