Abstract
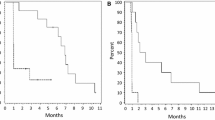
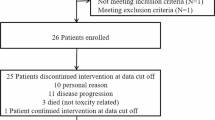
Antiangiogenic therapies for malignant gliomas often result in transient response, and recurrent disease is characterized by adoption of invasive and hypoxic phenotype. The notch signaling pathway is activated in gliomas, and augments cell migration and hypoxic response. Here we report a clinical study of the combination of bevacizumab and RO4929097, an inhibitor of the notch signaling cascade. A phase I clinical trial was conducted through the Adult Brain Tumor Consortium in subjects with recurrent malignant glioma. Primary objectives were to assess safety and to define the maximum tolerated dose of RO4929097 in combination with bevacizumab. Secondary objectives were to determine overall survival, progression free survival, radiographic response, pharmacokinetic evaluation, and tissue biomarker analysis. Thirteen subjects were enrolled. Of the three subjects treated with the highest dose of RO4929097, one grade 3 toxicity and one grade 2 toxicity were observed. Definitive maximum tolerated dose of RO4929097 in combination with bevacizumab was not identified due to manufacturer’s decision to halt drug production. 2 of 12 evaluable subjects demonstrated radiographic response; one subject experienced CR and the second PR. The median overall survival was 10.9 months with a median progression-free survival of 3.7 months. Two subjects remained free of disease progression at 6 months from treatment initiation. PK evaluation did not identify clinically significant drug–drug interactions. All analyzed tissue specimens revealed activation of notch signaling. Combination of RO4929097 and bevacizumab was well-tolerated. Given the compelling scientific rationale, additional studies of antiangiogenic and notch signaling inhibitors should be considered.



Similar content being viewed by others
References
Wen PY, Kesari S (2008) Malignant gliomas in adults. N Engl J Med 359:492–507. doi:10.1056/NEJMra0708126
Bolos V, Grego-Bessa J, de la Pompa JL (2007) Notch signaling in development and cancer. Endocr Rev 28:339–363. doi:10.1210/er.2006-0046
Bray SJ (2006) Notch signalling: a simple pathway becomes complex. Nat Rev Mol Cell Biol 7:678–689. doi:10.1038/nrm2009
Nefedova Y, Gabrilovich D (2008) Mechanisms and clinical prospects of Notch inhibitors in the therapy of hematological malignancies. Drug Resist Updat 11:210–218. doi:10.1016/j.drup.2008.09.002
Fan X, Matsui W, Khaki L, Stearns D, Chun J, Li YM, Eberhart CG (2006) Notch pathway inhibition depletes stem-like cells and blocks engraftment in embryonal brain tumors. Cancer Res 66:7445–7452. doi:10.1158/0008-5472.CAN-06-0858
Aster JC, Pear WS, Blacklow SC (2008) Notch signaling in leukemia. Annu Rev Pathol 3:587–613. doi:10.1146/annurev.pathmechdis.3.121806.154300
Staal FJ, Langerak AW (2008) Signaling pathways involved in the development of T-cell acute lymphoblastic leukemia. Haematologica 93:493–497. doi:10.3324/haematol.12917
Chen Y, De Marco MA, Graziani I, Gazdar AF, Strack PR, Miele L, Bocchetta M (2007) Oxygen concentration determines the biological effects of NOTCH-1 signaling in adenocarcinoma of the lung. Cancer Res 67:7954–7959. doi:10.1158/0008-5472.CAN-07-1229
Konishi J, Kawaguchi KS, Vo H, Haruki N, Gonzalez A, Carbone DP, Dang TP (2007) Gamma-secretase inhibitor prevents Notch3 activation and reduces proliferation in human lung cancers. Cancer Res 67:8051–8057. doi:10.1158/0008-5472.CAN-07-1022
