Abstract
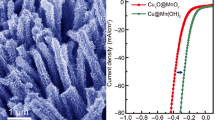
Developing a facile strategy for the construction of advanced electrocatalysts to accelerate oxygen evolution kinetics to meet the needs of new energy conversion technologies is highly desirable. Herein, a novel and facile route is devised to grow the amorphous RuxOy on CuO/Cu2O nanowire arrays. Experimental results show that the typical product displays an outstanding electrocatalytic oxygen evolution activity involving a low overpotential of 236 mV at 20 mA cm−2 and an excellent durability. The reasons for which is associated with the synergistic effect of amorphous RuxOy and CuO/Cu2O nanowire arrays, as well as the existence of abundant oxygen vacancies. This study exhibits new insights into using the composite of amorphous RuxOy and copper (Cu)–based oxides as high-activity catalysts for oxygen evolution.






Similar content being viewed by others
References
Anantharaj S, Reddy PN, Kundu S (2017) Core-oxidized amorphous cobalt phosphide nanostructures: an advanced and highly efficient oxygen evolution catalyst. Inorg Chem 56:1742–1756
Chen D, Dong CL, Zou Y, Su D, Huang YC, Tao L, Dou S, Shen S, Wang S (2017a) In situ evolution of highly dispersed amorphous CoOx clusters for oxygen evolution reaction. Nanoscale 9:11969–11975
Chen H, Gao Y, Sun L (2017b) Highly active three-dimensional NiFe/Cu2O nanowires/Cu foam electrode for water oxidation. Chemsuschem 10:1475–1481
Deng Y, Handoko AD, Du Y, Xi S, Yeo BS (2016) In situ Raman spectroscopy of copper and copper oxide surfaces during electrochemical oxygen evolution reaction: identification of Cu-III oxides as catalytically active species. ACS Catal 6:2473–2481
Dong C et al (2017) Modest oxygen-defective amorphous manganese-based nanoparticle mullite with superior overall electrocatalytic performance for oxygen reduction reaction. Small 13:1603903
Du Z, Wang Y, Li J, Liu J (2019) Facile fabrication of Pt-Ni alloy nanoparticles supported on reduced graphene oxide as excellent electrocatalysts for hydrogen evolution reaction in alkaline environment. J Nanopart Res 21:13
Ferrero GA, Preuss K, Fuertes AB, Sevilla M, Titirici MM (2016) The influence of pore size distribution on the oxygen reduction reaction performance in nitrogen doped carbon microspheres. J Mater Chem A 4:2581–2589
Gu Y, Wang Y, An W, Men Y, Rui Y, Fan X, Li B (2019) A novel strategy to boost the oxygen evolution reaction activity of NiFe-LDHs with in situ synthesized 3D porous reduced graphene oxide matrix as both the substrate and electronic carrier. New J Chem 43:6555–6562
Handoko AD, Deng S, Deng Y, Cheng AWF, Chan KW, Tan HR, Pan Y, Tok ES, Sow CH, Yeo BS (2016) Enhanced activity of H2O2-treated copper(II) oxide nanostructures for the electrochemical evolution of oxygen. Catal Sci Technol 6:269–274
He D, Wang G, Liu G, Suo H, Zhao C (2017) Construction of leaf-like CuO–Cu2O nanocomposites on copper foam for high-performance supercapacitors. Dalton T 46:3318–3324
He D, Xing S, Sun B, Cai H, Suo H, Zhao C (2016) Design and construction of three-dimensional flower-like CuO hierarchical nanostructures on copper foam for high performance supercapacitor. Electrochim Acta 210:639–645
Hou C-C, Fu W-F, Chen Y (2016) Self-supported Cu-based nanowire arrays as noble-metal-free electrocatalysts for oxygen evolution. Chemsuschem 9:2069–2073
Hu F, Zhu S, Chen S, Li Y, Ma L, Wu T, Zhang Y, Wang C, Liu C, Yang X, Song L, Yang X, Xiong Y (2017) Amorphous metallic NiFeP: a conductive bulk material achieving high activity for oxygen evolution reaction in both alkaline and acidic media. Adv Mater 29:1606570
Jin L, Zheng X, Liu W, Cao L, Cao Y, Yao T, Wei S (2017) Integration of plasmonic and amorphous effects in MoO3−x spheres for efficient photoelectrochemical water oxidation. J Mater Chem A 5:12022–12026
