Abstract
Herein, we demonstrate that the performance of mesoporous silica SBA-15 and SBA-15-Ag during plasma ammonia synthesis depends on the plasma conditions. At high power, the mesoporous silica SBA-15 without Ag produces the largest amount of ammonia, but the addition of Ag provides a minor benefit at lower powers. Plasma conditions were analyzed through optical emission spectroscopy using N2, N2+, and NH molecular bands and Hα line. Stark broadening of Hα line was used to find electron density, and N2 molecular bands were used to assess N2 vibrational excitation, important for plasma nitrogen decomposition. At similar input conditions, reactors with SBA-15 have higher electron density and higher N2 vibrational temperature. Consistent with higher electron density, SBA-15 reactors have stronger N2+ emission intensity relative to the neutral N2. The addition of Ag results in higher N2 rotational temperature, possibly due to localized heating. From the materials point of view, SBA-15 is a more robust catalyst with good surface area retention after plasma exposure due to the lack of local heating generated when a metal is in the structure. We identify two possible regimes during ammonia synthesis, a metal and a surface-plasma driven. At lower plasma densities, the addition of metal is beneficial, while at higher power and plasma density, the best performance is achieved without the aid of a metal catalyst.
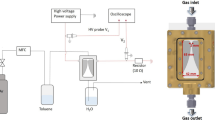
Graphical Abstract
Mesoporous materials for Plasma Catalytic Ammonia Synthesis, at certain plasma conditions lead to different regimes, a plasma/surface and a metal dominated regimes of ammonia production.













Similar content being viewed by others
References
Reiter AJ, Kong S-C (2011) Combustion and emissions characteristics of compression-ignition engine using dual ammonia-diesel fuel. Fuel 90(1):87–97
Carreon ML (2019) Plasma catalytic ammonia synthesis: state of the art and future directions. J Phys D Appl Phys 52(48):483001
Degnan T (2018) New catalytic developments may promote development of smaller scale ammonia plants. Focus Catalysts 1
Klerke A, Christensen CH, Nørskov JK, Vegge T (2008) Ammonia for hydrogen storage: challenges and opportunities. J Mater Chem 18(20):2304–2310
Rees NV, Compton RG (2011) Carbon-free energy: a review of ammonia-and hydrazine-based electrochemical fuel cells. Energy Environ Sci 4(4):1255–1260
Wang W, Herreros JM, Tsolakis A, York APE (2013) Ammonia as hydrogen carrier for transportation; investigation of the ammonia exhaust gas fuel reforming. Int J Hydrogen Energy 38(23):9907–9917
Zamfirescu C, Dincer I (2009) Ammonia as a green fuel and hydrogen source for vehicular applications. Fuel Process Technol 90(5):729–737
Lan R, Irvine JTS, Tao S (2012) Ammonia and related chemicals as potential indirect hydrogen storage materials. Int J Hydrogen Energy 37(2):1482–1494
Cortright RD, Davda R, Dumesic JA (2002) Hydrogen from catalytic reforming of biomass-derived hydrocarbons in liquid water. Nature 418(6901):964–967
Christensen CH, Johannessen T, Sørensen RZ, Nørskov JK (2006) Towards an ammonia-mediated hydrogen economy? Catal Today 111(1–2):140–144
Erisman JW, Sutton MA, Galloway J, Klimont Z, Winiwarter W (2008) How a century of ammonia synthesis changed the world. Nat Geosci 1(10):636–639
Schlögl R (2008) Ammonia synthesis, Handbook of heterogeneous catalysis. Wiley-VCH2008, pp. 2501–2575
Patil BS (2017) Plasma (catalyst)-assisted nitrogen fixation: reactor development for nitric oxide and ammonia production. Technische Universiteit Eindhoven, Eindhoven, pp 1–223
Tanabe Y, Nishibayashi Y (2013) Developing more sustainable processes for ammonia synthesis. Coord Chem Rev 257(17):2551–2564
http://energy.globaldata.com/media-center/press-releases/oil-and-gas/, global-ammonia-capacity-to-reach-almost-250-million-tons-per-year-by-2018-says-globaldata. 2018.