Peacock CD, Watkins DN (2008) Cancer stem cells and the ontogeny of lung cancer. J Clin Oncol 26:2883–2889. doi:10.1200/JCO.2007.15.2702
Kanamori M, Kawaguchi T, Nigro JM, Feuerstein BG, Berger MS, Miele L, Pieper RO (2007) Contribution of Notch signaling activation to human glioblastoma multiforme. J Neurosurg 106:417–427. doi:10.3171/jns.2007.106.3.417
Shih AH, Holland EC (2006) Notch signaling enhances nestin expression in gliomas. Neoplasia 8:1072–1082. doi:10.1593/neo.06526
Zeng Q, Li S, Chepeha DB, Giordano TJ, Li J, Zhang H, Polverini PJ, Nor J, Kitajewski J, Wang CY (2005) Crosstalk between tumor and endothelial cells promotes tumor angiogenesis by MAPK activation of Notch signaling. Cancer Cell 8:13–23. doi:10.1016/j.ccr.2005.06.004
Li JL, Sainson RC, Oon CE, Turley H, Leek R, Sheldon H, Bridges E, Shi W, Snell C, Bowden ET, Wu H, Chowdhury PS, Russell AJ, Montgomery CP, Poulsom R, Harris AL (2011) DLL4-Notch signaling mediates tumor resistance to anti-VEGF therapy in vivo. Cancer Res 71:6073–6083. doi:10.1158/0008-5472.CAN-11-1704
Soeda A, Hara A, Kunisada T, Yoshimura S, Iwama T, Park DM (2015) The evidence of glioblastoma heterogeneity. Sci Rep 5:7979. doi:10.1038/srep07979
Park DM, Li J, Okamoto H, Akeju O, Kim SH, Lubensky I, Vortmeyer A, Dambrosia J, Weil RJ, Oldfield EH, Park JK, Zhuang Z (2007) N-CoR pathway targeting induces glioblastoma derived cancer stem cell differentiation. Cell Cycle 6:467–470
Wu J, Wiegand R, LoRusso P, Li J (2011) Validation and implementation of a liquid chromatography/tandem mass spectrometry assay for quantitation of the total and unbound RO4929097, a gamma-secretase inhibitor targeting Notch signaling, in human plasma. J Chromatogr B Analyt Technol Biomed Life Sci 879:1537–1543. doi:10.1016/j.jchromb.2011.03.045
Shah VP, Midha KK, Findlay JW, Hill HM, Hulse JD, McGilveray IJ, McKay G, Miller KJ, Patnaik RN, Powell ML, Tonelli A, Viswanathan CT, Yacobi A (2000) Bioanalytical method validation—a revisit with a decade of progress. Pharm Res 17:1551–1557
Gabrielsson J (1994) Pharmacokinetic/pharmacodynamic data analysis: concepts and applications. Swedish Pharmaceutical Press, Uppsala
Lacey LF, Keene ON, Pritchard JF, Bye A (1997) Common noncompartmental pharmacokinetic variables: are they normally or log-normally distributed? J Biopharm Stat 7:171–178. doi:10.1080/10543409708835177
Mizuta E, Tsubotani A (1985) Preparation of mean drug concentration–time curves in plasma. A study on the frequency distribution of pharmacokinetic parameters. Chem Pharm Bull (Tokyo) 33:1620–1632
Sahlgren C, Gustafsson MV, Jin S, Poellinger L, Lendahl U (2008) Notch signaling mediates hypoxia-induced tumor cell migration and invasion. Proc Natl Acad Sci USA 105:6392–6397. doi:10.1073/pnas.0802047105
Bolos V, Mira E, Martinez-Poveda B, Luxan G, Canamero M, Martinez AC, Manes S, de la Pompa JL (2013) Notch activation stimulates migration of breast cancer cells and promotes tumor growth. Breast Cancer Res 15:R54. doi:10.1186/bcr3447
Gustafsson MV, Zheng X, Pereira T, Gradin K, Jin S, Lundkvist J, Ruas JL, Poellinger L, Lendahl U, Bondesson M (2005) Hypoxia requires notch signaling to maintain the undifferentiated cell state. Dev Cell 9:617–628. doi:10.1016/j.devcel.2005.09.010
Qiang L, Wu T, Zhang HW, Lu N, Hu R, Wang YJ, Zhao L, Chen FH, Wang XT, You QD, Guo QL (2012) HIF-1alpha is critical for hypoxia-mediated maintenance of glioblastoma stem cells by activating Notch signaling pathway. Cell Death Differ 19:284–294. doi:10.1038/cdd.2011.95
