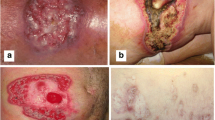
Abstract
Chromoblastomycosis (CBM) is a chronic, progressive fungal disease of the skin and subcutaneous tissue caused by a group of dematiaceous fungi. Verrucous lesions present parasite-rich granulomas and predominance of a Th2 patterns of cytokines. The inflammasome constitutes a macromolecular protein complex that play a role in the activation of caspase 1 that cleaves pro-IL1β and pro-IL18, essential mediators of inflammation, and also activates pyroptosis. We intended to explore the presence and a possible role of inflammasome elements in cutaneous human lesions in CBM, considering the expression of IL1β, IL18, caspase 1, NLRP1, and also RIPK3, a key downstream component of necroptosis signaling. 35 skin biopsies of cutaneous lesions of verrucous form of CBM and 10 biopsies from normal skin were selected. The diagnosis was based on histological and clinical analysis. An immunohistochemical protocol was performed. The histopathological analysis evidenced epidermis with hyperkeratosis, irregular acanthosis, and micro abscesses. The dermis presented suppurative granulomas and inflammatory infiltrate composed by giant cells, macrophages, epithelioid cells, lymphocytes, and some eosinophils. Positive cells were distributed in the inflammatory infiltrate, with an increased number of cells expressing caspase 1, IL1β and IL18. Cells expressing RIPK3 and NLRP1 were less frequent. The intense presence of caspase 1, IL1β and IL18, allied to NLRP1 expression, suggest that inflammasome and pyroptosis could play a role in the immune response against fungal agents of CBM. Our results, allied to data from literature, could suggest that inflammasome-mediated response and pyroptosis could be a target to be explored to decrease CBM lesions.



Similar content being viewed by others
References
Santos DWCL, de Azevedo CMPES, Vicente VA, Queiroz-Telles F, Rodrigues AM, de Hoog GS, et al. The global burden of chromoblastomycosis. PLoS Negl Trop Dis. 2021;12(15):e0009611. https://doi.org/10.1371/journal.pntd.0009611.
Silva JP, de Souza W, Rozental S. Chromoblastomycosis: a retrospective study of 325 cases on Amazonic Region (Brazil). Mycopathologia. 1998–1999;143:171–5. https://doi.org/10.1023/a:1006957415346.
Queiroz-Telles F, Nucci M, Colombo AL, Tobón A, Restrepo A. Mycoses of implantation in Latin America: an overview of epidemiology, clinical manifestations, diagnosis and treatment. Med Mycol. 2011;49:225–36. https://doi.org/10.3109/13693786.2010.539631.
Queiroz-Telles F, de Hoog S, Santos DW, Salgado CG, Vicente VA, Bonifaz A, et al. Chromoblastomycosis. Clin Microbiol Rev. 2017;30:233–76. https://doi.org/10.1128/CMR.00032-16.
Queiroz-Telles F, Esterre P, Perez-Blanco M, Vitale RG, Salgado CG, Bonifaz A. Chromoblastomycosis: an overview of clinical manifestations, diagnosis and treatment. Med Mycol. 2009;47:3–15. https://doi.org/10.1080/13693780802538001.
d’Avila SC, Pagliari C, Duarte MI. The cell-mediated immune reaction in the cutaneous lesion of chromoblastomycosis and their correlation with different clinical forms of the disease. Mycopathologia. 2003;156(2):51–60. https://doi.org/10.1023/a:1022948329193.
Silva AA, Criado PR, Nunes RS, da Silva WL, Kanashiro-Galo L, Duarte MI, et al. In situ immune response in human chromoblastomycosis—a possible role for regulatory and Th17 T cells. PLoS Negl Trop Dis. 2014;8:e3162. https://doi.org/10.1371/journal.pntd.0003162.
Martinon F, Burns K, Tschopp J. The inflammasome: a molecular platform triggering activation of inflammatory caspases and processing of pro IL-beta. Mol Cell. 2002;10:417–26. https://doi.org/10.1016/s1097-2765(02)00599-3.
Siqueira IM, Wüthrich M, Li M, Wang H, Las-Casas LO, de Castro RJA, et al. Early immune response against Fonsecaea pedrosoi requires Dectin-2-mediated Th17 activity, whereas Th1 response, aided by Treg cells, is crucial for fungal clearance in later stage of experimental chromoblastomycosis. PLoS Negl Trop Dis. 2020;14(6):e0008386. https://doi.org/10.1371/journal.pntd.0008386.
