Abstract
Microglial activation and release of inflammatory cytokines and chemokines are crucial events in neuroinflammation. Microglial cells interact and respond to other inflammatory cells such as T cells and mast cells as well as inflammatory mediators secreted from these cells. Recent studies have shown that neuroinflammation causes and accelerates neurodegenerative disease such as Parkinson’s disease (PD) pathogenesis. 1-methyl-4-phenyl-pyridinium ion (MPP+), the active metabolite of neurotoxin 1-methyl-4-phenyl-1,2,3,6-tetrahydro pyridine activates glial cells and mediate neurodegeneration through release of inflammatory mediators. We have shown that glia maturation factor (GMF) activates glia and induces neuroinflammation and neurodegeneration and that MPP+ activates mast cells and release proinflammatory cytokines and chemokines. The chemokine (C-C motif) ligand 2 (CCL2) levels have been shown to be elevated and play a role in PD pathogenesis. In the present study, we analyzed if MPP+ activates mouse and human mast cells to release chemokine CCL2. Mouse bone marrow-derived mast cells (BMMCs) and human umbilical cord blood-derived cultured mast cells (hCBMCs) were incubated with MPP+ (10 µM) for 24 h and CCL2 levels were measured in the supernatant media by ELISA. MPP+-significantly induced CCL2 release from BMMCs and hCBMCs. Additionally, GMF overexpression in BMMCs obtained from wild-type mice released significantly more CCL2, while BMMCs obtained from GMF-deficient mice showed less CCL2 release. Further, we show that MPP+-induced CCL2 release was greater in BMMCs–astrocyte co-culture conditions. Uncoupling protein 4 (UCP4) which is implicated in neurodegenerative diseases including PD was detected in BMMCs by immunocytochemistry. Our results suggest that mast cells may play role in PD pathogenesis.





Similar content being viewed by others
References
Dong H, Zhang X, Qian Y (2014) Mast cells and neuroinflammation. Med Sci Monit Basic Res 20:200–206
Lyman M, Lloyd DG, Ji X, Vizcaychipi MP, Ma D (2014) Neuroinflammation: the role and consequences. Neurosci Res 79:1–12
Jarrott B, Williams SJ (2015) Chronic brain inflammation: the neurochemical basis for drugs to reduce inflammation. Neurochem Res. doi:10.1007/s11064-015-1661-7
Trudler D, Nash Y, Frenkel D (2015) New insights on Parkinson’s disease genes: the link between mitochondria impairment and neuroinflammation. J Neural Transm 122:1409–1419
Stojkovska I, Wagner BM, Morrison BE (2015) Parkinson’s disease and enhanced inflammatory response. Exp Biol Med (Maywood) 240:1387–1395
Czlonkowska A, Kohutnicka M, Kurkowska-Jastrzebska I, Czlonkowski A (1996) Microglial reaction in MPTP (1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine) induced Parkinson’s disease mice model. Neurodegeneration 5:137–143
Zhai A, Zhu X, Wang X, Chen R, Wang H (2013) Secalonic acid A protects dopaminergic neurons from 1-methyl-4-phenylpyridinium (MPP(+))-induced cell death via the mitochondrial apoptotic pathway. Eur J Pharmacol 713:58–67
Khan MM, Kempuraj D, Zaheer S, Zaheer A (2014) Glia maturation factor deficiency suppresses 1-methyl-4-phenylpyridinium-induced oxidative stress in astrocytes. J Mol Neurosci 53:590–599
Lim R, Miller JF, Zaheer A (1989) Purification and characterization of glia maturation factor beta: a growth regulator for neurons and glia. Proc Natl Acad Sci USA 86:3901–3905
