Abstract
Atmospheric nonthermal plasma (ANTP) is used for various biological applications including seed quality improvements in crop production. However, the underlying mechanisms of plasma-induced seed action are not fully explained and operating large-scale ANTP on seeds is highly challenging. Two large-scale ANTPs, streamer corona plasma (SCP) and dielectric barrier discharge (DBD) plasma, were used to enhance rice seed vigor through surface modification and functionalization. The SCP and DBD plasma were conducted under the same power source and applied to rice seeds to modify their surface from being highly hydrophobic to being highly hydrophilic, as defined by the apparent contact angle measurement. The results show that SCP requires less treatment time (~ 2 min) for surface activation than the DBD plasma process (~ 10 min). Both plasma-treated seeds showed higher seed vigor than non-treated seeds. For 50% of the viable seeds to emerge, the SCP-treated seeds took on average about 62 h., while the untreated rice seeds took around 72 h. The germination percentage of all conditions is ~ 92% which is a typical proportion of good seed quality. The microstructure of the rice seed surface suggests that the bombardment of highly energetic ions and the reaction of reactive oxygen and nitrogen species on the seed surface cause morphological changes via surface etching and functionalization without any adverse effects on seed nutrition.
Graphical abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
With the expansion of the global population, the demand for rice as a staple food is rapidly growing [1,2,3]. Various technologies and methods have been introduced to improve rice seed quality and increase crop yield to meet food security requirements [1, 3,4,5]. Numerous indicators are used to assess seed quality such as seed lot purity, seed viability, and seed health, however, seed vigor is considered one of the most significant parameters [4, 6,7,8,9]. The level of seed vigor indicates the ability of the seeds to germinate and establish seedlings rapidly, uniformly, and robustly in a field across diverse environmental conditions. As a result, using seed vigor enhancement technology to achieve rapid and uniform rice seed germination has proven to be an effective method. Hence, it is a primary objective within the agricultural industry's breeding and seed production programs [4]. Several techniques, such as chemical treatments (chemicals, fungicides, and plant growth regulators) and physical treatments (ultrasonic scratching and electric field treatments), have been used for seed enhancement [5, 10,11,12,13,14,15]. However, both are labor-intensive, safety concerns, and costly due to large quantities of chemical consumption and chemical residues [5, 15]. In contrast, with a mixture of free radicals, negative and positive ions, molecules, reactive species (RS), and electrons, atmospheric nonthermal plasma—ANTP—technologies have been developed as an alternative nonthermal emerging technology for food and agricultural applications. ANTP technology is capable of inactivating or decontamination of microorganisms without inducing thermal damage and produces no hazardous residues [5, 16,17,18].
Initially, low-pressure plasma was used to treat biomaterials, including seeds, because it produces homogeneously distributed plasma in an entire operating area [17, 19, 20]. However, the low-pressure plasma requires a vacuum system to generate and control the plasma phase, this is considerably expensive and complex [16, 19]. Additionally, certain biomaterials are unable to withstand low-pressure settings without deteriorating [21, 22]. Subsequently, the advent of ANTP has attracted substantial attention due to its ability to treat biological and agricultural materials in an ambient environment. As a result, ANTP, also known as ‘cold plasma’, has been utilized for many agricultural purposes, from seeds to harvest [5, 15, 21, 23,24,25], particularly in rice production [17, 26,27,28]. Several reports demonstrate that ANTP treatment can enhance seed imbibition and germination [21, 25, 27, 29]. Seed germination requires a complicated network of hormones, metabolism, and developmental changes that do not occur if the seed is not in the ideal levels of moisture, oxygen, temperature, and light [21, 30]. Our previous report showed the sterilization capability of streamer plasma [27] however, there is no report on the subsequent effect on rice seed vigor or the underlying mechanism behind the phenomena. Only a few ANTP structures can be practically used in large-scale applications that can treat more than 10,000 seeds for each operation, especially in agriculture. Generally, scaling up an atmospheric cold plasma system is challenging due to the difficulty in controlling the plasma state which involves several reactor designs and process parameters, such as a high electric field, high discharge voltage, and a high gas flow rate, all of which require a large power source [19, 31, 32]. All previous research has been conducted at the laboratory stage and scale, with ~ 20 g of rice seed/grain being the maximum treated capacity [17, 27, 33]. However, nutrition and field tests require at least 0.3–1 kg to meet the standardized experimental parameters and volume. Therefore, scaling up the ANTP area is currently the primary challenge that needs attention. Recently, electrical discharge phenomena such as corona discharge and dielectric barrier discharge (DBD) have become a promising approach for producing ANTP systems because they can take place at atmospheric pressure. Electrical gas discharge mainly relies on the electric field distribution between the electrode geometrical configurations and dielectric permittivity [32, 34, 35]. Generated by highly localized electric fields associated with sharp electrodes, corona discharge requires relatively low power and appears in several forms, such as streamer corona, glow corona, and spark discharge [16, 31, 36, 37]. However, corona discharge plasma is extremely difficult to control because it can quickly spark (arc) [31, 36]. In contrast, the DBD structure typically consists of two parallel electrode plates with dielectric layers placed between the electrodes which provide micro discharge filaments of nanosecond duration with more stability and homogeneity [16, 37, 38]. However, with the proper electrode and power source design, both electrical discharge plasma can be obtained with low operational power.
