Abstract
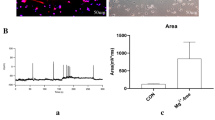
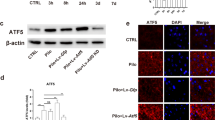
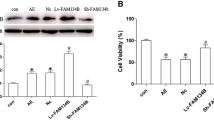
The mitochondrial unfolded protein response (mtUPR) has been shown to restore protein homeostasis and cell function under stress, and recent studies have confirmed that the activating transcription factor 4 (ATF4) regulates mtUPR. However, the role of ATF4-mediated mtUPR in a hippocampal neuronal culture model of seizures remains unclear. Our results showed that the expression of mtUPR-related proteins (HSP60 and CLpP) increased in primary hippocampal neurons with seizures induced by a magnesium-free solution, suggesting mtUPR activation. Furthermore, ATF4 overexpression by lentiviral vector transfection enhanced the expression of HSP60 and CLpP, whereas ATF4 low expression by lentiviral vector transfection weakened the expression of HSP60 and CLpP. In addition, ATF4 overexpression increased neuronal viability and reduced seizure-induced apoptosis. ATF4 overexpression reduced reactive oxygen species (ROS) production and improved mitochondrial membrane potential damage during seizures. Moreover, ATF4 overexpression reduced the BCL2-associated X protein (Bax) expression and increased the expression of B-cell lymphoma 2 (BCL2). In contrast, ATF4 expression showed the opposite trend. In conclusion, our results showed that ATF4-mediated mtUPR may delay the cascade activation of apoptotic pathways by reducing ROS-mediated oxidative stress, thereby attenuating seizure-induced stress injury.








Similar content being viewed by others
Data Availability
All data generated by the current study are available upon reasonable request.
Code Availability
Not applicable.
References
Misrani A, Tabassum S, Li Y (2021). Mitochondrial dysfunction and oxidative stress in Alzheimer’s Disease[J].Frontiers in Aging Neuroscience,13
Shamima R (2018) Mitochondrial diseases and status epilepticus[J].Epilepsia,
Lax NZ, Gorman GS, Turnbull DM (2017) Review: Central nervous system involvement in mitochondrial disease. Neuropathol Appl Neurobiol 43(2):102–118. https://doi.org/10.1111/nan.12333
Bindoff LA, Engelsen BA (2012) Mitochondrial diseases and epilepsy. Epilepsia 53(Suppl 4):92–97. https://doi.org/10.1111/j.1528-1167.2012.03618.x
Naresh NU, Haynes CM (2019) Signaling and regulation of the mitochondrial unfolded protein Response[J]. Cold Spring Harbor perspectives in biology
Svagua T, Martini M, Martini M et al (2020) Mitochondrial unfolded protein response, mitophagy and other mitochondrial quality control mechanisms in heart disease and aged heart[J]. Croatian Med J 61(2):126–138
Hu D, Liu Z, Qi X (2021) UPR(mt) activation protects against MPP(+)-induced toxicity in a cell culture model of Parkinson’s disease. Biochem Biophys Res Commun 569:17–22
Melber A, Haynes CM (2018) UPR(mt) regulation and output: a stress response mediated by mitochondrial-nuclear communication. Cell Res 28(3):281–295
Wortel IMN, van der Meer LT, Kilberg MS, van Leeuwen FN (2017) Surviving stress: modulation of ATF4-Mediated stress responses in normal and malignant cells. Trends Endocrinol Metab 28(11):794–806
Quirós PM, Prado MA, Zamboni N, D’Amico D, Williams RW, Finley D, Gygi SP, Auwerx J Multi-omics analysis identifies ATF4 as a key regulator of the mitochondrial stress response in mammals.The Journal of cell biology2017, 216 (7),2027–2045
Nargund AM, Fiorese CJ, Pellegrino MW, Deng P, Haynes CM (2015) Mitochondrial and nuclear accumulation of the transcription factor ATFS-1 promotes OXPHOS recovery during the UPR(mt). Mol Cell 58(1):123–133
Jiang D, Cui H, Xie N, Banerjee S, Liu RM, Dai H, Thannickal VJ, Liu G (2020) ATF4 mediates mitochondrial unfolded protein response in alveolar epithelial cells. Am J Respir Cell Mol Biol 63(4):478–489
Brewer GJ, Torricelli JR, Evege EK, Price PJ (1993) Optimized survival of hippocampal neurons in B27-supplemented Neurobasal, a new serum-free medium combination. J Neurosci Res 35(5):567–576