Hong J, Prawer S, Murphy AB (2018) Plasma catalysis as an alternative route for ammonia production: status, mechanisms, and prospects for progress. ACS Sustain Chem Eng 6(1):15–31
Hess C, Lercher J, Naraschewski F, Kondratenko E, Baerns M, Trunschke A, Grasselli R (2011) Nanostructured catalysts: selective oxidations, Royal Society of Chemistry
Shah J, Gorky F, Psarras P, Seong B, Gómez-Gualdrón DA, Carreon ML (2020) Enhancement of the yield of ammonia by hydrogen-sink effect during plasma catalysis. ChemCatChem 12(4):1200–1211
Norby T, Widerøe M, Glöckner R, Larring Y (2004) Hydrogen in oxides. Dalton Trans 19:3012–3018
Efimchenko VS, Fedotov VK, Kuzovnikov MA, Zhuravlev AS, Bulychev BM (2013) Hydrogen solubility in amorphous silica at pressures up to 75 kbar. J Phys Chem B 117(1):422–425
Hashizume K, Ogata K, Nishikawa M, Tanabe T, Abe S, Akamaru S, Hatano Y (2013) Study on kinetics of hydrogen dissolution and hydrogen solubility in oxides using imaging plate technique. J Nucl Mater 442(1–3):S880–S884
Meletov KP, Efimchenko VS (2021) Raman study of hydrogen-saturated silica glass. Int J Hydrogen Energy
Peng P, Chen P, Addy M, Cheng Y, Anderson E, Zhou N, Schiappacasse C, Zhang Y, Chen D, Hatzenbeller R, Liu Y, Ruan R (2019) Atmospheric plasma-assisted ammonia synthesis enhanced via synergistic catalytic absorption. ACS Sustain Chem Eng 7(1):100–104
Aika K (2017) Role of alkali promoter in ammonia synthesis over ruthenium catalysts—effect on reaction mechanism. Catal Today 286:14–20
Sugiyama K, Kiyoshi A, Masaaki O, Hiroshi M, Tsuneo M, N.O. (1986) Ammonia synthesis by means of plasma over MgO catalyst. Plasma Chem Plasma Process 6(2):179–193
Ozaki A (1981) Development of alkali-promoted ruthenium as a novel catalyst for ammonia synthesis. Acc Chem Res 14(1):16–21
Reddy RR, Ahammed YN, Gopal KR, Raghuram DV (1998) Optical electronegativity and refractive index of materials. Opt Mater 10(2):95–100
Suttikul T, Sreethawong T, Sekiguchi H, Chavadej S (2011) Ethylene epoxidation over alumina-and silica-supported silver catalysts in low-temperature ac dielectric barrier discharge. Plasma Chem Plasma Process 31(2):273–290
Wannagat U, The Silicon-Nitrogen Bond (1978) Biochemistry of Silicon and related Problems, Springer, Boston, pp 77–90
James BD, DeSantis DA, Saur G, Hydrogen Production Pathways Cost Analysis (2013–2016), 2016, DOE-StrategicAnalysis-6231-1., Strategic Analysis Inc., Arlington, VA (United States)
Taguchi A, Schüth F (2005) Ordered mesoporous materials in catalysis. Microporous Mesoporous Mater 77(1):1–45
Rao Y, Antonelli DM (2009) Mesoporous transition metal oxides: characterization and applications in heterogeneous catalysis. J Mater Chem 19(14):1937–1944
Beck JS (1992) A new family of mesoporous molecular sieves prepared with liquid crystal templates. J Am Chem Soc 114:10834–10843
Kresge CT, Leonowicz ME, Roth WJ, Vartuli JC, Beck JS (1992) Ordered mesoporous molecular sieves synthesized by a liquid-crystal template mechanism. Nature 359:710–712
Carreon MA, Guliants VV (2005) Ordered meso-and macroporous binary and mixed metal oxides. Eur J Inorg Chem 2005:27–43
Shah J, Wu T, Lucero J, Carreon MA, Carreon ML (2018) Nonthermal plasma synthesis of ammonia over Ni-MOF-74. ACS Sustain Chem Eng 7(1):377–383
Shah JR, Gorky F, Lucero J, Carreon MA, Carreon ML (2020) Ammonia synthesis via atmospheric plasma catalysis: zeolite 5A, a case of study. Ind Eng Chem Res 59(11):5167–5176