Here, we present two types of large-scale electrical discharge plasma systems, streamer corona plasma (SCP) and DBD plasma, used for treating 1 kg of rice seeds each. We studied and compared the effects of the SCP and DBD plasma on rice seed enhancement, germination, and radicle emergence—RE—speed under the same power source. Optical emission spectroscopy was performed to elucidate the underlying mechanism and essential reactive species that affect seed quality during treatment. A scanning electron microscope was employed to understand the plasma-induced physiochemical response of the seed surface after SCP and DBD plasma applications. The primary nutritional value of rice seeds was defined to substantiate whether the application of plasma degrades physical and physiological seed quality. This overall investigation is helpful not only for future ANTP design parameters but also for improving the efficiency and effectiveness of cold plasma for seed enhancement.
Materials and Methods
Experimental Setup
Two large-scale ANTPs, SCP and DBD plasma, were designed and fabrication. Then, the developed ANTPs were validated using optical emission spectroscopy (OES) and apparent contact angle (ACA) measurement.
Atmospheric Nonthermal Plasma Design and Fabrication
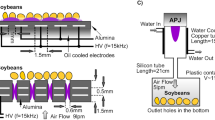
Figure 1a demonstrates the atmospheric nonthermal SCP treatment of rice seeds. The SCP is generated by using a hybrid structure, combining the corona discharge plasma and DBD plasma [16, 27, 32]. Rice seeds were placed on a dielectric layer laid on top of the grounded electrode. There are approximately 20.3 seeds per 1 g, so an electronic balance was used to count the rice seeds before treatment. Through this way, we treated approximately 19,500 seeds. The top electrode was the sharp tip array connected to a high voltage alternating current (AC) power supply (7 kV with a repetition rate of 1 kHz). The gap distance between the top and bottom electrodes was set to 0.7 cm, and the operational gas was air mixed with Ar (Ar-air) by flowing argon to mix with the surrounding air at a flow rate of 2 L/min. The SCP was generated and directly radiated to the seed under ambient atmospheric conditions (Fig. 1a-bottom). Although the SCP method applies plasma disproportionally to the top of the seed surface, our recent work shows that due to the combination of corona and the DBD structure the whole seed surface (top and bottom) is exposed to plasma and is consequently modified [28, 32, 39]. Figure 1b demonstrates the DBD plasma structure employing the same plasma source as the SCP. The DBD structure comprises of two metal plate (top and bottom) electrodes, and glass containers which serve as two dielectric layers covering the top and bottom planar electrodes with a gap of approximately 0.7 cm. In the DBD process, the working gases are also a mixture of Ar and air. The Ar with a flow rate of 0.5 L/min is injected into the plasma active area which is controlled but a flow meter. Thus, DBD provides the microscale filament discharge plasma entirely inside a closed container and covers all seed surfaces (Fig. 1b-bottom). However, both electrical discharge plasma structures have fundamental differences in electric field distribution, electrode structures, and cold plasma generation [16, 31]. For example, the SCP employed a sharp tip electrode to induce a highly localized electric field (105 V/cm) to generate corona discharge plasma [16, 31, 32]. In contrast, DBD employs two dielectric layers to induce a large electric field in the airgap and is significantly low in the dielectric layer itself (103 V/cm) [39]. As a result, the ANTPs will have different amounts of plasma components and interaction rates on the rice seeds. After treatments, the plasma-treated seed was thoroughly mixed and drawn randomly for validating the plasma treatments and evaluate their effects on seed quality.
Electrical discharge plasma on rice seed. a Schematic diagram of streamer corona plasma (SCP) treatment of rice seeds (top) and the optical image (bottom, scale bar = 7 mm; Ar and HV denote argon and high voltage, respectively). b Schematic diagram of dielectric barrier discharge (DBD) plasma treatment of rice seeds (top) and the optical image (bottom, scale bar = 7 mm)
Validation of the Atmospheric Nonthermal Plasma
To elucidate and compare the rice seed enhancement after surface modification and functionalization via SCP and DBD plasma, rice seeds were treated via each plasma process until the seed surface became modified entirely, defined by the ACA measurement. Optical emission spectroscopy (Thorlabs CCS200, wavelength 200−1000 nm) coupled with fiber optics was employed to acquire the optical emission spectrum of reactive species generated during plasma treatment. For apparent contact angle measurement, the ACA of a 0.25 µl water droplet from a well-controlled micropipette (0.1–2 µl, Bravo, CAPP, Denmark) was measured under an optical microscope in conjunction with computer-aided measurement.