Sombati S, Delorenzo RJ (1995) Recurrent spontaneous seizure activity in hippocampal neuronal networks in culture. J Neurophysiol 73(4):1706–1711
Mackenzie-Gray Scott C, Parrish RR, Walsh D, Racca C, Cowell RM, Trevelyan AJ (2022) PV-specific loss of the transcriptional coactivator PGC-1α slows down the evolution of epileptic activity in an acute ictogenic model. J Neurophysiol. Jan 1;127(1):86–98. doi: 10.1152/jn.00295.2021. Epub 2021 Nov 17. PMID: 34788174; PMCID: PMC8721902
Rho JM, Boison D (2022) The metabolic basis of epilepsy. Nat Rev Neurol 18(6):333–347. https://doi.org/10.1038/s41582-022-00651-8
Yang N, Guan QW, Chen FH et al (2020) Antioxidants targeting mitochondrial oxidative stress: promising Neuroprotectants for Epilepsy. Oxid Med Cell Longev. 2020:6687185. Published 2020 Nov 25 https://doi.org/10.1155/2020/6687185
Wang X, Yu X, Li Y et al (2022) ATF5 attenuates apoptosis in hippocampal neurons with seizures evoked by Mg2+-Free medium via regulating mitochondrial unfolded protein response [published online ahead of print, 2022 Aug 8. Neurochem Res. https://doi.org/10.1007/s11064-022-03702-0
Jin SM, Youle RJ (2013) The accumulation of misfolded proteins in the mitochondrial matrix is sensed by PINK1 to induce PARK2/Parkin-mediated mitophagy of polarized mitochondria. Autophagy 9(11):1750–1757
Cappello F, Conway de Macario E, Marasà L, Zummo G, Macario AJ (2008) Hsp60 expression, new locations, functions and perspectives for cancer diagnosis and therapy. Cancer Biol Ther 7(6):801–809
Emre S (2019) Tianren, et al. Mitochondrial unfolded protein response: a stress response with implications for fertility and reproductive aging.[J]. Fertility and sterility
Singh S, Singh TG, Rehni AK, Sharma V, Singh M, Kaur R (2021) Reviving mitochondrial bioenergetics: a relevant approach in epilepsy. Mitochondrion 58:213–226
Zsurka G, Kunz WS (2015) Mitochondrial dysfunction and seizures: the neuronal energy crisis. Lancet Neurol 14(9):956–966
Vovk T, Grabnar I, Martinc B (2014) Antioxidants as a preventive treatment for epileptic process: a review of the current Status[J]. Curr Neuropharmacol 12(6):–
Sinha K, Das J, Pal PB et al (2013) Oxidative stress: the mitochondria-dependent and mitochondria-independent pathways of apoptosis[J]. Arch Toxicol 87(7):1157–1180
Valentin R, Grabow S, Davids MS (2018) The rise of apoptosis: targeting apoptosis in hematologic malignancies. Blood 132(12):1248–1264
Acknowledgements
We acknowledge the support of the Translational Medicine Platform of Academy of Medical Sciences, Zhengzhou University.
Funding
This work was supported by National Natural Science Foundation of China (81971214) and the Outstanding Young Talent Cultivation Project of Henan Science and Technology Innovation Talents(YXKC2022037).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design.XMY,XYW, and YYX performed the experiments. XMY, XYW, and TTP analyzed the data. The first draft of the manuscript was written by XMY and XYW. CW and NCX contributed to the study conception and design, and revised the manuscript. All authors have read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing Interests
All other authors declare that there is no potential conflict of interest.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Ethical Statement
All methods were performed carried out in accordance with relevant guidelines and regulations and were approved by the Animal Care and Use Commission of Zhengzhou University.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yu, X., Wang, X., Xie, Y. et al. Activating Transcription Factor 4-mediated Mitochondrial Unfolded Protein Response Alleviates Hippocampal Neuronal Damage in an In Vitro Model of Epileptiform Discharges. Neurochem Res 48, 2253–2264 (2023). https://doi.org/10.1007/s11064-023-03910-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-023-03910-2