Gorky F, Carreon MA, Carreon ML (2020) Experimental strategies to increase ammonia yield in plasma catalysis over LTA and BEA zeolites. IOP SciNotes 1(2):024801
Gorky F, Guthrie SR, Smoljan CS, Crawford JM, Carreon MA, Carreon ML (2021) Plasma ammonia synthesis over mesoporous silica SBA-15. J Phys D 54(26):264003
vanʻt Veer K, Reniers F, Bogaerts A (2020) Zero-dimensional modeling of unpacked and packed bed dielectric barrier discharges: the role of vibrational kinetics in ammonia synthesis. Plasma Sources Sci Technol 29(4):045020
Brandenburg R (2017) Dielectric barrier discharges: progress on plasma sources and on the understanding of regimes and single filaments. Plasma Sources Sci Technol 26(5):053001
Gao M, Zhang Y, Wang H, Guo B, Zhang Q, Bogaerts A (2018) Mode transition of filaments in packed-bed dielectric barrier discharges. Catalysts 8(6):248
Hala A (2018) Plasma dynamics in a packed bed dielectric barrier discharge (DBD) operated in helium. J Phys D Appl Phys 51(11):11LT02
Kruszelnicki J, Hala A, Kushner MJ (2020) Formation of surface ionization waves in a plasma enhanced packed bed reactor for catalysis applications. Chem Eng J 382:123038
Engeling KW, Kruszelnicki J, Kushner MJ, Foster JE (2018) Time-resolved evolution of micro-discharges, surface ionization waves and plasma propagation in a two-dimensional packed bed reactor. Plasma Sources Sci Technol 27(8):085002
Cheng H, Ma M, Zhang Y, Liu D, Lu X (2020) The plasma enhanced surface reactions in a packed bed dielectric barrier discharge reactor. J Phys D Appl Phys 53(14):144001
Li Y, Yang D-Z, Qiao J-J, Zhang L, Wang W-Z, Zhao Z-L, Zhou X-F, Yuan H, Wang W-C (2020) The dynamic evolution and interaction with dielectric material of the discharge in packed bed reactor. Plasma Sources Sci Technol 29(5):055004
Shah J, Wang W, Bogaerts A, Carreon ML (2018) Ammonia synthesis by radio frequency plasma catalysis: revealing the underlying mechanisms. ACS Appl Energy Mater 1(9):4824–4839
Dongyuan Z, Sun J, Li Q, Stucky GD (2000) Morphological control of highly ordered mesoporous silica SBA-15. Chem Mater 12:275–279
Crawford JM, Anderson R, Gasvoda RJ, Kovach NC, Smoljan CS, Jasinski JB, Trewyn BG, Agarwal S, Gómez-Gualdrón DA, Carreon MA (2020) Vacancy healing as a desorption tool: oxygen triggered removal of stored ammonia from NiO1–x/MOR validated by experiments and simulations. ACS Appl Energy Mater 3(9):8233–8239
Gorky F, Lucero JM, Crawford JM, Blake B, Carreon MA, Carreon ML (2021) Plasma-induced catalytic conversion of nitrogen and hydrogen to ammonia over zeolitic imidazolate frameworks ZIF-8 and ZIF-67. ACS Appl Mater Interfaces 13(18):21338–21348
Hong T, De León F (2015) Lissajous curve methods for the identification of nonlinear circuits: calculation of a physical consistent reactive power. IEEE Trans Circuits Syst I Regul Pap 62(12):2874–2885
Peeters FJJ, Van de Sanden MCM (2014) The influence of partial surface discharging on the electrical characterization of DBDs. Plasma Sources Sci Technol 24(1):015016
Gordon IE, Rothman LS, Hargreaves RJ, Hashemi R, Karlovets EV, Skinner FM, Conway EK, Hill C, Kochanov RV, Tan Y (2021) The HITRAN2020 molecular spectroscopic database. J Quant Spectroscopy Radiat Transf 107949
LCO, Optical Diagnostics and Collisional-Radiative Models, June 30 – July 3, 2008, VKI Course on Hypersonic Entry and Cruise Vehicles, Stanford University.
