Abstract
Purpose
Outcomes for patients with recurrent high-grade glioma (HGG) progressing on bevacizumab (BEV) are dismal. Fractionated stereotactic radiosurgery (FSRS) has been shown to be feasible and safe when delivered in this setting, but prospective evidence is lacking. This single-institution randomized trial compared FSRS plus BEV-based chemotherapy versus BEV-based chemotherapy alone for BEV-resistant recurrent malignant glioma.
Materials and methods
HGG patients on BEV with tumor progression after 2 previous treatments were randomized to 1) FSRS plus BEV-based chemotherapy or 2) BEV-based chemotherapy with irinotecan, etoposide, temozolomide, or carboplatin. FSRS was delivered as 32 Gy (8 Gy × 4 fractions within 2 weeks) to the gross target volume and 24 Gy (6 Gy × 4 fractions) to the clinical target volume (fluid-attenuated inversion recovery abnormality). The primary endpoints were local control (LC) at 2 months and progression-free survival (PFS).
Results
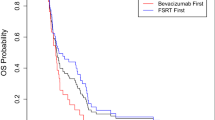
Of the 35 patients enrolled, 29 had glioblastoma (WHO IV) and 6 had anaplastic glioma (WHO III). The median number of prior recurrences was 3. Patients treated with FSRS had significantly improved PFS (5.1 vs 1.8 months, P < .001) and improved LC at 2 months (82% [14/17] vs 27% [4/15], P = .002). The overall median survival was 6.6 months (7.2 months with FSRS vs 4.8 months with chemotherapy alone, P = .11).
Conclusions
FSRS combined with BEV-based chemotherapy in recurrent HGG patients progressing on BEV is feasible and improves LC and PFS when compared to treatment with BEV-based chemotherapy alone.


Similar content being viewed by others
Data availability
Data will be available from the authors upon request.
References
Ostrom QT, Gittleman H, Farah P, Ondracek A, Chen Y, Wolinsky Y, Stroup NE, Kruchko C, Barnholtz-Sloan JS (2013) CBTRUS statistical report: primary brain and central nervous system tumors diagnosed in the United States in 2006–2010. Neuro Oncol 15:ii1–ii56. https://doi.org/10.1093/neuonc/not151
Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, Belanger K, Brandes AA, Marosi C, Bogdahn U, Curschmann J, Janzer RC, Ludwin SK, Gorlia T, Allgeier A, Lacombe D, Cairncross JG, Eisenhauer E, Mirimanoff RO, European Organisation for Research, and Treatment of Cancer Brain Tumor, and Radiotherapy Groups, National Cancer Institute of Canada Clinical Trials Group (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352:987–996. https://doi.org/10.1056/NEJMoa043330
van den Bent MJ, Brandes AA, Taphoorn MJ, Kros JM, Kouwenhoven MC, Delattre JY, Bernsen HJ, Frenay M, Tijssen CC, Grisold W, Sipos L, Enting RH, French PJ, Dinjens WN, Vecht CJ, Allgeier A, Lacombe D, Gorlia T, Hoang-Xuan K (2013) Adjuvant procarbazine, lomustine, and vincristine chemotherapy in newly diagnosed anaplastic oligodendroglioma: long-term follow-up of EORTC brain tumor group study 26951. J Clin Oncol 31:344–350. https://doi.org/10.1200/JCO.2012.43.2229
Cairncross G, Wang M, Shaw E, Jenkins R, Brachman D, Buckner J, Fink K, Souhami L, Laperriere N, Curran W, Mehta M (2013) Phase III trial of chemoradiotherapy for anaplastic oligodendroglioma: long-term results of RTOG 9402. J Clin Oncol 31:337–343. https://doi.org/10.1200/JCO.2012.43.2674
Wick W, Roth P, Hartmann C, Hau P, Nakamura M, Stockhammer F, Sabel MC, Wick A, Koeppen S, Ketter R, Vajkoczy P, Eyupoglu I, Kalff R, Pietsch T, Happold C, Galldiks N, Schmidt-Graf F, Bamberg M, Reifenberger G, Platten M, von Deimling A, Meisner C, Wiestler B, Weller M, Neurooncology Working Group of the German Cancer Society (2016) Long-term analysis of the NOA-04 randomized phase III trial of sequential radiochemotherapy of anaplastic glioma with PCV or temozolomide. Neuro Oncol 18:1529–1537. https://doi.org/10.1093/neuonc/now133
Sneed PK, Gutin PH, Larson DA, Malec MK, Phillips TL, Prados MD, Scharfen CO, Weaver KA, Wara WM (1994) Patterns of recurrence of glioblastoma multiforme after external irradiation followed by implant boost. Int J Radiat Oncol Biol Phys 29:719–727. https://doi.org/10.1016/0360-3016(94)90559-2
