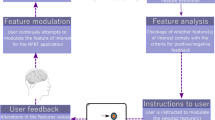
Abstract
The pursuit of effective motion rehabilitation devices has been a prominent focus of research in recent decades. However, the efficacy of such devices relies heavily on their ability to induce motor learning. Thus, understanding the neuroscientific principles underlying motor learning is crucial. This paper highlights a significant number of studies investigating the roles of various brain regions such as the basal ganglia, cerebellum, and motor cortex in motor learning, either individually or in interactive processes. However, due to the theoretical nature of many proposed ideas, definitive conclusions about acceptable brain interactive mechanisms facilitating motor learning are challenging. Addressing the lack of a comprehensive review paper to scrutinize and compare these hypotheses, identify weaknesses, and offer new directions for researchers, this study provides a theoretical perspective review. Excluding works solely focused on neurophysiological connections or computational models, it categorizes selected papers into topics related to the contributions of basal ganglia, cerebellum, motor/sensory cortex, and super-learning mechanisms in motor learning. The analysis suggests that concepts emphasizing super-learning hypotheses and information transmission mechanisms offer valuable insights into the processes underlying motor learning, warranting greater attention for designing rehabilitation interventions. Nonetheless, further experimental evidence is necessary to validate these hypotheses.
Similar content being viewed by others
Data availability
My manuscript has no associated data.
References
Papale AE, Hooks BM. Circuit changes in motor cortex during motor skill learning. Neuroscience. 2018;368:283-97.
Hwang EJ, Dahlen JE, Hu YY, Aguilar K, Yu B, Mukundan M, et al. Disengagement of motor cortex from movement control during long-term learning. Science advances. 2019;5(10):eaay0001.
Makino H, Ren C, Liu H, Kim AN, Kondapaneni N, Liu X, et al. Transformation of cortex-wide emergent properties during motor learning. Neuron. 2017;94(4):880-90. e8.
Leisman G, Braun-Benjamin O, Melillo R. Cognitive-motor interactions of the basal ganglia in development. Frontiers in systems neuroscience. 2014;8:16.
Granato G, Cartoni E, Da Rold F, Mattera A, Baldassarre G. A Computational Model of Representation Learning in the Brain Cortex, Integrating Unsupervised and Reinforcement Learning. arXiv preprint arXiv:210603688. 2021.
Bédard P, Sanes JN. Basal ganglia-dependent processes in recalling learned visual-motor adaptations. Experimental brain research. 2011;209:385-93.
Zhang W, Guo L, Liu D. Concurrent interactions between prefrontal cortex and hippocampus during a spatial working memory task. Brain Structure and Function. 2022;227(5):1735-55.
Kitazawa S, Kimura T, Yin P-B. Cerebellar complex spikes encode both destinations and errors in arm movements. Nature. 1998;392(6675):494-7.
Streng ML, Popa LS, Ebner TJ. Climbing fibers control Purkinje cell representations of behavior. Journal of Neuroscience. 2017;37(8):1997-2009.
Hull C. Prediction signals in the cerebellum: beyond supervised motor learning. elife. 2020;9:e54073.
Herrojo Ruiz M, Maudrich T, Kalloch B, Sammler D, Kenville R, Villringer A, et al. Modulation of neural activity in frontopolar cortex drives reward-based motor learning. Scientific reports. 2021;11(1):20303.
Wang JX, Kurth-Nelson Z, Kumaran D, Tirumala D, Soyer H, Leibo JZ, et al. Prefrontal cortex as a meta-reinforcement learning system. Nature neuroscience. 2018;21(6):860-8.
Ito M, Doya K. Multiple representations and algorithms for reinforcement learning in the cortico-basal ganglia circuit. Current opinion in neurobiology. 2011;21(3):368-73.
Gershman SJ, Moustafa AA, Ludvig EA. Time representation in reinforcement learning models of the basal ganglia. Frontiers in computational neuroscience. 2014;7:194.
Rosahl SK, Knight RT. Role of prefrontal cortex in generation of the contingent negative variation. Cerebral Cortex. 1995;5(2):123-34.
Bostan AC, Strick PL. The basal ganglia and the cerebellum: nodes in an integrated network. Nature Reviews Neuroscience. 2018;19(6):338-50.
Milardi D, Quartarone A, Bramanti A, Anastasi G, Bertino S, Basile GA, et al. The cortico-basal ganglia-cerebellar network: past, present and future perspectives. Frontiers in systems neuroscience. 2019;13:61.
Spampinato DA, Celnik PA, Rothwell JC. Cerebellar–motor cortex connectivity: one or two different networks? Journal of Neuroscience. 2020;40(21):4230-9.
Bukhari Q, Ruf SF, Guell X, Whitfield-Gabrieli S, Anteraper S. Interaction between cerebellum and cerebral cortex, evidence from dynamic causal modeling. The Cerebellum. 2022;21(2):225-33.