Li W, Kashiwamura S, Ueda H, Sekiyama A, Okamura H. Protection of CD8+ T cells from activation-induced cell death by IL-18. J Leukoc Biol. 2007;82(1):142–51. https://doi.org/10.1189/jlb.0706431.
Bueter CL, Lee CK, Rathinam VAK, Healy GJ, Taron CH, Specht CA, Levitz SM. Chitosan but not chitin activates the inflammasome by a mechanism dependent upon phagocytosis. J Biol Chem. 2011;286(41):35447–55. https://doi.org/10.1074/jbc.M111.274936.
Marty-Roix R, Vladimer GI, Pouliot K, Weng D, Buglione-Corbett R, West K, et al. Identification of QS-21 as an inflammasome-activating molecular component of saponin adjuvants. J Biol Chem. 2016;291(3):1123–36. https://doi.org/10.1074/jbc.M115.683011.
Wallace HL, Wang L, Gardner CL, Corkum CP, Grant MD, Hirasawa K, Russell RS. Crosstalk between pyroptosis and apoptosis in hepatitis C virus-induced cell death. Front Immunol. 2022;13:788138. https://doi.org/10.3389/fimmu.2022.788138.
Ivanov K, Garanina E, Rizvanov A, Khaiboullina S. Inflammasomes as targets for adjuvants. Pathogens. 2020;9(4):252. https://doi.org/10.3390/pathogens9040252.
Martynova E, Rizvanov A, Urbanowicz RA, Khaiboullina S. Inflammasome contribution to the activation of Th1, Th2, and Th17 immune responses. Front Microbiol. 2022;13:851835. https://doi.org/10.3389/fmicb.2022.851835.
Ketelut-Carneiro N, Silva GK, Rocha FA, Milanezi CM, Cavalcanti-Neto FF, Zamboni DS, et al. IL-18 triggered by the NLRP3 inflammasome induces host innate resistance in a pulmonary model of fungal infection. J Immunol. 2015;194:4507–17. https://doi.org/10.4049/jimmunol.1402321.
Van de Veerdonk FL, Joosten LAB, Shaw PJ, Smeekens SP, Malireddi RKS, Van der Meer JWM, et al. The inflammasome drives protective Th1 and Th17 cellular responses in disseminated candidiasis. Eur J Immunol. 2011;41(8):2260–8. https://doi.org/10.1002/eji.201041226.
Hise AG, Tomalka J, Ganesan S, Patel K, Hall BA, Brown GD, et al. An essential role for the NLRP3 inflammasome in host defense against the human fungal pathogen Candida albicans. Cell Host Microbe. 2009;5:487–97. https://doi.org/10.1016/j.chom.2009.05.002.
Kapplusch F, Schulze F, Rabe-Matschewsky S, Russ S, Herbig M, Heymann MC, et al. CASP1 variants influence subcellular caspase-1 localization, pyroptosome formation, pro-inflammatory cell death and macrophage deformability. Clin Immunol. 2019;208:108232. https://doi.org/10.1016/j.clim.2019.06.008.
Cookson BT, Brennan MA. Pro-inflammatory programmed cell death. Trends Microbiol. 2001;9:113–4. https://doi.org/10.1016/s0966-842x(00)01936-3.
Lamkanfi M, Dixit VM. Mechanisms and functions of inflammasomes. Cell. 2014;157:1013–22. https://doi.org/10.1016/j.cell.2014.04.007.
Man SM, Karki R, Kanneganti TD. Molecular mechanisms and functions of pyroptosis, inflammatory caspases and inflammasomes in infectious diseases. Immunol Rev. 2017;277(1):61–75. https://doi.org/10.1111/imr.12534.
Dhuriya YK, Sharma D. Necroptosis: a regulated inflammatory mode of cell death. J Neuroinflammation. 2018;15:199. https://doi.org/10.1186/s12974-018-1235-0.
Guo H, Kaiser WJ, Mocarski ES. Manipulation of apoptosis and necroptosis signaling by herpesviruses. Med Microbiol Immunol. 2015;204:439–48. https://doi.org/10.1007/s00430-015-0410-5.
Shlomovitz I, Zargrian S, Gerlic M. Mechanisms of RIPK3-induced inflammation. Immunol Cell Biol. 2017;95:166–72. https://doi.org/10.1038/icb.2016.124.
Lamkanfi M, Malireddi RKS, Kanneganti T-D. Fungal zymosan and mannan activate the cryopyrin inflammasome. J Biol Chem. 2009;284:20574–81. https://doi.org/10.1074/jbc.M109.023689.