Lim R, Zaheer A (1991) Structure and function of glia maturation factor beta. Adv Exp Med Biol 296:161–164
Kaplan R, Zaheer A, Jaye M, Lim R (1991) Molecular cloning and expression of biologically active human glia maturation factor-beta. J Neurochem 57:483–490
Zaheer A, Fink BD, Lim R (1993) Expression of glia maturation factor beta mRNA and protein in rat organs and cells. J Neurochem 60:914–920
Zaheer S, Wu Y, Sahu SK, Zaheer A (2011) Suppression of neuro inflammation in experimental autoimmune encephalomyelitis by glia maturation factor antibody. Brain Res 1373:230–239
Zaheer A, Zaheer S, Sahu SK, Knight S, Khosravi H, Mathur SN, Lim R (2007) A novel role of glia maturation factor: induction of granulocyte–macrophage colony-stimulating factor and pro-inflammatory cytokines. J Neurochem 101:364–376
Khan MM, Zaheer S, Thangavel R, Patel M, Kempuraj D, Zaheer A (2015) Absence of Glia maturation factor protects dopaminergic neurons and improves motor behavior in mouse model of parkinsonism. Neurochem Res 40:980–990
Zaheer S, Wu Y, Sahu SK, Zaheer A (2010) Overexpression of glia maturation factor reinstates susceptibility to myelin oligodendrocyte glycoprotein-induced experimental autoimmune encephalomyelitis in glia maturation factor deficient mice. Neurobiol Dis 40:593–598
Zaheer A, Mathur SN, Lim R (2002) Overexpression of glia maturation factor in astrocytes leads to immune activation of microglia through secretion of granulocyte–macrophage-colony stimulating factor. Biochem Biophys Res Commun 294:238–244
Skaper SD, Facci L, Giusti P (2014) Neuroinflammation, microglia and mast cells in the pathophysiology of neurocognitive disorders: a review. CNS Neurol Disord Drug Targets 13:1654–1666
Nelissen S, Lemmens E, Geurts N, Kramer P, Maurer M, Hendriks J, Hendrix S (2013) The role of mast cells in neuroinflammation. Acta Neuropathol 125:637–650
Skaper SD, Giusti P, Facci L (2012) Microglia and mast cells: two tracks on the road to neuroinflammation. FASEB J 26:3103–3117
Kim DY, Jeoung D, Ro JY (2010) Signaling pathways in the activation of mast cells cocultured with astrocytes and colocalization of both cells in experimental allergic encephalomyelitis. J Immunol 185:273–283
Kempuraj D, Thangavel R, Yang E, Pattani S, Zaheer S, Santillan DA, Santillan MK, Zaheer A (2015) Dopaminergic toxin 1-methyl-4-phenylpyridinium, proteins alpha-synuclein and Glia maturation factor activate mast cells and release inflammatory mediators. PLoS One 10:e0135776
Conti B, Sugama S, Lucero J, Winsky-Sommerer R, Wirz SA, Maher P, Andrews Z, Barr AM, Morale MC, Paneda C, Pemberton J, Gaidarova S, Behrens MM, Beal F, Sanna PP, Horvath T, Bartfai T (2005) Uncoupling protein 2 protects dopaminergic neurons from acute 1,2,3,6-methyl-phenyl-tetrahydropyridine toxicity. J Neurochem 93:493–501
Tagen M, Elorza A, Kempuraj D, Boucher W, Kepley CL, Shirihai OS, Theoharides TC (2009) Mitochondrial uncoupling protein 2 inhibits mast cell activation and reduces histamine content. J Immunol 183:6313–6319
Chu AC, Ho PW, Kwok KH, Ho JW, Chan KH, Liu HF, Kung MH, Ramsden DB, Ho SL (2009) Mitochondrial UCP4 attenuates MPP+-and dopamine-induced oxidative stress, mitochondrial depolarization, and ATP deficiency in neurons and is interlinked with UCP2 expression. Free Radic Biol Med 46:810–820
Ho PW, Ho JW, Liu HF, So DH, Tse ZH, Chan KH, Ramsden DB, Ho SL (2012) Mitochondrial neuronal uncoupling proteins: a target for potential disease-modification in Parkinson’s disease. Transl Neurodegener 1:3
