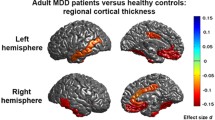
Objectives. To identify the structural features of the gray matter of the cerebral cortex in patients with depression at clinical high risk of psychosis. Materials and methods. Nineteen right-handed male patients with juvenile depression who met the criteria for a high risk of psychosis, along with 20 sex- and age-matched mentally healthy subjects as a control group, underwent clinical and MRI investigations. T1-weighted images were processed in FreeSurfer 7.1.1 to obtain average cortical gray matter thickness, subcortical volume, and amygdalar nucleus volume for each subject. Between-group comparisons were made and correlations with psychometric measures (SOPS, HDRS) were calculated. Results. Lower cortical thickness was found in patients in the left (p = 0.002) and right (p = 0.003) postcentral gyrus, along with greater thickness in the right posterior cingulate cortex (p = 0.003) and the anterior part of the anterior cingulate cortex (p = 0.001). Conclusions. This picture may reflect changes in the cerebral cortex at the early stages of the endogenous process, including reductions in gray matter in some areas and changes in the opposite direction in others (a relationship between the latter and altered ontogenesis and/or certain compensatory changes cannot be excluded).
Similar content being viewed by others
References
Fusar-Poli, P., “The clinical high-risk state for psychosis (CHR-P), Version, II,” Schizophr. Bull., 43, No. 1, 44–47 (2017).
Fusar-Poli, P., Cappucciati, M., Rutigliano, G., et al., “Towards a standard psychometric diagnostic interview for subjects at ultra high risk of psychosis: CAARMS versus SIPS,” Psychiatry J., 2016, 1–11 (2016).
Woods, S. W., Miller, T. J., and McGlashan, T. H., “The ‘prodromal’ patient: both symptomatic and at-risk,” CNS Spectr., 6, No. 3, 223–232 (2001).
Lencz, T., Smith, C. W., Authera, A., et al., “Nonspecific and attenuated negative symptoms in patients at clinical high-risk for schizophrenia,” Schizophr. Res., 68, No. 1, 37–48 (2004).
Gross, G., “Prodrome und Vorpostensyndrome schizophrener Erkrankungen,” in: Schizophrenie und Zyklothymie. Ergebnisse und Probleme, Gross, G. (ed.), Thieme, Stuttgart (1969).
Kaleda, V. G. and Omelchenko, M. A., “Dynamics and questions of prognosis of juvenile depression with attenuated schizophrenia spectrum symptoms,” Obozr. Psikhiatr. Med. Psikhol., No. 1, 42–52 (2021).
Girolamo, G., McGorry, P., and Sartorius, N. (eds.), Age of Onset of Mental Disorders: Etiopathogenetic and Treatment Implications Springer, Cham, Switzerland (2019).
Bardenshtein, L. M. and Aleshkina, G. A., “Depressive and manic disorders in the structure of the first psychotic episode in adolescence and young adulthood,” Zh. Nevrol. Psikhiatr., 115, No. 1–2, 3–6 (2015).
Del Re, E. C., Stone, W. S., Bouix, S., et al., “Baseline cortical thickness reductions in clinical high risk for psychosis: Brain regions associated with conversion to psychosis versus non-conversion as assessed at one-year follow-up in the Shanghai-At-Risk-for-Psychosis (SHARP) Study,” Schizophr. Bull., 47, No. 2, 562–574 (2021).
Jalbrzikowski, M., Hayes, R. A., Wood, S. J., et al., “Association of structural magnetic resonance imaging measures with psychosis onset in individuals at clinical high risk for developing psychosis: An ENIGMA Working Group mega-analysis,” JAMA Psychiatry, 78, No. 7, 753–766 (2021).
Liloia, D., Brasso, C., Cauda, F., et al., “Updating and characterizing neuroanatomical markers in high-risk subjects, recently diagnosed and chronic patients with schizophrenia: A revised coordinate-based meta-analysis,” Neurosci. Biobehav. Rev., 123, 83–103 (2021).
Tomyshev, A. S., Lebedeva, I. S., Akhadov, T. A., et al., “Alterations in white matter microstructure and cortical thickness in individuals at ultra-high risk of psychosis: A multimodal tractography and surface-based morphometry study,” Psychiatry Res. Neuroimaging, 289, 26–36 (2019).
