Abstract
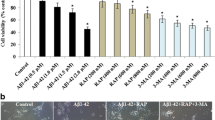
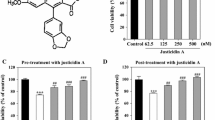
Beta-amyloid (Aβ), the hallmark protein in Alzheimer’s disease (AD), induces neurotoxicity that involves oxidative stress and mitochondrial dysfunction, leading to cell death. Carnosic acid (CA), a polyphenolic diterpene isolated from the herb rosemary (Rosemarinus officinalis), was investigated in our study to assess its neuroprotective effect and underlying mechanism against Aβ-induced injury in human neuroblastoma SH-SY5Y cells. We found that CA pretreatment alleviated the Aβ25–35-induced loss of cell viability, inhibited both Aβ1–42 accumulation and tau hyperphosphorylation, reduced reactive oxygen species generation, and maintained the mitochondrial membrane potential. Moreover, CA increased the microtubule-associated protein light chain 3 (LC3)-II/I ratio and decreased SQSTM1(p62), indicating that CA could induce autophagy. Autophagy inhibitor 3-methyladenine (3-MA) attenuated the neuroprotective effect of CA, suggesting that autophagy was involved in the neuroprotection of CA. It was also observed that CA activated AMP-activated protein kinase (AMPK) but inhibited mammalian target of rapamycin (mTOR). Furthermore, blocking AMPK with si-AMPKα successfully inhibited the upregulation of LC3-II/I, prevented the downregulation of phosphorylation of mTOR and SQSTM1(p62), indicating that CA induced autophagy in SH-SY5Y cells via the activation of AMPK. These results suggested that CA might be a potential agent for preventing AD.






Similar content being viewed by others
Abbreviations
- Aβ:
-
Beta-amyloid
- AD:
-
Alzheimer’s disease
- CA:
-
Carnosic acid
- ROS:
-
Reactive oxygen species
- MMP:
-
Mitochondrial membrane potential
- LC3:
-
Microtubule-associated protein light chain 3
- 3-MA:
-
3-Methyladenine
- AMPK:
-
AMP-activated protein kinase
- mTOR:
-
Mammalian target of rapamycin
- NFT:
-
Neurofibrillary tangles
- CMA:
-
Chaperone mediated autophagy
- MTT:
-
3-(4,5-Dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide
- DMSO:
-
Dimethyl sulfoxide
- RT:
-
Room temperature
- PBS:
-
Phosphate buffered saline
- PI:
-
Propidium iodide
References
Querfurth HW, LaFerla FM (2010) Alzheimer’s disease. N Engl J Med 362(4):329–344
Murrow L, Debnath J (2013) Autophagy as a stress-response and quality-control mechanism: implications for cell injury and human disease. Annu Rev Pathol 8:105–137
Tan CC, Yu JT, Tan MS, Jiang T, Zhu XC, Tan L (2014) Autophagy in aging and neurodegenerative diseases: implications for pathogenesis and therapy. Neurobiol Aging 35(5):941–957
Ghavami S, Shojaei S, Yeganeh B, Ande SR, Jangamreddy JR, Mehrpour M, Christoffersson J, Chaabane W, Moghadam AR, Kashani HH, Hashemi M, Owji AA, Los MJ (2014) Autophagy and apoptosis dysfunction in neurodegenerative disorders. Prog Neurobiol 112:24–49
Nixon RA, Yang DS (2011) Autophagy failure in Alzheimer’s disease-locating the primary defect. Neurobiol Dis 43(1):38–45. doi:10.1016/j.nbd.2011.01.021
Tramutola A, Triplett JC, Di Domenico F, Niedowicz DM, Murphy MP, Coccia R, Perluigi M, Butterfield DA (2015) Alteration of mTOR signaling occurs early in the progression of Alzheimer disease (AD): analysis of brain from subjects with pre-clinical AD, amnestic mild cognitive impairment and late-stage AD. J Neurochem 133(5):739–749
Yang DS, Stavrides P, Mohan PS, Kaushik S, Kumar A, Ohno M, Schmidt SD, Wesson D, Bandyopadhyay U, Jiang Y, Pawlik M, Peterhoff CM, Yang AJ, Wilson DA, George-Hyslop P, Westaway D, Mathews PM, Levy E, Cuervo AM, Nixon RA (2011) Reversal of autophagy dysfunction in the TgCRND8 mouse model of Alzheimer’s disease ameliorates amyloid pathologies and memory deficits. Brain 134:258–277. doi:10.1093/brain/awq341
