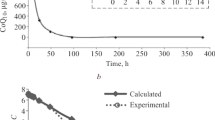
The dynamics of ubidecarenone (coenzyme Q10) levels in rat blood plasma and liver were monitored for two days after i.v. injection of solubilized ubidecarenone at doses of 10 and 30 mg/kg. The kinetic curves of the drug in plasma were exponential for both doses. The areas under the concentration—time curves differed by 8.6 times for the 10 and 30 mg/kg doses. Normalization to the dose did not superimpose them. The drug accumulated gradually in liver with the areas under the concentration—time curves differing by a factor of 4.4. These data indicated that ubidecarenone pharmacokinetics in plasma and liver were nonlinear after intravascular injection.


Similar content being viewed by others
References
J. Garrido-Maraver, M. D. Cordero, M. Oropesa-Avila, et al., Front. Biosci. (Landmark Ed.), 19, 619 – 633 (2014).
M. Spindler,M. F. Beal, and C. Henchcliffe, Neuropsychiatr. Dis. Treat., 5, 597 – 610 (2009).
E. I. Kalenikova, E. A. Gorodetskaya, and O. S. Medvedev, Pharmaceutical Analysis [in Russian], Vol. 16, Argamak-Media, Moscow (2013), pp. 616 – 653.
H. N. Bhagavan and R. K. Chopra, Free Radical Res., 40(5), 445 – 453 (2006).
Acknowledgments
The studies were performed under the auspices of a grant from the Russian Scientific Foundation (Project No. 14-15-00126).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiko-Farmatsevticheskii Zhurnal, Vol. 48, No. 12, pp. 7 – 8, December, 2014.
Rights and permissions
About this article
Cite this article
Kalenikova, E.I., Gorodetskaya, E.A., Belousova, M.A. et al. Estimation of the Linearity of Ubidecarenone Pharmacokinetics After Intravenous Administration. Pharm Chem J 48, 775–776 (2015). https://doi.org/10.1007/s11094-015-1193-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-015-1193-0