Typically, non-treated rice seed surfaces are highly hydrophobic, having ACA 120° after an 0.25 µL water droplet application (Fig. 2a-top). Therefore, each plasma process was performed on rice seeds until the seed surface was transformed entirely to a superhydrophilic state (ACA 0°) (Fig. 2a middle and bottom). The result shows that the SCP requires considerably less treatment time (2 min) to complete hydrophilic surface transformation when compared to the DBD plasma (10 min). The high efficiency of the modified surface via SCP is probably due to the energy requirement for producing SCP being much less than that of DBD at the same power source and electrode distance [31, 39], resulting in a stronger impact ionization and consequently a higher physical–chemical reaction on the seed surface. To verify this hypothesis, we performed optical emission spectroscopy during DBD (Fig. 2b-top) and SCP (Fig. 2b-bottom) because the change in ACA is attributed to the physical, by etching process via energetic particles, and chemical reaction of reactive oxygen and nitrogen species (RONS) on the seed surface during plasma treatment, turning hydrophobic to hydrophilic [27, 40]. The OES shows that the RONS generated in the range of ultraviolet spectra (250–450 nm) [18, 37], ·OH (309 nm), N2+ (391 nm), N2, or NO lines (316, 337, 358, 427.5 nm) in the DBD structure (Fig. 2b top-inset) is significantly lower than those presented in the SCP structure (Fig. 2b-bottom). As a result, the SCP requires a shorter treatment time for surface activation. RONS are recognized as the most active plasma components for seed treatment due to their ability to initiate oxidation processes critical for the degradation of organic compounds [41, 42], inactivate microorganisms [27, 28, 37], and enhance surface wettability [23, 43].
Electrical discharge plasma on rice seed. a The apparent contact angle—ACA—of water droplets on (top) non-treated, (middle) after 2 min streamer corona plasma—SCP—treatment and (bottom) after 10 min dielectric barrier discharge—DBD—seed (scale bar 3.5 mm). b Optical emission spectra (OES) of DBD plasma (top) and SCP (bottom) during treatment from the same plasma source and electrode distance
Application of the SCP and DBD Plasma for Rice Seed Enhancement
Seed Germination and Vigor Test
Seeds (Oryza sativa var. Indica cv. KDML105) were obtained from the Rice Department. Before the experiment, the samples were stored at 5 °C and 12% relative humidity (RH). Germination tests were conducted immediately after plasma treatment following ISTA guidelines [9], using four replicates of 100 seeds each. The seeds were placed on moistened blotter paper and stored in a transparent box in a plant-growth chamber (Daihan Labtech, Model LGC-5201) that was set to 20 °C in the dark for 16 h. and 30 °C under cool white lamps (~ 1200 lx) for 8 h. at 85% RH.
Seed vigor was verified using the RE test [9]. According to the International Seed Testing Association (ISTA), seed vigor is “the sum of those properties that determine the activity and performance of seed lots of acceptable germination in a wide range of environments” [9]. Currently, [8] propose that the extent of previous deterioration and the repair period can determine the length of the lag period and therefore the percentage germination in rice. After seed imbibition, cellular repair activities promoted recovery from damage incurred during quiescence, and the speed and efficacy of repair were closely linked to RE and germination performance [44, 45]. As a result, the RE test was designed to evaluate rice seed vigor after plasma treatment in this study. Seed RE was evaluated using techniques similar to those expressed for the germination test, but the RE was counted at 4 h. intervals up to 104 h. after the start time. Calculation of the RE time (tRE), the mean RE time (MRET) and the time required for 50% of viable seeds to emerge (t50RE), were conducted using GERMINATOR software [46]. The software was used in combination with the least sum of squares method to find the right parameters to fit the cumulative RE curves to the four-parameter Hill function.
where y is the cumulative RE percentage at time x (hr.), y0 is the intercept on the y axis (≥ 0), a is the maximum cumulative RE percentage (≤ 100), b is controlling the shape and steepness of the curve and c is the t50RE. The MRET describes the average time for a seed to RE, or the delay (lag period) from the start of imbibition to RE. In Fig. 4a, seed lot has the longest average delay (high MRET), is the latest to start to germinate and has the greatest spread of RE over time. Following the seed ageing/repair hypothesis [6], the lower the values of MRET and t50RE are the better the seed quality (high seed vigor).
Scanning Electron Microscopy (SEM)
Plasma-treated and non-treated rice seeds with a moisture content of 5 − 8% were attached to aluminum stubs, sputtered with platinum in a vacuum evaporator, and visualized with a scanning electron microscope (S-4800, HITACHI, Japan).
Nutrient Contents
Seed moisture content was evaluated from three hundred grams of rice seed samples using the analytical methods adopted by the Association of Official Analytical Chemists [47]. The major nutrients, carbohydrate, fat, and protein, were analyzed according to the in-house method TE-CH-042 based on AOAC [48].
Statistical Analysis
The data reported in this article are the means of quadruplicate analyses, along with their standard errors. To determine the significance of the differences between the means, one-way ANOVA and Tukey's HSD post hoc tests were performed for analytical analysis. The threshold for a substantial difference was set at 5%. The applications of these statistical analyses were carried out using the R software package [49].