Pearse RWB, Gaydon AG (1976) The identification of molecular spectra. Chapman and Hall, London
Belostotskiy SG, Ouk T, Donnelly VM, Economou DJ, Sadeghi N (2010) Gas temperature and electron density profiles in an argon dc microdischarge measured by optical emission spectroscopy. J Appl Phys 107(5):053305
Mijatović Z, Djurović S, Gavanski L, Gajo T, Favre A, Morel V, Bultel A (2020) Plasma density determination by using hydrogen Balmer Hα spectral line with improved accuracy. Spectrochim Acta Part B Atom Spectrosc 166:105–821
Gigosos MA, Gonzalez MA, Cardenoso V (2003) Computer simulated Balmer-alpha,-beta and-gamma Stark line profiles for non-equilibrium plasmas diagnostics. Spectrochim Acta, Part B 58(8):1489–1504
Gigosos MA, Cardeñoso V (1996) New plasma diagnosis tables of hydrogen Stark broadening including ion dynamics. J Phys B: At Mol Opt Phys 29(20):4795
Van der Horst RM, Verreycken T, Van Veldhuizen EM, Bruggeman PJ (2012) Time-resolved optical emission spectroscopy of nanosecond pulsed discharges in atmospheric-pressure N2 and N2/H2O mixtures. J Phys D Appl Phys 45(34):345201
Kepple P, Griem HR (1968) Improved Stark profile calculations for the hydrogen lines Hα, Hβ, Hγ, and Hδ. Phys Rev 173(1):317
Griem HR (1966) Plasma spectroscopy. Mc Graw‐Hill, New York (1964), Spectral Line Broadening by Plasmas
Drake GWF (2006) Atomic, molecular and optical physics. Springer, New York
Iwamoto M, Akiyama M, Aihara K, Deguchi T (2017) Ammonia synthesis on wool-like Au, Pt, Pd, Ag, or Cu electrode catalysts in nonthermal atmospheric-pressure plasma of N2 and H2. ACS Catal 7(10):6924–6929
Liu T-W, Gorky F, Carreon ML, Gómez-Gualdrón DA (2022) Energetics of reaction pathways enabled by N and H radicals during catalytic, plasma-assisted NH3 synthesis. ACS Sustain Chem Eng
Patil BS, Cherkasov N, Srinath NV, Lang J, Ibhadon AO, Wang Q, Hessel V (2021) The role of heterogeneous catalysts in the plasma-catalytic ammonia synthesis. Catal Today 362:2–10
Barboun P, Mehta P, Herrera FA, Go DB, Schneider WF, Hicks JC (2019) Distinguishing plasma contributions to catalyst performance in plasma-assisted ammonia synthesis. ACS Sustain Chem Eng 7(9):8621–8630
Pipa AV, Brandenburg R (2019) The equivalent circuit approach for the electrical diagnostics of dielectric barrier discharges: the classical theory and recent developments. Atoms 7(1):14
Baskaran S, Liu J, Domansky K, Kohler N, Li X, Coyle C, Fryxell GE, Thevuthasan S, Williford RE (2000) Low dielectric constant mesoporous silica films through molecularly templated synthesis. Adv Mater 12(4):291–294
Wang Y, Craven M, Yu X, Ding J, Bryant P, Huang J, Tu X (2019) Plasma-Enhanced catalytic synthesis of ammonia over a Ni/Al2O3 catalyst at near-room temperature: insights into the importance of the catalyst surface on the reaction mechanism. ACS Catal. https://doi.org/10.1021/acscatal.9b02538)
Gorky F, Best A, Jasinski J, Allen BJ, Alba-Rubio AC, Carreon ML (2021) Plasma catalytic ammonia synthesis on Ni nanoparticles: the size effect. J Catal 393:369–380
Wu D, Yao L, Ricci M, Li J, Xie R, Peng Z (2021) Ambient synthesis of pt-reactive metal alloy and high-entropy alloy nanocatalysts utilizing hydrogen cold plasma. Chem Mater
Lu X, Naidis GV, Laroussi M, Reuter S, Graves DB, Ostrikov K (2016) Reactive species in non-equilibrium atmospheric-pressure plasmas: generation, transport, and biological effects. Phys Rep 630:1–84
Neyts EC, Ostrikov K, Sunkara MK, Bogaerts A (2015) Plasma catalysis: synergistic effects at the nanoscale. Chem Rev 115(24):13408–13446
Mehta P, Barboun P, Herrera FA, Kim J, Rumbach P, Go DB, Hicks JC, Schneider WF (2018) Overcoming ammonia synthesis scaling relations with plasma-enabled. Catalysis 1(4):269–275
Acknowledgements
The work by SG and HF was supported by the Princeton Collaborative Research Facility (PCRF), which is supported by the U.S. Department of Energy (DOE) under Contract No. DE-AC02-09CH11466. The work by MLC and FG was supported by DOE, Office of Science Fusion Energy Sciences Award No. DE-SC0021309. All plasma diagnostic resources used in this work were provided by the PCRF. MLC wants to thank Dr. Jacek Jasinski for his help with the collected TEM images and insightful discussion about the same.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gershman, S., Fetsch, H., Gorky, F. et al. Identifying Regimes During Plasma Catalytic Ammonia Synthesis. Plasma Chem Plasma Process 42, 731–757 (2022). https://doi.org/10.1007/s11090-022-10258-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11090-022-10258-y