Salminen A, Kaarniranta K, Kauppinen A, Ojala J, Haapasalo A, Soininen H, Hiltunen M (2013) Impaired autophagy and APP processing in Alzheimer’s disease: the potential role of Beclin 1 interactome. Prog Neurobiol 106:33–54. doi:10.1016/j.pneurobio.2013.06.002
Vingtdeux V, Giliberto L, Zhao HT, Chandakkar P, Wu QL, Simon JE, Janle EM, Lobo J, Ferruzzi MG, Davies P, Marambaud P (2010) AMP-activated protein kinase signaling activation by resveratrol modulates amyloid-beta peptide metabolism. J Biol Chem 285(12):9100–9113. doi:10.1074/jbc.M109.060061
Li LX, Zhang SF, Zhang X, Li T, Tang Y, Liu H, Yang WD, Le WD (2013) Autophagy enhancer carbamazepine alleviates memory deficits and cerebral amyloid-beta pathology in a mouse model of Alzheimer’s disease. Curr Alzheimer Res 10(4):433–441
Scherz-Shouval R, Elazar Z (2007) ROS, mitochondria and the regulation of autophagy. Trends Cell Biol 17(9):422–427. doi:10.1016/j.tcb.2007.07.009
Wang K, Klionsky DJ (2011) Mitochondria removal by autophagy. Autophagy 7(3):297–300. doi:10.4161/auto.7.3.14502
Lee J, Giordano S, Zhang JH (2012) Autophagy, mitochondria and oxidative stress: cross-talk and redox signalling. Biochem J 441:523–540. doi:10.1042/bj20111451
Filomeni G, Graziani I, De Zio D, Dini L, Centonze D, Rotilio G, Ciriolo MR (2012) Neuroprotection of kaempferol by autophagy in models of rotenone-mediated acute toxicity: possible implications for Parkinson’s disease. Neurobiol Aging 33(4):767–785. doi:10.1016/j.neurobiolaging.2010.05.021
Ashabi G, Ahmadiani A, Abdi A, Abraki SB, Khodagholi F (2013) Time course study of A beta formation and neurite outgrowth disruption in differentiated human neuroblastoma cells exposed to H2O2: protective role of autophagy. Toxicol In Vitro 27(6):1780–1788. doi:10.1016/j.tiv.2013.05.005
Wu JJ, Quijano C, Chen E, Liu HJ, Cao L, Fergusson MM, Rovira II, Gutkind S, Daniels MP, Komatsu M, Finkel T (2009) Mitochondrial dysfunction and oxidative stress mediate the physiological impairment induced by the disruption of autophagy. Aging USA 1(4):425–437
Rezaie T, McKercher SR, Kosaka K, Seki M, Wheeler L, Viswanath V, Chun T, Joshi R, Valencia M, Sasaki S, Tozawa T, Satoh T, Lipton SA (2012) Protective effect of carnosic acid, a pro-electrophilic compound, in models of oxidative stress and light-induced retinal degeneration. Investig Ophthalmol Vis Sci 53(12):7847–7854
Wang T, Takikawa Y, Tabuchi T, Satoh T, Kosaka K, Suzuki K (2012) Carnosic acid (CA) prevents lipid accumulation in hepatocytes through the EGFR/MAPK pathway. J Gastroenterol 47(7):805–813
Barni MV, Carlini MJ, Cafferata EG, Puricelli L, Moreno S (2012) Carnosic acid inhibits the proliferation and migration capacity of human colorectal cancer cells. Oncol Rep 27(4):1041–1048
Lopez-Jimenez A, Garcia-Caballero M, Medina MA, Quesada AR (2013) Anti-angiogenic properties of carnosol and carnosic acid, two major dietary compounds from rosemary. Eur J Nutr 52(1):85–95
Hou CW, Lin YT, Chen YL, Wang YH, Chou JL, Ping LY, Jeng KC (2012) Neuroprotective effects of carnosic acid on neuronal cells under ischemic and hypoxic stress. Nutr Neurosci 7:7
Chen JH, Ou HP, Lin CY, Lin FJ, Wu CR, Chang SW, Tsai CW (2012) Carnosic acid prevents 6-hydroxydopamine-induced cell death in SH-SY5Y cells via mediation of glutathione synthesis. Chem Res Toxicol 25(9):1893–1901
Azad N, Rasoolijazi H, Joghataie MT, Soleimani S (2011) Neuroprotective effects of carnosic Acid in an experimental model of Alzheimer’s disease in rats. Cell J 13(1):39–44
