Abstract
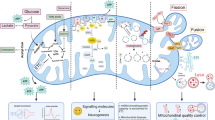
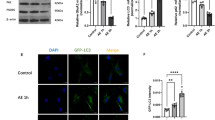
Mitochondrial-associated endoplasmic reticulum (ER) membranes (MAMs) regulate calcium (Ca2+) homeostasis via Ca2+ transport-related proteins such as inositol-1,4,5-triphosphate receptor (IP3R). FAM134B-mediated ER-phagy plays an important role in ER homeostasis. However, it remains unknown whether FAM134B-mediated ER-phagy affects mitochondrial Ca2+ homeostasis and cell death through MAMs. In this study, we demonstrated that colocalization degree of FAM134B with LC3 and the LC3-II/LC3-I ratio were elevated in the hippocampal neuronal culture (HNC) model of acquired epilepsy (AE), which indicate an increased level of autophagy. In this model, FAM134B overexpression enhanced ER-phagy, while FAM134B downregulation had the opposite effect. Additionally, FAM134B overexpression significantly reversed the increases in IP3R expression and mitochondrial Ca2+ concentration and the decrease in the ER Ca2+ concentration in this model. FAM134B overexpression also ameliorated the AE-induced ultrastructural damage in neuronal mitochondria, decrease in mitochondrial membrane potential (mMP), cytochrome c (CytC) release and caspase-3 activation, while FAM134B downregulation induced the opposite effects. Altogether, our data indicate that FAM134B-mediated ER-phagy can attenuate AE-induced neuronal apoptosis, possibly by modulating the IP3R in MAMs to alter Ca2+ exchange between ER and mitochondria and thus inhibit mitochondrial structural damage, a decrease in mMP, release of CytC and mitochondrial apoptosis.







Similar content being viewed by others
References
Schwarz DS, Blower MD (2016) The endoplasmic reticulum: structure, function and response to cellular signaling. Cell Mol Life Sci 73:79–94. https://doi.org/10.1007/s00018-015-2052-6
Walter P, Ron D (2011) The unfolded protein response: from stress pathway to homeostatic regulation. Science 334:1081–1086. https://doi.org/10.1126/science.1209038
De Leonibus C, Cinque L, Settembre C (2019) Emerging lysosomal pathways for quality control at the endoplasmic reticulum. FEBS Lett 593:2319–2329. https://doi.org/10.1002/1873-3468.13571
Song S, Tan J, Miao Y, Zhang Q (2018) Crosstalk of ER stress-mediated autophagy and ER-phagy: Involvement of UPR and the core autophagy machinery. J Cell Physiol 233:3867–3874. https://doi.org/10.1002/jcp.26137
Loi M, Fregno I, Guerra C, Molinari M (2018) Eat it right: ER-phagy and recovER-phagy. Biochem Soc Trans 46:699–706. https://doi.org/10.1042/BST20170354
Khaminets A, Heinrich T, Mari M, Grumati P, Huebner AK, Akutsu M, Liebmann L, Stolz A, Nietzsche S, Koch N, Mauthe M, Katona I, Qualmann B, Weis J, Reggiori F, Kurth I, Hubner CA, Dikic I (2015) Regulation of endoplasmic reticulum turnover by selective autophagy. Nature 522:354–358. https://doi.org/10.1038/nature14498
Bhaskara RM, Grumati P, Garcia-Pardo J, Kalayil S, Covarrubias-Pinto A, Chen W, Kudryashev M, Dikic I, Hummer G (2019) Curvature induction and membrane remodeling by FAM134B reticulon homology domain assist selective ER-phagy. Nat Commun 10:2370. https://doi.org/10.1038/s41467-019-10345-3
Chiramel AI, Best SM (2018) Role of autophagy in Zika virus infection and pathogenesis. Virus Res 254:34–40. https://doi.org/10.1016/j.virusres.2017.09.006