Norden AD, Young GS, Setayesh K, Muzikansky A, Klufas R, Ross GL, Ciampa AS, Ebbeling LG, Levy B, Drappatz J, Kesari S, Wen PY (2008) Bevacizumab for recurrent malignant gliomas: efficacy, toxicity, and patterns of recurrence. Neurology 70:779–787. https://doi.org/10.1212/01.wnl.0000304121.57857.38
Vredenburgh JJ, Desjardins A, Herndon JE 2nd, Marcello J, Reardon DA, Quinn JA, Rich JN, Sathornsumetee S, Gururangan S, Sampson J, Wagner M, Bailey L, Bigner DD, Friedman AH, Friedman HS (2007) Bevacizumab plus irinotecan in recurrent glioblastoma multiforme. J Clin Oncol 25:4722–4729. https://doi.org/10.1200/JCO.2007.12.2440
Tipping M, Eickhoff J, Ian Robins H (2017) Clinical outcomes in recurrent glioblastoma with bevacizumab therapy: an analysis of the literature. J Clin Neurosci 44:101–106. https://doi.org/10.1016/j.jocn.2017.06.070
Li Y, Ali S, Clarke J, Cha S (2017) Bevacizumab in recurrent glioma: patterns of treatment failure and implications. Brain Tumor Res Treat 5:1–9
Dirks P, Bernstein M, Muller PJ, Tucker WS (1993) The value of reoperation for recurrent glioblastoma. Can J Surg 36:271–275
Hudes RS, Corn BW, Werner-Wasik M, Andrews D, Rosenstock J, Thoron L, Downes B, Curran WJ Jr (1999) A phase I dose escalation study of hypofractionated stereotactic radiotherapy as salvage therapy for persistent or recurrent malignant glioma. Int J Radiat Oncol Biol Phys 43:293–298. https://doi.org/10.1016/s0360-3016(98)00416-7
Souhami L, Seiferheld W, Brachman D, Podgorsak EB, Werner-Wasik M, Lustig R, Schultz CJ, Sause W, Okunieff P, Buckner J, Zamorano L, Mehta MP, Curran WJ Jr (2004) Randomized comparison of stereotactic radiosurgery followed by conventional radiotherapy with carmustine to conventional radiotherapy with carmustine for patients with glioblastoma multiforme: report of Radiation Therapy Oncology Group 93–05 protocol. Int J Radiat Oncol Biol Phys 60:853–860. https://doi.org/10.1016/j.ijrobp.2004.04.011
Imber BS, Kanungo I, Braunstein S, Barani IJ, Fogh SE, Nakamura JL, Berger MS, Chang EF, Molinaro AM, Cabrera JR, McDermott MW, Sneed PK, Aghi MK (2017) Indications and efficacy of gamma knife stereotactic radiosurgery for recurrent glioblastoma: 2 decades of institutional experience. Neurosurgery 80:129–139. https://doi.org/10.1227/NEU.0000000000001344
Larson EW, Peterson HE, Lamoreaux WT, MacKay AR, Fairbanks RK, Call JA, Carlson JD, Ling BC, Demakas JJ, Cooke BS, Lee CM (2014) Clinical outcomes following salvage Gamma Knife radiosurgery for recurrent glioblastoma. World J Clin Oncol 5:142–148. https://doi.org/10.5306/wjco.v5.i2.142
Patel M, Siddiqui F, Jin JY, Mikkelsen T, Rosenblum M, Movsas B, Ryu S (2009) Salvage reirradiation for recurrent glioblastoma with radiosurgery: radiographic response and improved survival. J Neurooncol 92:185–191. https://doi.org/10.1007/s11060-008-9752-9
Torcuator RG, Thind R, Patel M, Mohan YS, Anderson J, Doyle T, Ryu S, Jain R, Schultz L, Rosenblum M, Mikkelsen T (2010) The role of salvage reirradiation for malignant gliomas that progress on bevacizumab. J Neurooncol 97:401–407. https://doi.org/10.1007/s11060-009-0034-y
Kim EY, Yechieli R, Kim JK, Mikkelsen T, Kalkanis SN, Rock J, Rosenblum M, Ryu S (2014) Patterns of failure after radiosurgery to two different target volumes of enhancing lesions with and without FLAIR abnormalities in recurrent glioblastoma multiforme. J Neurooncol 116:291–297. https://doi.org/10.1007/s11060-013-1290-4
Benedict SH, Yenice KM, Followill D, Galvin JM, Hinson W, Kavanagh B, Keall P, Lovelock M, Meeks S, Papiez L, Purdie T, Sadagopan R, Schell MC, Salter B, Schlesinger DJ, Shiu AS, Solberg T, Song DY, Stieber V, Timmerman R, Tome WA, Verellen D, Wang L, Yin FF (2010) Stereotactic body radiation therapy: the report of AAPM Task Group 101. Med Phys 37:4078–4101. https://doi.org/10.1118/1.3438081