Results and Discussion
Application of the SCP and DBD Plasma for Rice Seed Enhancement
Responses of rice seed quality to large-scale SCP and DBD plasma were determined by following the sequence in the methodology. Figure 3a shows the effects of plasma on the RE percentage of rice seeds after DBD and SCP treatments. The 100 rice seeds were separately exposed to DBD and SCP plasma for 10 and 2 min, respectively. Then, the RE test was performed immediately after the plasma treatments in accordance with ISTA guidelines [9], with four replicates of each treatment (see also Materials and Methods). RE was counted after producing a 2 mm radicle (Fig. 3d and e). GERMINATOR software [46] was utilized to calculate the tRE, t50RE, and MRET, indicating the seed vigor level (Fig. 4a and b). The results showed that plasma-treated seeds require less tRE than non-treated seeds (Fig. 3a). For example, at 55 h., after the set time the percentage of RE of DBD- and SCP-treated seeds was 18.5% and 24.5%, respectively, while that of non-treated seeds was only 8.25%. From seed aging/repair hypothesis, the RE speed would imply the vigor and field emergence of indica rice seed [8, 50]. DNA repair mechanisms can reverse damage to cellular components, restoring cellular function prior to the initiation of RE and growth post-germination [44]. DNA repair is linked to the cell expansion in RE in the many species including rice seeds [44]. This can delay RE and field establishment of grain crops [6, 45]. Based on this hypothesis, the results suggested that both SCP and DBD provide higher seed vigor than non-treated seeds. Figure 3b. shows the effect of plasma treatment on RE time. The results showed that the SCP-treated rice seeds had the lowest t50RE, ~ 62 h. (Fig. 3b left, blue). However, there was no significant difference in t50RE between DBD-treated seeds (red color) and non-treated seeds (black color), at approximately 70 h. The MRET of all conditions was similar to the t50RE, indicating no significant deviation of RE between seed lots. Thus, the overall result confirms that the SCP strongly impacts rice seed functionalization and modification, resulting in higher seed vigor enhancement. Interestingly, the germination percentage after DBD and SCP treatments was not different from non-treated seeds (control) (Fig. 3c-left). The germination percentage was approximately 92%, which is the typical percentage of good seed quality [6, 27]. On average, 14-day seedlings of plasma-treated seeds showed longer shoot lengths than the control group. Statistically significant increases in shoot length were observed in response to the DBD plasma treatment (Fig. 3c). The large-scale SCP and DBD plasma had no effect on root and shoot growth of rice seeds. Seedlings showed all their essential structures that well developed and healthy (Fig. 3f). Thus, the results indicated that neither plasma treatment harmed the seeds but improved the characteristics of seedling growth. Seed vigor enhancement via plasma treatments demonstrates its ability to yield vigorous seedlings that become robust and uniform plants.
Seed vigor and quality enhancement via electrical discharge plasma. a Single counts of radicle emergence in rice seed (Oryza sativa L.) after cold plasma process. b Radicle emergence time (t50RE) and mean radicle emergence time (MRET) of treated rice seeds. c Germination percentage and seedling length of rice seeds after cold plasma treatment. d, e Representative picture of radicle emergence at 55 h and 77 h after beginning to germinate of DBD-treated seeds, respectively (scale bar 3.5 cm). f Representative pictures of seedlings following the control, DBD treatment and SPC treatment; from left to right (scale bar 3.5 cm). The error bar denotes the standard error (n = 4). Means within a column of each bar with the same lowercase letters are not significant at p ≤ 0.05 based on Tukey’s honest significant difference test
Rice seed vigor test using GERMINATOR software via cumulative radicle emergence curves. a Cumulative radicle emergence data is used as input for the curve fitting module. Multiple radicle emergence parameters are automatically extracted. Gmax indicates the maximum germination capacity of a seed lot. The time required for 50% of viable seeds to germinate and mean radicle emergence time are the t50RE and MRET, respectively. b Cumulative radicle emergence curves of (red) non-treated, (blue) after 2 min streamer corona plasma treatment and (green) after 10 min dielectric barrier discharge seed. Error bars denote the confidence intervals error bars (n = 4; p < 0.05); missing error bars indicate that they are smaller than the symbols
Evaluation of the Post-discharge Reaction on Rice Seed Surface Morphology and Physicochemical Alteration
The overall result suggests that both electrical discharge plasma, SCP and DBD, can enhance rice seed vigor via surface modification and functionalization. Thus, to gain deeper insight into their interactions and mechanisms on the seed surface, SEM was employed to examine rice seed morphology after plasma applications. Figure 5a demonstrates the natural rice seed morphology (before the plasma process), showing small needle-like features on the entire seed surface with a thin seed coat covering the seed (Fig. 5b). It was observed that after plasma treatments, some needle-like structures were damaged or broken (Fig. 5c and e) during the nanoscale plasma etching process [32]. Furthermore, both plasma-treated seeds had smoother surfaces (Fig. 5d and f) than the non-treated seeds (Fig. 5b) and had a cleaner surface without epicuticular wax that decrease surface wetting and moisture loss of rice seeds. Seed surface erosion presented in the SEM image indicates that the seeds interacted with active plasma species, the electric field, oxygen radicals and, bombarded ions during treatment, resulting in modifications of the seed coat. Figure 5g demonstrates the overall physicochemical interaction of plasma on the seed surface, which increases hydrophilicity and water permeability, both of which are crucial factors for seed germination [51]. Electrical discharge plasma-induced seed surface modification involves several physical and chemical processes. For example, a built-in high electric field, 105 V/cm in the electrical charge plasma system [31], can enhance the high energy electron injection and consequently impact the ionization process [16, 31], leading to several RONS and ion generation depending upon the operational gas ambient conditions [5, 21, 37]. Furthermore, the bombardment of highly energetic ions and particles plays an essential role in morphological alterations via surface etching [37] which causes ruptures of the surface membrane, potentially facilitating water absorption. Several RONS and active ions, such as NO, N2O, ·OH, O3 and H2O2, are responsible for surface functionalization, for example, by increasing polar groups such as carbonyl, carboxyl, hydroxyl, and amino groups on the seed surface [21, 52]. Furthermore, it has been shown that lipid peroxidation following plasma therapy might cause oxidative damage to the membrane bilayer [53, 54] resulting in enhanced water permeability and thus enhanced imbibition.