Lewis SC, Uchiyama LF, Nunnari J (2016) ER-mitochondria contacts couple mtDNA synthesis with mitochondrial division in human cells. Science 353:aaf5549. https://doi.org/10.1126/science.aaf5549
Perrone M, Caroccia N, Genovese I, Missiroli S, Modesti L, Pedriali G, Vezzani B, Vitto VAM, Antenori M, Lebiedzinska-Arciszewska M, Wieckowski MR, Giorgi C, Pinton P (2020) The role of mitochondria-associated membranes in cellular homeostasis and diseases. Int Rev Cell Mol Biol 350:119–196. https://doi.org/10.1016/bs.ircmb.2019.11.002
Szabadkai G, Bianchi K, Varnai P, De Stefani D, Wieckowski MR, Cavagna D, Nagy AI, Balla T, Rizzuto R (2006) Chaperone-mediated coupling of endoplasmic reticulum and mitochondrial Ca2+ channels. J Cell Biol 175:901–911. https://doi.org/10.1083/jcb.200608073
Catoni C, Cali T, Brini M (2019) Calcium, dopamine and neuronal calcium sensor 1: their contribution to Parkinson’s disease. Front Mol Neurosci 12:55. https://doi.org/10.3389/fnmol.2019.00055
Nunnari J, Suomalainen A (2012) Mitochondria: in sickness and in health. Cell 148:1145–1159. https://doi.org/10.1016/j.cell.2012.02.035
Giorgi C, Wieckowski MR, Pandolfi PP, Pinton P (2011) Mitochondria associated membranes (MAMs) as critical hubs for apoptosis. Commun Integr Biol 4:334–335. https://doi.org/10.4161/cib.4.3.15021
Sombati S, Delorenzo RJ (1995) Recurrent spontaneous seizure activity in hippocampal neuronal networks in culture. J Neurophysiol 73:1706–1711. https://doi.org/10.1152/jn.1995.73.4.1706
Hinze SJ, Jackson MR, Lie S, Jolly L, Field M, Barry SC, Harvey RJ, Shoubridge C (2017) Incorrect dosage of IQSEC2, a known intellectual disability and epilepsy gene, disrupts dendritic spine morphogenesis. Transl Psychiatry 7:e1110. https://doi.org/10.1038/tp.2017.81
Li Y, Wang C, Lian Y, Zhang H, Meng X, Yu M, Li Y, Xie N (2020) Role of the mitochondrial calcium uniporter in Mg(2+)-free-induced epileptic hippocampal neuronal apoptosis. Int J Neurosci 130:1024–1032. https://doi.org/10.1080/00207454.2020.1715978
Bento CF, Ashkenazi A, Jimenez-Sanchez M, Rubinsztein DC (2016) The Parkinson’s disease-associated genes ATP13A2 and SYT11 regulate autophagy via a common pathway. Nat Commun 7:11803. https://doi.org/10.1038/ncomms11803
Xie N, Li Y, Wang C, Lian Y, Zhang H, Li Y, Meng X, Du L (2020) FAM134B attenuates seizure-induced apoptosis and endoplasmic reticulum stress in hippocampal neurons by promoting autophagy. Cell Mol Neurobiol 40:1297–1305. https://doi.org/10.1007/s10571-020-00814-5
Fu J, Peng L, Wang W, He H, Zeng S, Chen TC, Chen Y (2019) Sodium valproate reduces neuronal apoptosis in acute pentylenetetrzole-induced seizures via inhibiting ER stress. Neurochem Res 44:2517–2526. https://doi.org/10.1007/s11064-019-02870-w
Deegan S, Saveljeva S, Gorman AM, Samali A (2013) Stress-induced self-cannibalism: on the regulation of autophagy by endoplasmic reticulum stress. Cell Mol Life Sci 70:2425–2441. https://doi.org/10.1007/s00018-012-1173-4
Wilkinson S (2020) Emerging principles of selective ER autophagy. J Mol Biol 432:185–205. https://doi.org/10.1016/j.jmb.2019.05.012
Carvajal FJ, Mattison HA, Cerpa W (2016) Role of NMDA receptor-mediated glutamatergic signaling in chronic and acute neuropathologies. Neural Plast 2016:2701526. https://doi.org/10.1155/2016/2701526
Diaz-Prieto N, Herrera-Peco I, de Diego AM, Ruiz-Nuno A, Gallego-Sandin S, Lopez MG, Garcia AG, Cano-Abad MF (2008) Bcl2 mitigates Ca2+ entry and mitochondrial Ca2+ overload through downregulation of L-type Ca2+ channels in PC12 cells. Cell Calcium 44:339–352. https://doi.org/10.1016/j.ceca.2008.01.007