Rasoolijazi H, Azad N, Joghataei MT, Kerdari M, Nikbakht F, Soleimani M (2013) The protective role of carnosic acid against beta-amyloid toxicity in rats. Sci World J 24(917082):1–5
Meng P, Yoshida H, Matsumiya T, Imaizumi T, Tanji K, Xing F, Hayakari R, Dempoya J, Tatsuta T, Aizawa-Yashiro T, Mimura J, Kosaka K, Itoh K, Satoh K (2013) Carnosic acid suppresses the production of amyloid-beta 1-42 by inducing the metalloprotease gene TACE/ADAM17 in SH-SY5Y human neuroblastoma cells. Neurosci Res 75(2):94–102
Yoshida H, Meng P, Matsumiya T, Tanji K, Hayakari R, Xing F, Wang L, Tsuruga K, Tanaka H, Mimura J, Kosaka K, Itoh K, Takahashi I, Imaizumi T (2014) Carnosic acid suppresses the production of amyloid-beta 1-42 and 1-43 by inducing an alpha-secretase TACE/ADAM17 in U373MG human astrocytoma cells. Neurosci Res 79:83–93
Duan XY, Pan Q, Fan JG (2011) Carnosic acid promotes liver autophagy in rats with nonalcoholic steatohepatitis. J Gastroenterol Hepatol 26:174
Ma W, Yuan L, Yu H, Ding B, Xi Y, Feng J, Xiao R (2010) Genistein as a neuroprotective antioxidant attenuates redox imbalance induced by beta-amyloid peptides 25-35 in PC12 cells. Int J Dev Neurosci 28(4):289–295
Deng YN, Shi J, Liu J, Qu QM (2013) Celastrol protects human neuroblastoma SH-SY5Y cells from rotenone-induced injury through induction of autophagy. Neurochem Int 63(1):1–9
Wong E, Cuervo AM (2010) Autophagy gone awry in neurodegenerative diseases. Nat Neurosci 13(7):805–811
Li L, Zhang X, Le W (2010) Autophagy dysfunction in Alzheimer’s disease. Neurodegener Dis 7(4):265–271
Tsuruma K, Tanaka Y, Shimazawa M, Hara H (2010) Induction of amyloid precursor protein by the neurotoxic peptide, amyloid-beta 25–35, causes retinal ganglion cell death. J Neurochem 113(6):1545–1554
Stancu IC, Vasconcelos B, Terwel D, Dewachter I (2014) Models of beta-amyloid induced Tau-pathology: the long and “folded” road to understand the mechanism. Mol Neurodegener 9(51):1326–1750
Zhang L, Yu H, Zhao X, Lin X, Tan C, Cao G, Wang Z (2010) Neuroprotective effects of salidroside against beta-amyloid-induced oxidative stress in SH-SY5Y human neuroblastoma cells. Neurochem Int 57(5):547–555
Naldi M, Fiori J, Pistolozzi M, Drake AF, Bertucci C, Wu R, Mlynarczyk K, Filipek S, De Simone A, Andrisano V (2012) Amyloid beta-peptide 25–35 self-assembly and its inhibition: a model undecapeptide system to gain atomistic and secondary structure details of the Alzheimer’s disease process and treatment. ACS Chem Neurosci 3(11):952–962
Miller DM, Singh IN, Wang JA, Hall ED (2015) Nrf2-ARE activator carnosic acid decreases mitochondrial dysfunction, oxidative damage and neuronal cytoskeletal degradation following traumatic brain injury in mice. Exp Neurol 264:103–110
Katsuragi Y, Ichimura Y, Komatsu M (2015) p62/SQSTM1 functions as a signaling hub and an autophagy adaptor. FEBS J 282(24):4672–4678
Gao Q, Liu H, Yao Y, Geng L, Zhang X, Jiang L, Shi B, Yang F (2015) Carnosic acid induces autophagic cell death through inhibition of the Akt/mTOR pathway in human hepatoma cells. J Appl Toxicol 35(5):485–492
Xue Z, Zhang S, Huang L, He Y, Fang R, Fang Y (2013) Increased expression of Beclin-1-dependent autophagy protects against beta-amyloid-induced cell injury in PC12 cells [corrected]. J Mol Neurosci 51(1):180–186
Levine B, Kroemer G (2008) Autophagy in the pathogenesis of disease. Cell 132(1):27–42
Dai ZJ, Gao J, Ma XB, Kang HF, Wang BF, Lu WF, Lin S, Wang XJ, Wu WY (2012) Antitumor effects of rapamycin in pancreatic cancer cells by inducing apoptosis and autophagy. Int J Mol Sci 14(1):273–285