Rice seed surface morphology and physicochemical interaction of plasma. SEM images of the rice seed surface a, b before plasma treatment, c, d after DBD plasma treatment and e, f after SCP plasma treatment. The scale bars on the left and right images represent 500 µm and 100 μm, respectively. g The overall physicochemical interaction of plasma on the seed surface increases the hydrophilicity and water permeability, which are essential factors for seed germination. h The primary nutritional content in the seeds after plasma processes. The vertical bars represent the standard error of the means
In terms of seed vigor enhancement, RONS can infiltrate seeds, which promotes physiological responses [18, 55]. Typically, the seed coat characteristics and its permeability to water and oxygen are involved in the germination process [56]. Thus, enhanced seed imbibition can resume the metabolism of quiescent seeds and stimulate embryo rehydration and metabolism activation [18, 51]. Furthermore, the germination of a seed involves a complex network of plant hormones such as abscisic acid (ABA), gibberellic acid (GA), brassinosteroids, ethylene, and auxin [21, 57]. Specifically, ABA and GA play an important role in the process, ABA inhibits seed germination during embryo maturation, while GA enhances the germination process [21, 57]. Direct plasma treatment of seeds in ambient air allows the seed to be exposed to ions and the electric field and a mixture of short-lived and long-lived RONS (e.g., NO, NO2–, NO3–, O, OH, O3, H2O2) (Fig. 5g). Therefore, by modifying cell wall polysaccharides, RONS can increase germination by weakening the seed coat. H2O2 and NOx may affect ABA levels, limiting dormancy while promoting GA biosynthesis for germination. [21, 37, 57, 58]. Additionally, by modulating NO levels through NO2−, NO3− in seeds ABA levels are reduced while GA levels are increased [21, 58]. As a result, seed vigor and germination processes are improved, even in a large-scale SCP system that simultaneously rendered 10,000–20,000 rice seeds. Although plasma treatment can enhance seed vigor and surface modification, it raises an obvious concern about the primary nutritional content in the seeds. Thus, seed nutritional contents were investigated after plasma treatments. Figure 5h shows that the carbohydrate, fat, and protein contents of plasma-treated seeds, as well as the seed moisture content, were not significantly different from those of non-treated seeds. The results suggest that plasma can improve seed vigor primarily through surface functionalization but has no adverse effects on seed nutrition.
Conclusions
In conclusion, this research has shown that large-scale electrical discharge plasma treatments (SCP and DBD) can significantly enhance the vigor of rice seeds and the growth parameters of seedlings compared with untreated seeds. The morphological change of the rice seed surface demonstrated the effectiveness of plasma interaction on the seed surface, resulting in improved water uptake and the permeability of the seeds and thus triggering the initiation of germination and eventually becoming an essential factor affecting fast radicle emergence (RE) and germination parameters. Plasma-treated seeds required ~ 13% less RE time than non-treated seeds at 55 h. after the set time, while the final germination percentage of plasma-treated and non-treated seeds was not significantly different. The overall results indicate that both electrical discharge plasma treatments do not harm seeds and have become potential alternative seed enhancement methods in seed technology.