Androutsellis-Theotokis A, Leker RR, Soldner F, Hoeppner DJ, Ravin R, Poser SW, Rueger MA, Bae SK, Kittappa R, McKay RD (2006) Notch signalling regulates stem cell numbers in vitro and in vivo. Nature 442:823–826. doi:10.1038/nature04940
Soeda A, Park M, Lee D, Mintz A, Androutsellis-Theotokis A, McKay R, Engh J, Iwama T, Kunisada T, Kassam A (2009) Hypoxia promotes expansion of the CD133-positive glioma stem cells through activation of HIF-1α. Oncogene 28:3949–3959
Bar EE, Lin A, Mahairaki V, Matsui W, Eberhart CG (2010) Hypoxia increases the expression of stem-cell markers and promotes clonogenicity in glioblastoma neurospheres. Am J Pathol 177:1491–1502. doi:10.2353/ajpath.2010.091021
Hovinga KE, Shimizu F, Wang R, Panagiotakos G, Van Der Heijden M, Moayedpardazi H, Correia AS, Soulet D, Major T, Menon J, Tabar V (2010) Inhibition of notch signaling in glioblastoma targets cancer stem cells via an endothelial cell intermediate. Stem cells 28:1019–1029. doi:10.1002/stem.429
Villa JC, Chiu D, Brandes AH, Escorcia FE, Villa CH, Maguire WF, Hu CJ, de Stanchina E, Simon MC, Sisodia SS, Scheinberg DA, Li YM (2014) Nontranscriptional role of Hif-1alpha in activation of gamma-secretase and notch signaling in breast cancer. Cell Rep 8:1077–1092. doi:10.1016/j.celrep.2014.07.028
Diaz-Padilla I, Hirte H, Oza AM, Clarke BA, Cohen B, Reedjik M, Zhang T, Kamel-Reid S, Ivy SP, Hotte SJ, Razak AA, Chen EX, Brana I, Wizemann M, Wang L, Siu LL, Bedard PL (2013) A phase Ib combination study of RO4929097, a gamma-secretase inhibitor, and temsirolimus in patients with advanced solid tumors. Invest New Drugs 31:1182–1191. doi:10.1007/s10637-013-0001-5
Tolcher AW, Messersmith WA, Mikulski SM, Papadopoulos KP, Kwak EL, Gibbon DG, Patnaik A, Falchook GS, Dasari A, Shapiro GI, Boylan JF, Xu ZX, Wang K, Koehler A, Song J, Middleton SA, Deutsch J, Demario M, Kurzrock R, Wheler JJ (2012) Phase I study of RO4929097, a gamma secretase inhibitor of Notch signaling, in patients with refractory metastatic or locally advanced solid tumors. J Clin Oncol 30:2348–2353. doi:10.1200/JCO.2011.36.8282
Shih T, Lindley C (2006) Bevacizumab: an angiogenesis inhibitor for the treatment of solid malignancies. Clin Ther 28:1779–1802. doi:10.1016/j.clinthera.2006.11.015
Sahebjam S, Bedard PL, Castonguay V, Chen Z, Reedijk M, Liu G, Cohen B, Zhang WJ, Clarke B, Zhang T, Kamel-Reid S, Chen H, Ivy SP, Razak AR, Oza AM, Chen EX, Hirte HW, McGarrity A, Wang L, Siu LL, Hotte SJ (2013) A phase I study of the combination of ro4929097 and cediranib in patients with advanced solid tumours (PJC-004/NCI 8503). Br J Cancer 109:943–949. doi:10.1038/bjc.2013.380
Xu R, Shimizu F, Hovinga K, Beal K, Karimi S, Droms L, Peck KK, Gutin P, Iorgulescu JB, Kaley T, DeAngelis L, Pentsova E, Nolan C, Grommes C, Chan T, Bobrow D, Hormigo A, Cross JR, Wu N, Takebe N, Panageas K, Ivy P, Supko JG, Tabar V, Omuro A (2016) Molecular and clinical effects of Notch inhibition in glioma patients: a phase 0/I trial. Clin Cancer Res. doi:10.1158/1078-0432.CCR-16-0048
Funding
CA UM1 137443 awarded to ABTC.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
Timothy F. Cloughesy has been compensated by Roche/Genentech for consulting work and expert testimony. No other authors report conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pan, E., Supko, J.G., Kaley, T.J. et al. Phase I study of RO4929097 with bevacizumab in patients with recurrent malignant glioma. J Neurooncol 130, 571–579 (2016). https://doi.org/10.1007/s11060-016-2263-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-016-2263-1