Lim R, Zaheer A, Khosravi H, Freeman JH Jr, Halverson HE, Wemmie JA, Yang B (2004) Impaired motor performance and learning in glia maturation factor-knockout mice. Brain Res 1024:225–232
Kim DY, Hong GU, Ro JY (2011) Signal pathways in astrocytes activated by cross-talk between of astrocytes and mast cells through CD40-CD40L. J Neuroinflammation 8:25
Sayed BA, Walker ME, Brown MA (2011) Cutting edge: mast cells regulate disease severity in a relapsing-remitting model of multiple sclerosis. J Immunol 186:3294–3298
Zaheer A, Yorek MA, Lim R (2001) Effects of glia maturation factor overexpression in primary astrocytes on MAP kinase activation, transcription factor activation, and neurotrophin secretion. Neurochem Res 26:1293–1299
Zaheer A, Yang B, Cao X, Lim R (2004) Decreased copper-zinc superoxide dismutase activity and increased resistance to oxidative stress in glia maturation factor-null astrocytes. Neurochem Res 29:1473–1480
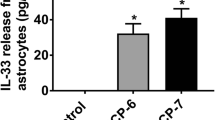
Kempuraj D, Khan MM, Thangavel R, Xiong Z, Yang E, Zaheer A (2013) Glia maturation factor induces interleukin-33 release from astrocytes: implications for neurodegenerative diseases. J Neuroimmune Pharmacol 8:643–650
Kempuraj D, Saito H, Kaneko A, Fukagawa K, Nakayama M, Toru H, Tomikawa M, Tachimoto H, Ebisawa M, Akasawa A, Miyagi T, Kimura H, Nakajima T, Tsuji K, Nakahata T (1999) Characterization of mast cell-committed progenitors present in human umbilical cord blood. Blood 93:3338–3346
Kempuraj D, Asadi S, Zhang B, Manola A, Hogan J, Peterson E, Theoharides TC (2010) Mercury induces inflammatory mediator release from human mast cells. J Neuroinflammation 7:20
Santillan MK, Leslie KK, Hamilton WS, Boese BJ, Ahuja M, Hunter SK, Santillan DA (2014) Collection of a lifetime: a practical approach to developing a longitudinal collection of women’s healthcare biological samples. Eur J Obstet Gynecol Reprod Biol 179:94–99
Bulfone-Paus S, Bahri R (2015) Mast cells as regulators of T cell responses. Front Immunol 6:394
Theoharides TC, Valent P, Akin C (2015) Mast cells, mastocytosis, and related disorders. N Engl J Med 373:163–172
Theoharides TC, Alysandratos KD, Angelidou A, Delivanis DA, Sismanopoulos N, Zhang B, Asadi S, Vasiadi M, Weng Z, Miniati A, Kalogeromitros D (2012) Mast cells and inflammation. Biochim Biophys Acta 1822:21–33
Silver R, Curley JP (2013) Mast cells on the mind: new insights and opportunities. Trends Neurosci 36:513–521
Chikahisa S, Kodama T, Soya A, Sagawa Y, Ishimaru Y, Sei H, Nishino S (2013) Histamine from brain resident MAST cells promotes wakefulness and modulates behavioral states. PLoS One 8:e78434
Skaper SD, Facci L, Barbierato M, Zusso M, Bruschetta G, Impellizzeri D, Cuzzocrea S, Giusti P (2015) N-Palmitoylethanolamine and neuroinflammation: a novel therapeutic strategy of resolution. Mol Neurobiol 52:1034–1042
Kalesnikoff J, Galli SJ (2008) New developments in mast cell biology. Nat Immunol 9:1215–1223
Mekori YA, Metcalfe DD (2000) Mast cells in innate immunity. Immunol Rev 173:131–140
Sismanopoulos N, Delivanis DA, Alysandratos KD, Angelidou A, Therianou A, Kalogeromitros D, Theoharides TC (2012) Mast cells in allergic and inflammatory diseases. Curr Pharm Des 18:2261–2277
Madrigal JL, Caso JR (2014) The chemokine (C-C motif) ligand 2 in neuroinflammation and neurodegeneration. Adv Exp Med Biol 824:209–219