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Abbreviations
- ABA:
-
Abscisic acid
- AC:
-
Alternating current
- ACA:
-
Apparent contact angle
- ANOVA:
-
Analysis of variance
- ANTP:
-
Atmospheric nonthermal plasma
- DBD:
-
Dielectric barrier discharge
- GA:
-
Gibberellic acid
- MRET:
-
Mean radicle emergence time
- OES:
-
Optical emission spectroscopy
- RONS:
-
Reactive oxygen and nitrogen species
- SCP:
-
Streamer corona plasma
- SEM:
-
Scanning electron microscope
- tRE :
-
Radicle emergence time
- t50RE :
-
The time required for 50% of viable seeds to emerge
References
Alexandratos N, Bruinsma J (2012) World agriculture towards 2030/2050: the 2012 revision In: FAO Agricultural Development Economics Division (ed) Agricultural Development Economics (ESA) Working Paper No. 12–03, Food and Agriculture Organization of the United Nations, Rome, Italy
International Rice Research Institute (IRRI) (2019) Rice to zero hunger, Los Baños, Laguna, Philippines. https://www.irri.org/world-food-day-2019-rice-zero-hunger
Basra SMA, Farooq M, Tabassam R, Ahmad N (2005) Physiological and biochemical aspects of pre-sowing seed treatments in fine rice (Oryza sativa L.). Seed Sci Technol 33:623–628. https://doi.org/10.15258/sst.2005.33.3.09
Finch-Savage WE, Bassel GW (2016) Seed vigour and crop establishment: extending performance beyond adaptation. J Exp Bot 67:567–591. https://doi.org/10.1093/jxb/erv490
Misra N, Yadav B, Roopesh M, Jo C (2019) Cold plasma for effective fungal and mycotoxin control in foods: mechanisms, inactivation effects, and applications. Compr Rev Food Sci Food Saf 18:106–120. https://doi.org/10.1111/1541-4337.12398
Matthews S, Noli E, Demir I, Khajeh-Hosseini M, Wagner MH (2012) Evaluation of seed quality: from physiology to international standardization. Seed Sci Res 22:S69–S73. https://doi.org/10.1017/S0960258511000365
Powell AA (2006) Seed vigour and its assessment. In: Basra AS (ed) Handbook of Seed Science and Technology. Food Products Press, Binghamton, USA
Onwimol D, Chanmprasert W, Changsee P, Rongsangchaichareon T (2016) Seed vigor classification using analysis of mean radicle emergence time and single counts of radicle emergence in rice (Oryza sativa L.) and mung bean (Vigna radiata (L.) Wilczek). Agric. Nat. Resour. 50:345–350. https://doi.org/10.1016/j.anres.2016.12.003
International Seed Testing Association (ISTA) (2018) International Rules for Seed Testing. Bassersdorf, Switzerland
Zehra A, Shaikh F, Ansari R, Gul B, Khan M (2013) Effect of ascorbic acid on seed germination of three halophytic grass species under saline conditions. Grass Forage Sci 68:339–344. https://doi.org/10.1111/j.1365-2494.2012.00899.x
Guangwu Z, Xuwen J (2014) Roles of gibberellin and auxin in promoting seed germination and seedling vigor in Pinus massoniana. For Sci 60:367–373. https://doi.org/10.5849/forsci.12-143
Goussous S, Samarah N, Alqudah A, Othman M (2010) Enhancing seed germination of four crop species using an ultrasonic technique. Exp Agric 46:231. https://doi.org/10.1017/S0014479709991062
Xu G, Wang X-T, Gan C-L, Fang Y-Q, Zhang M (2012) Biological effects of low energy nitrogen ion implantation on Jatropha curcas L. seed germination. Nucl Instrum Methods Phys Res B 287:76–84. https://doi.org/10.1016/j.nimb.2012.05.038
Ahmed Z, Manzoor MF, Ahmad N, Zeng XA, Din ZU, Roobab U, Qayum A, Siddique R, Siddeeg A, Rahaman A (2020) Impact of pulsed electric field treatments on the growth parameters of wheat seeds and nutritional properties of their wheat plantlets juice. Food Sci Nutr 8:2490–2500. https://doi.org/10.1002/fsn3.1540
Volin JC, Denes FS, Young RA, Park SM (2000) Modification of seed germination performance through cold plasma chemistry technology. Crop Sci 40:1706–1718. https://doi.org/10.2135/cropsci2000.4061706x
Bogaerts A, Neyts E, Gijbels R, Van der Mullen J (2002) Gas discharge plasmas and their applications. Spectrochim Acta, B 57:609–658. https://doi.org/10.1016/S0584-8547(01)00406-2
Chen HH (2014) Investigation of properties of long-grain brown rice treated by low-pressure plasma. Food Bioprocess Technol 7:2484–2491. https://doi.org/10.1007/s11947-013-1217-2
Graves DB (2012) The emerging role of reactive oxygen and nitrogen species in redox biology and some implications for plasma applications to medicine and biology. J Phys D Appl Phys 45:263001. https://doi.org/10.1088/0022-3727/45/26/263001
Sakudo A, Yagyu Y, Onodera T (2019) Disinfection and sterilization using plasma technology: fundamentals and future perspectives for biological applications. Int J Mol Sci 20:5216. https://doi.org/10.3390/ijms20205216