O'Doherty JP, Dayan P, Friston K, Critchley H, Dolan RJ. Temporal difference models and reward-related learning in the human brain. Neuron. 2003;38(2):329-37.
Cacciola A, Milardi D, Livrea P, Flace P, Anastasi G, Quartarone A. The known and missing links between the cerebellum, basal ganglia, and cerebral cortex. The Cerebellum. 2017;16:753-5.
Mori F, Okada K-i, Nomura T, Kobayashi Y. The pedunculopontine tegmental nucleus as a motor and cognitive interface between the cerebellum and basal ganglia. Frontiers in neuroanatomy. 2016;10:109.
Hintzen A, Pelzer EA, Tittgemeyer M. Thalamic interactions of cerebellum and basal ganglia. Brain Structure and Function. 2018;223:569-87.
Nozaradan S, Schwartze M, Obermeier C, Kotz SA. Specific contributions of basal ganglia and cerebellum to the neural tracking of rhythm. Cortex. 2017;95:156-68.
Djurfeldt M, Ekeberg Ö, Graybiel AM. Cortex–basal ganglia interaction and attractor states. Neurocomputing. 2001;38:573-9.
Hélie S, Ell SW, Ashby FG. Learning robust cortico-cortical associations with the basal ganglia: an integrative review. Cortex. 2015;64:123-35.
Gaffan D, Easton A, Parker A. Interaction of inferior temporal cortex with frontal cortex and basal forebrain: double dissociation in strategy implementation and associative learning. Journal of Neuroscience. 2002;22(16):7288-96.
Walz A, Doppl K, Kaza E, Roschka S, Platz T, Lotze M. Changes in cortical, cerebellar and basal ganglia representation after comprehensive long term unilateral hand motor training. Behavioural brain research. 2015;278:393-403.
Ash H, Chang A, Ortiz RJ, Kulkarni P, Rauch B, Colman R, et al. Structural and functional variations in the prefrontal cortex are associated with learning in pre-adolescent common marmosets (Callithrix jacchus). Behavioural Brain Research. 2022;430:113920.
Akkal D, Dum RP, Strick PL. Supplementary motor area and presupplementary motor area: targets of basal ganglia and cerebellar output. Journal of Neuroscience. 2007;27(40):10659-73.
Luft AR, Manto M-U, Oulad Ben Taib N. Modulation of motor cortex excitability by sustained peripheral stimulation: the interaction between the motor cortex and the cerebellum. The Cerebellum. 2005;4:90-6.
Tanaka Y, Fujimura N, Tsuji T, Maruishi M, Muranaka H, Kasai T. Functional interactions between the cerebellum and the premotor cortex for error correction during the slow rate force production task: an fMRI study. Experimental brain research. 2009;193:143-50.
Kelly AC, Uddin LQ, Biswal BB, Castellanos FX, Milham MP. Competition between functional brain networks mediates behavioral variability. Neuroimage. 2008;39(1):527-37.
Gerraty RT, Davidow JY, Foerde K, Galvan A, Bassett DS, Shohamy D. Dynamic flexibility in striatal-cortical circuits supports reinforcement learning. Journal of Neuroscience. 2018;38(10):2442-53.
Yoo AH, Collins AG. How working memory and reinforcement learning are intertwined: A cognitive, neural, and computational perspective. Journal of cognitive neuroscience. 2022;34(4):551-68.
Granato G, Cartoni E, Da Rold F, Mattera A, Baldassarre G. Integrating unsupervised and reinforcement learning in human categorical perception: A computational model. Plos one. 2022;17(5):e0267838.
Todorov DI, Capps RA, Barnett WH, Latash EM, Kim T, Hamade KC, et al. The interplay between cerebellum and basal ganglia in motor adaptation: A modeling study. PLoS One. 2019;14(4):e0214926.
Caligiore D, Arbib MA, Miall RC, Baldassarre G. The super-learning hypothesis: Integrating learning processes across cortex, cerebellum and basal ganglia. Neuroscience & Biobehavioral Reviews. 2019;100:19-34.
Funding
No funding was received for conducting this study.
Author information
Authors and Affiliations
Contributions
Conceptualization, A.H.M.T, S.H, and H.K.; writing—original draft preparation, A.H.M.T, S.H, N.D, M.G, A.A, and H.K.; writing—review and editing, A.H.M.T and H.K.; supervision, A.H.M.T and H.K. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare no conflict of interest.
Ethics approval
The authors declare that this review article adheres to ethical standards and guidelines, including proper citation and attribution of sources. As this is just a review of other papers does not need other regulations regarding human and animal subjects.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Torbati, A.H.M., Jami, S., Kobravi, H. et al. Underlying interactive neural mechanism of motor learning governed by the cerebellum, the basal ganglia, and motor/sensory cortex: a review from theoretical perspective. Neurosci Behav Physi (2024). https://doi.org/10.1007/s11055-024-01583-0
Published:
DOI: https://doi.org/10.1007/s11055-024-01583-0