Stamatovic SM, Shakui P, Keep RF, Moore BB, Kunkel SL, Van Rooijen N, Andjelkovic AV (2005) Monocyte chemoattractant protein-1 regulation of blood–brain barrier permeability. J Cereb Blood Flow Metab 25:593–606
Yao Y, Tsirka SE (2014) Monocyte chemoattractant protein-1 and the blood–brain barrier. Cell Mol Life Sci 71:683–697
Reale M, Iarlori C, Thomas A, Gambi D, Perfetti B, Di Nicola M, Onofrj M (2009) Peripheral cytokines profile in Parkinson’s disease. Brain Behav Immun 23:55–63
Thangavel R, Stolmeier D, Yang X, Anantharam P, Zaheer A (2012) Expression of glia maturation factor in neuropathological lesions of Alzheimer’s disease. Neuropathol Appl Neurobiol 38:572–581
Zaheer S, Thangavel R, Sahu SK, Zaheer A (2011) Augmented expression of glia maturation factor in Alzheimer’s disease. Neuroscience 194:227–233
Zaheer A, Knight S, Zaheer A, Ahrens M, Sahu SK, Yang B (2008) Glia maturation factor overexpression in neuroblastoma cells activates glycogen synthase kinase-3beta and caspase-3. Brain Res 1190:206–214
Zaheer A, Zaheer S, Thangavel R, Wu Y, Sahu SK, Yang B (2008) Glia maturation factor modulates beta-amyloid-induced glial activation, inflammatory cytokine/chemokine production and neuronal damage. Brain Res 1208:192–203
Khan MM, Zaheer S, Nehman J, Zaheer A (2014) Suppression of glia maturation factor expression prevents 1-methyl-4-phenylpyridinium (MPP(+))-induced loss of mesencephalic dopaminergic neurons. Neuroscience 277:196–205
Reddy PH, Reddy TP (2011) Mitochondria as a therapeutic target for aging and neurodegenerative diseases. Curr Alzheimer Res 8:393–409
Andrews ZB, Horvath B, Barnstable CJ, Elsworth J, Yang L, Beal MF, Roth RH, Matthews RT, Horvath TL (2005) Uncoupling protein-2 is critical for nigral dopamine cell survival in a mouse model of Parkinson’s disease. J Neurosci 25:184–191
Ramsden DB, Ho PW, Ho JW, Liu HF, So DH, Tse HM, Chan KH, Ho SL (2012) Human neuronal uncoupling proteins 4 and 5 (UCP4 and UCP5): structural properties, regulation, and physiological role in protection against oxidative stress and mitochondrial dysfunction. Brain Behav 2:468–478
Negre-Salvayre A, Hirtz C, Carrera G, Cazenave R, Troly M, Salvayre R, Penicaud L, Casteilla L (1997) A role for uncoupling protein-2 as a regulator of mitochondrial hydrogen peroxide generation. FASEB J 11:809–815
Rousset S, Emre Y, Join-Lambert O, Hurtaud C, Ricquier D, Cassard-Doulcier AM (2006) The uncoupling protein 2 modulates the cytokine balance in innate immunity. Cytokine 35:135–142
Acknowledgments
This material is based upon work supported, in part, by the Department of Veterans Affairs, Veterans Health Administration, Office of Research and Development, Biomedical Laboratory Research and Development (BX002477-01, A.Z.), and by the National Institutes of Health (NIH) National Institute of Neurological Disorders and Stroke Grant NS073670 (A.Z.), and the Reproductive Scientist Development Program HD000849, RR024980 (MS) and CSTA U54TR001013. The Maternal Fetal Tissue Bank at the University of Iowa is supported by the University of Iowa Carver College of Medicine and Department of Obstetrics & Gynecology.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kempuraj, D., Thangavel, R., Fattal, R. et al. Mast Cells Release Chemokine CCL2 in Response to Parkinsonian Toxin 1-Methyl-4-Phenyl-Pyridinium (MPP+). Neurochem Res 41, 1042–1049 (2016). https://doi.org/10.1007/s11064-015-1790-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-015-1790-z