Nishioka T, Takai Y, Kawaradani M, Okada K, Tanimoto H, Misawa T, Kusakari S (2014) Seed disinfection effect of atmospheric pressure plasma and low pressure plasma on Rhizoctonia solani. Biocontrol Sci 19:99–102. https://doi.org/10.4265/bio.19.99
Ranieri P, Sponsel N, Kizer J, Rojas-Pierce M, Hernández R, Gatiboni L, Grunden A, Stapelmann K (2021) Plasma agriculture: review from the perspective of the plant and its ecosystem. Plasma Process Polym 18:2000162. https://doi.org/10.1002/ppap.202000162
Shi XM, Liao WL, Chang ZS, Zhang GJ, Wu XL, Dong XF, Yao CW, Ye BY, Li P, Xu GM, Chen SL, Cai JF (2015) Inactivation effect of low-temperature plasma on Pseudomonas aeruginosa for nosocomial anti-infection. IEEE Trans Plasma Sci 43:3211–3218. https://doi.org/10.1109/TPS.2015.2465158
Bormashenko E, Grynyov R, Bormashenko Y, Drori E (2012) Cold radiofrequency plasma treatment modifies wettability and germination speed of plant seeds. Sci Rep 2:741. https://doi.org/10.1038/srep00741
Guo Q, Meng Y, Qu G, Wang T, Yang F, Liang D, Hu S (2018) Improvement of wheat seed vitality by dielectric barrier discharge plasma treatment. Bioelectromagnetics 39:120–131. https://doi.org/10.1002/bem.22088
Ling L, Jiafeng J, Jiangang L, Minchong S, Xin H, Hanliang S, Yuanhua D (2014) Effects of cold plasma treatment on seed germination and seedling growth of soybean. Sci Rep 4:1–7. https://doi.org/10.1038/srep05859
Hashizume H, Kitano H, Mizuno H, Abe A, Yuasa G, Tohno S, Tanaka H, Ishikawa K, Matsumoto S, Sakakibara H (2021) Improvement of yield and grain quality by periodic cold plasma treatment with rice plants in a paddy field. Plasma Process Polym 18:2000181. https://doi.org/10.1002/ppap.202000181
Khamsen N, Onwimol D, Teerakawanich N, Dechanupaprittha S, Kanokbannakorn W, Hongesombut K, Srisonphan S (2016) Rice (Oryza sativa L.) seed sterilization and germination enhancement via atmospheric hybrid nonthermal discharge plasma. ACS Appl Materi Interfaces 8:19268–19275. https://doi.org/10.1021/acsami.6b04555
Ruangwong K, Rongsangchaicharean T, Thammaniphit C, Onwimol D, Srisonphan S (2020) Atmospheric corona discharge plasma for rice (Oryza sativa L.) seed surface modification fungi decontamination and shelf life extension. Plasma Med. https://doi.org/10.1615/PlasmaMed.2021036474
Sadhu S, Thirumdas R, Deshmukh RR, Annapure US (2017) Influence of cold plasma on the enzymatic activity in germinating mung beans (Vigna radiate). LWT 78:97–104. https://doi.org/10.1016/j.lwt.2016.12.026
Bentsink L, Koornneef M (2008) Seed dormancy and germination. The Arabidopsis book. American Society of Plant Biologists, Rockville, p 6
Chang J-S, Lawless PA, Yamamoto T (1991) Corona discharge processes. IEEE Trans Plasma Sci 19:1152–1166. https://doi.org/10.1109/27.125038
Srisonphan S (2018) Tuning surface wettability through hot carrier initiated impact ionization in cold plasma. ACS Appl Mater Interfaces 10:11297–11304. https://doi.org/10.1021/acsami.7b19495
Ikmal Misnal MF, Redzuan N, Firdaus Zainal MN, Raja Ibrahim RK, Ahmad N, Agun L (2021) Emerging cold plasma treatment on rice grains: a mini review. Chemosphere 274:129972. https://doi.org/10.1016/j.chemosphere.2021.129972
Kasap SO (2006) Principles of electronic materials and devices. McGraw-Hill, London
Kong D, He F, Yang B, Duan Z, Han R, Miao J, Yan X, Ouyang J (2021) Multiple current pulse behavior and its dynamics of atmospheric pressure plasma jet in a needle-to-ring configuration. J Phys D Appl Phys 54:405201. https://doi.org/10.1088/1361-6463/ac08cb
Teerakawanich N, Kasemsuwan V, Jitkajornwanich K, Kanokbannakorn W, Srisonphan S (2018) Microcorona discharge-mediated nonthermal atmospheric plasma for seed surface modification. Plasma Chem Plasma Process 38:817–830. https://doi.org/10.1007/s11090-018-9894-2
Laroussi M (2005) Low temperature plasma-based sterilization: overview and state-of-the-art. Plasma Processes Polym 2:391–400. https://doi.org/10.1002/ppap.200400078
Kogelschatz U (2003) Dielectric-barrier discharges: their history, discharge physics, and industrial applications. Plasma Chem Plasma Process 23:1–46. https://doi.org/10.1023/A:1022470901385
Srisonphan S, Ruangwong K, Thammaniphit C (2020) Localized electric field enhanced streamer cold plasma interaction on biological curved surfaces and its shadow effect. Plasma Chem Plasma Process 40:1253–1265. https://doi.org/10.1007/s11090-020-10098-8
Fritz JL, Owen MJ (1995) Hydrophobic recovery of plasma-treated polydimethylsiloxane. J Adhes 54:33–45. https://doi.org/10.1080/00218469508014379