Urbanek T, Kuczmik W, Basta-Kaim A, Gabryel B (2014) Rapamycin induces of protective autophagy in vascular endothelial cells exposed to oxygen-glucose deprivation. Brain Res 17:1–11
Cai Z, Yan LJ (2013) Rapamycin, autophagy, and Alzheimer’s disease. J Biochem Pharmacol Res 1(2):84–90
Butterfield DA, Boyd-Kimball D (2004) Amyloid beta-peptide(1-42) contributes to the oxidative stress and neurodegeneration found in Alzheimer disease brain. Brain Pathol 14(4):426–432
Kienlen-Campard P, Miolet S, Tasiaux B, Octave JN (2002) Intracellular amyloid-beta 1-42, but not extracellular soluble amyloid-beta peptides, induces neuronal apoptosis. J Biol Chem 277(18):15666–15670
Zhu XC, Yu JT, Jiang T, Tan L (2013) Autophagy modulation for Alzheimer’s disease therapy. Mol Neurobiol 48(3):702–714
Meng P, Yoshida H, Tanji K, Matsumiya T, Xing F, Hayakari R, Wang L, Tsuruga K, Tanaka H, Mimura J, Kosaka K, Itoh K, Takahashi I, Kawaguchi S, Imaizumi T (2015) Carnosic acid attenuates apoptosis induced by amyloid-beta 1–42 or 1–43 in SH-SY5Y human neuroblastoma cells. Neurosci Res 94:1–9
Tung YT, Wang BJ, Hu MK, Hsu WM, Lee H, Huang WP, Liao YF (2012) Autophagy: a double-edged sword in Alzheimer’s disease. J Biosci 37(1):157–165
Zhang JY, Peng C, Shi H, Wang S, Wang Q, Wang JZ (2009) Inhibition of autophagy causes tau proteolysis by activating calpain in rat brain. J Alzheimers Dis 16(1):39–47
Wang X, Wang W, Li L, Perry G, Lee HG, Zhu X (2014) Oxidative stress and mitochondrial dysfunction in Alzheimer’s disease. Biochim Biophys Acta 8(7):1
Zhang J (2013) Autophagy and mitophagy in cellular damage control. Redox Biol 1(1):19–23
Liu D, Pitta M, Jiang H, Lee JH, Zhang G, Chen X, Kawamoto EM, Mattson MP (2013) Nicotinamide forestalls pathology and cognitive decline in Alzheimer mice: evidence for improved neuronal bioenergetics and autophagy procession. Neurobiol Aging 34(6):1564–1580
Tizon B, Sahoo S, Yu H, Gauthier S, Kumar AR, Mohan P, Figliola M, Pawlik M, Grubb A, Uchiyama Y, Bandyopadhyay U, Cuervo AM, Nixon RA, Levy E (2010) Induction of autophagy by cystatin C: a mechanism that protects murine primary cortical neurons and neuronal cell lines. PLoS One 5(3):0009819
Omata Y, Lim YM, Akao Y, Tsuda L (2014) Age-induced reduction of autophagy-related gene expression is associated with onset of Alzheimer’s disease. Am J Neurodegener Dis 3(3):134–142
Mihaylova MM, Shaw RJ (2011) The AMPK signalling pathway coordinates cell growth, autophagy and metabolism. Nat Cell Biol 13(9):1016–1023
Kim J, Kundu M, Viollet B, Guan KL (2011) AMPK and mTOR regulate autophagy through direct phosphorylation of Ulk1. Nat Cell Biol 13(2):132–141
Jung CH, Ro SH, Cao J, Otto NM, Kim DH (2010) mTOR regulation of autophagy. FEBS Lett 584(7):1287–1295
Hosokawa N, Hara T, Kaizuka T, Kishi C, Takamura A, Miura Y, Iemura S, Natsume T, Takehana K, Yamada N, Guan JL, Oshiro N, Mizushima N (2009) Nutrient-dependent mTORC1 association with the ULK1-Atg13-FIP200 complex required for autophagy. Mol Biol Cell 20(7):1981–1991
Lipina C, Hundal HS (2014) Carnosic acid stimulates glucose uptake in skeletal muscle cells via a PME-1/PP2A/PKB signalling axis. Cell Signal 26(11):2343–2349
Acknowledgments
This work was supported by the Natural Science Foundation of China from Yong-Ning Deng (No.81301097).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to disclose.
Rights and permissions
About this article
Cite this article
Liu, J., Su, H. & Qu, QM. Carnosic Acid Prevents Beta-Amyloid-Induced Injury in Human Neuroblastoma SH-SY5Y Cells via the Induction of Autophagy. Neurochem Res 41, 2311–2323 (2016). https://doi.org/10.1007/s11064-016-1945-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-016-1945-6