Rieusset J, Fauconnier J, Paillard M, Belaidi E, Tubbs E, Chauvin MA, Durand A, Bravard A, Teixeira G, Bartosch B, Michelet M, Theurey P, Vial G, Demion M, Blond E, Zoulim F, Gomez L, Vidal H, Lacampagne A, Ovize M (2016) Disruption of calcium transfer from ER to mitochondria links alterations of mitochondria-associated ER membrane integrity to hepatic insulin resistance. Diabetologia 59:614–623. https://doi.org/10.1007/s00125-015-3829-8
Zhu L, Wang X, Wang Y (2021) Roles of FAM134B in diseases from the perspectives of organelle membrane morphogenesis and cellular homeostasis. J Cell Physiol. https://doi.org/10.1002/jcp.30377
Giorgi C, Baldassari F, Bononi A, Bonora M, De Marchi E, Marchi S, Missiroli S, Patergnani S, Rimessi A, Suski JM, Wieckowski MR, Pinton P (2012) Mitochondrial Ca(2+) and apoptosis. Cell Calcium 52:36–43. https://doi.org/10.1016/j.ceca.2012.02.008
Marchi S, Patergnani S, Missiroli S, Morciano G, Rimessi A, Wieckowski MR, Giorgi C, Pinton P (2018) Mitochondrial and endoplasmic reticulum calcium homeostasis and cell death. Cell Calcium 69:62–72. https://doi.org/10.1016/j.ceca.2017.05.003
La Rovere RM, Roest G, Bultynck G, Parys JB (2016) Intracellular Ca(2+) signaling and Ca(2+) microdomains in the control of cell survival, apoptosis and autophagy. Cell Calcium 60:74–87. https://doi.org/10.1016/j.ceca.2016.04.005
Vervliet T, Parys JB, Bultynck G (2016) Bcl-2 proteins and calcium signaling: complexity beneath the surface. Oncogene 35:5079–5092. https://doi.org/10.1038/onc.2016.31
Chen L, Sun Q, Zhou D, Song W, Yang Q, Ju B, Zhang L, Xie H, Zhou L, Hu Z, Yao H, Zheng S, Wang W (2017) HINT2 triggers mitochondrial Ca(2+) influx by regulating the mitochondrial Ca(2+) uniporter (MCU) complex and enhances gemcitabine apoptotic effect in pancreatic cancer. Cancer Lett 411:106–116. https://doi.org/10.1016/j.canlet.2017.09.020
Juhaszova M, Wang S, Zorov DB, Nuss HB, Gleichmann M, Mattson MP, Sollott SJ (2008) The identity and regulation of the mitochondrial permeability transition pore: where the known meets the unknown. Ann N Y Acad Sci 1123:197–212. https://doi.org/10.1196/annals.1420.023
Funding
This study was supported by the National Natural Science Foundation of China [Nos. 81701272, 81971214]; Provincial Ministry Co-construction Project from Medical Scientific and Technological Research Program of Henan Province [No. SB201902011] and Training plan for young backbone teachers of Zhengzhou University [No. 2019ZDGGJS056].
Author information
Authors and Affiliations
Contributions
NX and CW contributed to conception and design of the study; YL, YL, LD, JZ, NL, XH and WZ performed the experiments; NX and LM performed the statistical analysis; CW and YL wrote the manuscript. All authors contributed to manuscript revision read and approved the submitted version.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical Approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, C., Li, Y., Li, Y. et al. FAM134B-Mediated ER-Phagy in Mg2+-Free Solution-Induced Mitochondrial Calcium Homeostasis and Cell Death in Epileptic Hippocampal Neurons. Neurochem Res 46, 2485–2494 (2021). https://doi.org/10.1007/s11064-021-03389-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-021-03389-9