Wen PY, Macdonald DR, Reardon DA, Cloughesy TF, Sorensen AG, Galanis E, Degroot J, Wick W, Gilbert MR, Lassman AB, Tsien C, Mikkelsen T, Wong ET, Chamberlain MC, Stupp R, Lamborn KR, Vogelbaum MA, van den Bent MJ, Chang SM (2010) Updated response assessment criteria for high-grade gliomas: response assessment in neuro-oncology working group. J Clin Oncol 28:1963–1972. https://doi.org/10.1200/JCO.2009.26.3541
Cabrera AR, Cuneo KC, Desjardins A, Sampson JH, McSherry F, Herndon JE 2nd, Peters KB, Allen K, Hoang JK, Chang Z, Craciunescu O, Vredenburgh JJ, Friedman HS, Kirkpatrick JP (2013) Concurrent stereotactic radiosurgery and bevacizumab in recurrent malignant gliomas: a prospective trial. Int J Radiat Oncol Biol Phys 86:873–879. https://doi.org/10.1016/j.ijrobp.2013.04.029
Gutin PH, Iwamoto FM, Beal K, Mohile NA, Karimi S, Hou BL, Lymberis S, Yamada Y, Chang J, Abrey LE (2009) Safety and efficacy of bevacizumab with hypofractionated stereotactic irradiation for recurrent malignant gliomas. Int J Radiat Oncol Biol Phys 75:156–163. https://doi.org/10.1016/j.ijrobp.2008.10.043
Fetcko K, Lukas RV, Watson GA, Zhang L, Dey M (2017) Survival and complications of stereotactic radiosurgery: a systematic review of stereotactic radiosurgery for newly diagnosed and recurrent high-grade gliomas. Medicine (Baltimore) 96:e8293. https://doi.org/10.1097/MD.0000000000008293
Le Rhun E, Dhermain F, Vogin G, Reyns N, Metellus P (2016) Radionecrosis after stereotactic radiotherapy for brain metastases. Expert Rev Neurother 16:903–914. https://doi.org/10.1080/14737175.2016.1184572
Levin VA, Bidaut L, Hou P, Kumar AJ, Wefel JS, Bekele BN, Grewal J, Prabhu S, Loghin M, Gilbert MR, Jackson EF (2011) Randomized double-blind placebo-controlled trial of bevacizumab therapy for radiation necrosis of the central nervous system. Int J Radiat Oncol Biol Phys 79:1487–1495. https://doi.org/10.1016/j.ijrobp.2009.12.061
Abbassy M, Missios S, Barnett GH, Brewer C, Peereboom DM, Ahluwalia M, Neyman G, Chao ST, Suh JH, Vogelbaum MA (2018) Phase I trial of radiosurgery dose escalation plus bevacizumab in patients with recurrent/progressive glioblastoma. Neurosurgery 83:385–392. https://doi.org/10.1093/neuros/nyx369
Cuneo KC, Vredenburgh JJ, Sampson JH, Reardon DA, Desjardins A, Peters KB, Friedman HS, Willett CG, Kirkpatrick JP (2012) Safety and efficacy of stereotactic radiosurgery and adjuvant bevacizumab in patients with recurrent malignant gliomas. Int J Radiat Oncol Biol Phys 82:2018–2024. https://doi.org/10.1016/j.ijrobp.2010.12.074
Tsien C, Pugh S, Dicker AP, Raizer JJ, Matuszak MM, Lallana E, Huang J, Algan O, Taylor N, Portelance L, Villano J, Hamm J, Oh KS, Ali AN, Kim MM, Lindhorst S, Mehta MP (2019) Randomized phase II trial of re-irradiation and concurrent bevacizumab versus bevacizumab alone as treatment for recurrent glioblastoma (NRG Oncology/RTOG 1205): initial outcomes and RT plan quality report [abstract]. Int J Radiat Oncol Biol Phys 105:S78. https://doi.org/10.1016/j.ijrobp.2019.06.539
Funding
Funding was provided by the Hermelin Brain Tumor Center, the Department of Neurosurgery at Henry Ford Health System and funds raised with help of the Head for the Cure Foundation.
Author information
Authors and Affiliations
Contributions
Study design and oversight: SR, MSS, TW, TM. Manuscript composition and editing: DB, TW, SR, TM. Statistical analysis: LS. Collection of data: DB, AM, MS, MSS. Data analysis: TW, JS, SR, DB, AM. Approval and review of submitted manuscript: DB, AM, LS, JS, TM, MS, SR, MSS, TW.
Corresponding author
Ethics declarations
Conflict of interest:
The authors declare there are no conflicts of interest. ICMJE forms have been signed by all authors.
Ethical approval:
This study was approved by our institutional review board (HFHS-C # 11–01).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bergman, D., Modh, A., Schultz, L. et al. Randomized prospective trial of fractionated stereotactic radiosurgery with chemotherapy versus chemotherapy alone for bevacizumab-resistant high-grade glioma. J Neurooncol 148, 353–361 (2020). https://doi.org/10.1007/s11060-020-03526-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-020-03526-4