Sugiarto AT, Sato M (2001) Pulsed plasma processing of organic compounds in aqueous solution. Thin Solid Films 386:295–299. https://doi.org/10.1016/S0040-6090(00)01669-2
Schmid S, Jecklin MC, Zenobi R (2010) Degradation of volatile organic compounds in a non-thermal plasma air purifier. Chemosphere 79:124–130. https://doi.org/10.1016/j.chemosphere.2010.01.049
Encinas N, Díaz-Benito B, Abenojar J, Martínez M (2010) Extreme durability of wettability changes on polyolefin surfaces by atmospheric pressure plasma torch. Surf Coat Technol 205:396–402. https://doi.org/10.1016/j.surfcoat.2010.06.069
Waterworth WM, Bray CM, West CE (2019) Seeds and the art of genome maintenance. Front Plant Sci. https://doi.org/10.3389/fpls.2019.00706
Powell A, Matthews S (2012) Seed aging/repair hypothesis leads to new testing methods. Seed Technol 34:15–25
Joosen RV, Kodde J, Willems LA, Ligterink W, van der Plas LH, Hilhorst HW (2010) GERMINATOR: a software package for high-throughput scoring and curve fitting of Arabidopsis seed germination. Plant J 62:148–159. https://doi.org/10.1111/j.1365-313X.2009.04116.x
AOAC (1993) Official methods of analysis, 14th edn. Association of Official Analytical Chemist, USA
Aoac G (2016) Official methods of analysis of AOAC International. AOAC International, Rockville, MD
R Core Team (2020) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Luo Y, Lin C, Huang YT, Guan YJ, Hu J (2017) Single counts of radicle emergence can be used as a fast method to test seed vigour of indica rice. Seed Sci Technol 45:222–229. https://doi.org/10.15258/sst.2017.45.1.19
Abebe AT, Modi A (2009) Hydro-priming in dry bean (Phaseolus vulgaris L.). Res J Seed Sci 2:23–31. https://doi.org/10.3923/rjss.2009.23.31
Shi T, Shao M, Zhang H, Yang Q, Shen X (2011) Surface modification of porous poly (tetrafluoroethylene) film via cold plasma treatment. Appl Surf Sci 258:1474–1479. https://doi.org/10.1016/j.apsusc.2011.09.110
Gaunt LF, Beggs CB, Georghiou GE (2006) Bactericidal action of the reactive species produced by gas-discharge nonthermal plasma at atmospheric pressure: a review. IEEE Trans Plasma Sci 34:1257–1269. https://doi.org/10.1109/TPS.2006.878381
Arjunan KP, Sharma VK, Ptasinska S (2015) Effects of atmospheric pressure plasmas on isolated and cellular DNA—a review. Int J Mol Sci 16:2971–3016. https://doi.org/10.3390/ijms16022971
Živković S, Puač N, Giba Z, Grubišić D, Petrović ZL (2004) The stimulatory effect of non-equilibrium (low temperature) air plasma pretreatment on light-induced germination of Paulownia tomentosa seeds. Seed Sci Technol 32:693–701. https://doi.org/10.15258/sst.2004.32.3.05
Randeniya LK, de Groot GJ (2015) Non-thermal plasma treatment of agricultural seeds for stimulation of germination, removal of surface contamination and other benefits: a review. Plasma Process Polym 12:608–623. https://doi.org/10.1002/ppap.201500042
Bailly C (2019) The signalling role of ROS in the regulation of seed germination and dormancy. Biochem J 476:3019–3032. https://doi.org/10.1042/BCJ20190159
Bethke PC, Libourel IG, Aoyama N, Chung YY, Still DW, Jones RL (2007) The Arabidopsis aleurone layer responds to nitric oxide, gibberellin, and abscisic acid and is sufficient and necessary for seed dormancy. Plant Physiol 143:1173–1188. https://doi.org/10.1104/pp.106.093435
Acknowledgements
The Rice Department, Ministry of Agriculture and Cooperatives (Thailand) kindly provided the rice seed samples used in this research.
Funding
This project is funded by National Research Council of Thailand (NRCT) and Kasetsart University: N42A650281 and partially supported by the Kasetsart University Research and Development Institute (KURDI). S. Srisonphan acknowledges the funding support from the NSRF via the Program Management Unit for Human Resources & Institutional Development, Research and Innovation [grant number B05F640160].
Author information
Authors and Affiliations
Contributions
TR and SS were involved in the conceptualization, methodology, investigation, and writing of the original draft. DO was involved in the conceptualization, methodology, review, writing and editing. TR and SS contributed equally to this work.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that may have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rongsangchaicharean, T., Srisonphan, S. & Onwimol, D. Responses of Rice Seed Quality to Large-Scale Atmospheric Nonthermal Plasmas. Plasma Chem Plasma Process 42, 1127–1141 (2022). https://doi.org/10.1007/s11090-022-10261-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11090-022-10261-3