Kuang M, Li TT, Chen H, Zhang SM, Zhang LL, Zhang YX (2015) Hierarchical Cu2O/CuO/Co3O4 core-shell nanowires: synthesis and electrochemical properties. Nanotechnology 26:304002
Kulkarni AK, Panmand RP, Sethi YA, Kadam SR, Patil DR, Ghule AV, Kale BB (2018) 3D hierarchical heterostructures of Bi2W1-xMoxO6 with enhanced oxygen evolution reaction from water under natural sunlight. New J Chem 42:17597–17605
Li N, Xia WY, Wang J, Liu ZL, Li QY, Chen SZ, Xu CW, Lu XH (2015) Manganese oxides supported on hydrogenated TiO2 nanowire array catalysts for the electrochemical oxygen evolution reaction in water electrolysis. J Mater Chem A 3:21308–21313
Liu J et al (2018) The flexibility of an amorphous cobalt hydroxide nanomaterial promotes the electrocatalysis of oxygen evolution reaction. Small 14:1703514
Lv C, Yan C, Chen G, Ding Y, Sun J, Zhou Y, Yu G (2018) An amorphous noble-metal-free electrocatalyst enables N2 fixation under ambient conditions. Angew Chem 130:6181–6184
Qi J et al (2015) Porous nickel-iron oxide as a highly efficient electrocatalyst for oxygen evolution reaction. Adv Sci 2:1500199
Ren X, Ji X, Wei Y, Wu D, Zhang Y, Ma M, Liu Z, Asiri AM, Wei Q, Sun X (2018) In situ electrochemical development of copper oxide nanocatalysts within a TCNQ nanowire array: a highly conductive electrocatalyst for the oxygen evolution reaction. Chem Commun 54:1425–1428
Salvatore DA, Peña B, Dettelbach KE, Berlinguette CP (2016) Photodeposited ruthenium dioxide films for oxygen evolution reaction electrocatalysis. J Mater Chem A 5:1575–1580
Shan Z, Archana PS, Shen G, Gupta A, Bakker MG, Pan S (2015) NanoCOT: low-cost nanostructured electrode containing carbon, oxygen, and titanium for efficient oxygen evolution reaction. J Am Chem Soc 137:11996–12005
Siracusano S, Van Dijk N, Payne-Johnson E, Baglio V, Arico AS (2015) Nanosized IrOx and IrRuOx electrocatalysts for the O2 evolution reaction in PEM water electrolysers. Appl Catal B-Environ 164:488–495
Wu Z, Li P, Qin Q, Li Z, Liu X (2018) N-doped graphene combined with alloys (NiCo, CoFe) and their oxides as multifunctional electrocatalysts for oxygen and hydrogen electrode reactions. Carbon 139:35–44
Wu Z, Nie D, Song M, Jiao T, Fu G, Liu X (2019) Facile synthesis of Co-Fe-B-P nanochains as an efficient bifunctional electrocatalyst for overall water-splitting. Nanoscale 11:7506–7512
Yang M-Q, Wang J, Wu H, Ho GW (2018) Noble metal-free nanocatalysts with vacancies for electrochemical water splitting. Small 14:1703323
Yin J, Li Y, Lv F, Lu M, Sun K, Wang W, Wang L, Cheng F, Li Y, Xi P, Guo S (2017) Oxygen vacancies dominated NiS2/CoS2 interface porous nanowires for portable Zn–air batteries driven water splitting devices. Adv Mater 29:1704681
Yu X, Zhang X, Wang S, Feng G (2015) A computational study on water adsorption on Cu2O(111) surfaces: the effects of coverage and oxygen defect. Appl Surf Sci 343:33–40
Zhang R, El-Refaei SM, Russo PA, Pinna N (2018) Metal phosphonate coordination networks and frameworks as precursors of electrocatalysts for the hydrogen and oxygen evolution reactions. J Nanopart Res 20:146
Funding
This work was supported by the National Natural Science Foundation of China (51908567) and the China Postdoctoral Science Foundation (2018M643311).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOC 2714 kb)
Rights and permissions
About this article
Cite this article
Yuan, W., Wu, Y., Wang, X. et al. Constructing amorphous RuxOy on CuO/Cu2O nanowire arrays for improved oxygen evolution. J Nanopart Res 22, 125 (2020). https://doi.org/10.1007/s11051-020-04859-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-020-04859-x