Tomyshev, A. S., Lebedeva, I. S., Omelchenko, M. A., and Kaleda, V. G., “ MRI markers of changes in the supragranular layer of the cerebral cortex in patients at clinically high risk of developing endogenous psychoses,” Byull. Eksperim. Biol. Med., 171, No. 4, 500–506 (2021).
Zhao, Y., Zhang, Q., Shah, C., et al., “Cortical thickness abnormalities at different stages of the illness course in schizophrenia: A systematic review and meta-analysis,” JAMA Psychiatry, 79, No. 6, 560–570 (2022).
Malla, A., Shah, J., Iyer, S., et al., “Youth mental health should be a top priority for health care in Canada,” Can. J. Psychiatry, 63, No. 4, 216–222 (2018).
Zimmerman, M., Martinez, J. H., Young, D., et al., “Severity classification on the Hamilton Depression Rating Scale,” J. Affect. Disord., 150, No. 2, 384–388 (2013).
Miller, T. J., McGlashan, T. H., Woods, S. W., et al., “Symptom assessment in schizophrenic prodromal states,” Psychiatr. Q., 70, No. 4, 273–287 (1999).
Fischl, B., “FreeSurfer,” NeuroImage, 62, No. 2, 774–781 (2012).
Desikan, R. S., Segonne, F., Fischl, B., et al., “An automated labeling system for subdividing the human cerebral cortex on MRI scans into gyral based regions of interest,” NeuroImage, 31, No. 3, 968–980 (2006).
Saygin, Z. M., Kliemann, D., Iglesias, J. E., et al., “High-resolution magnetic resonance imaging reveals nuclei of the human amygdala: manual segmentation to automatic atlas,” NeuroImage, 155, 370–382 (2017).
Benjamini, Y. and Hochberg, Y., “On the adaptive control of the false discovery rate in multiple testing with independent statistics,” J. Educat. Behav. Stat., 25, No. 1, 60–83 (2016).
Kropf, E., Syan, S. K., Minuzzi, L., and Frey, B. N., “From anatomy to function: the role of the somatosensory cortex in emotional regulation,” Rev. Brasil. Psiquiatria, 41, No. 3, 261–269 (2019).
Binnewies, J., Nawijn, L., Brandmaier, A. M., et al., “Associations of depression and regional brain structure across the adult lifespan: Pooled analyses of six population-based and two clinical cohort studies in the European Lifebrain consortium,” NeuroImage Clin., 36, 103180 (2022).
Schmaal, L., Hibar, D. P., Samann, P. G., et al., “Cortical abnormalities in adults and adolescents with major depression based on brain scans from 20 cohorts worldwide in the ENIGMA Major Depressive Disorder Working Group,” Mol. Psychiatry, 22, No. 6, 900–909 (2017).
Opel, N., Goltermann, J., Hermesdorf, M., et al., “Cross-disorder analysis of brain structural abnormalities in six major psychiatric disorders: A secondary analysis of mega- and meta-analytical findings from the ENIGMA Consortium,” Biol. Psychiatry, 88, No. 9, 678–686 (2020).
Frangou, S., Modabbernia, A., Williams, S. C. R., et al., “Cortical thickness across the lifespan: Data from 17,075 healthy individuals aged 3–90 years,” Hum. Brain Mapp., 43, No. 1, 431–451 (2022).
Deng, W., Addington, J., Bearden, C. E., et al., “Depression predicts global functional outcomes in individuals at clinical high risk for psychosis,” Psychiatr. Res. Clin. Pract., 3, No. 4, 163–171 (2021).
Woodberry, K. A., Shapiro, D. I., Bryant, C., and Seidman, L. J., “Progress and future directions in research on the psychosis prodrome: A review for clinicians,” Harv. Rev. Psychiatry, 24, No. 2, 87–103 (2016).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Zhurnal Nevrologii i Psikhiatrii imeni S. S. Korsakova, Vol. 123, No. 6, pp. 94–99, June, 2023.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Dudina, A.N., Tomyshev, A.S., Omelchenko, M.A. et al. Structural Features of the Brain in Juvenile Depression with Clinical High Risk of Psychosis. Neurosci Behav Physi 54, 16–21 (2024). https://doi.org/10.1007/s11055-024-01562-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11055-024-01562-5