Vonk AG, Netea MG, van Krieken JH, Iwakura Y, van der Meer JWM, Kullberg BJ. Endogenous interleukin (IL)–1α and IL-1β Are crucial for host defense against disseminated candidiasis. J Infect Dis. 2006;193:1419–26. https://doi.org/10.1086/503363.
Karki R, Man SM, Malireddi RKS, Gurung P, Vogel P, Lamkanfi M, et al. Concerted activation of the AIM2 and NLRP3 inflammasomes orchestrates host protection against Aspergillus infection. Cell Host Microbe. 2015;17:357–68. https://doi.org/10.1016/j.chom.2015.01.006.
Briard B, Malireddi RKS, Kanneganti T-D. Role of inflammasomes/pyroptosis and PANoptosis during fungal infection. PLoS Pathog. 2021;17(3):e1009358. https://doi.org/10.1371/journal.ppat.1009358.
Castro RJA, Siqueira IM, Jerônimo MS, Basso AMM, Veloso PHH Jr, Magalhães KG, et al. The major chromoblastomycosis etiologic agent Fonsecaea pedrosoi activates the NLRP3 inflammasome. Front Immunol. 2017;8:1572. https://doi.org/10.3389/fimmu.2017.01572.
Brito AC, Bittencourt MJS. Chromoblastomycosis: an etiological, epidemiological, clinical, diagnostic, and treatment update. An Bras Dermatol. 2018;93(4):495–506. https://doi.org/10.1590/abd1806-4841.20187321.
Silva AAL, Criado PR, Nunes RS, Kanashiro-Galo L, Seixas Duarte MI, Sotto MN, Pagliari C. Langerhans cells express IL-17A in the epidermis of chromoblastomycosis lesions. Biomed Hub. 2017;2(2):1–8. https://doi.org/10.1159/000477954.
Joosten LA, Netea MG, Dinarello CA. Interleukin-1beta in innate inflammation, autophagy and immunity. Semin Immunol. 2013;25:416–24. https://doi.org/10.1016/j.smim.2013.10.018.
Dinarello CA, Novick D, Kim S, Kaplanski G. Interleukin-18 and IL-18 binding protein. Front Immunol. 2013;4:289. https://doi.org/10.3389/fimmu.2013.00289.
Silva LM, Sousa JR, Hirai KE, Dias LB Jr, Furlaneto IP, Carneiro FRO, et al. The inflammasome in leprosy skin lesions: an immunohistochemical evaluation. Infect Drug Resist. 2018;11:2231–40. https://doi.org/10.2147/IDR.S172806.
Pontillo A, Laurentino W, Crovella S, Pereira AC. NLRP1 haplotypes associated with leprosy in Brazilian patients. Infect Genet Evol. 2013;19:274–9. https://doi.org/10.1016/j.meegid.2013.06.006.
Chavarría-Smith J, Vance RE. The NLRP1 inflammasomes. Immunol Rev. 2015;265(1):22–34. https://doi.org/10.1111/imr.12283.
Awad F, Assrawi E, Louvrier C, Jumeau C, Georgin-Lavialle S, Grateau G, et al. Inflammasome biology, molecular pathology and therapeutic implications. Pharmacol Ther. 2018;187:133–49. https://doi.org/10.1016/j.pharmthera.2018.02.011.
Evavold CL, Kagan JC. How inflammasomes inform adaptive immunity. J Mol Biol. 2018;430(2):217–37. https://doi.org/10.1016/j.jmb.2017.09.019.
Williams TJ, Gonzales-Huerta LE, Armstrong-James D. Fungal-induced programmed cell death. J Fungi (Basel). 2021;7:231. https://doi.org/10.3390/jof7030231.
Gong W, Shi Y, Ren J. Research progresses of molecular mechanism of pyroptosis and its related diseases. Immunobiology. 2020;225:151884. https://doi.org/10.1016/j.imbio.2019.11.019.
Tavares AH, Burgel PH, Bocca AL. Turning up the heat: inflammasome activation by fungal pathogens. PLoS Pathog. 2015;11:e1004948. https://doi.org/10.1371/journal.ppat.1004948.
Wellington M, Koselny K, Sutterwala FS, Krysan DJ. Candida albicans triggers NLRP3-mediated pyroptosis in macrophages. Eukaryot Cell. 2014;13:329–40. https://doi.org/10.1128/EC.00336-13.
Lian H, Fang XL, Li Q, Liu S, Wei Q, Hua X, et al. NLRP3 inflammasome-mediated pyroptosis pathway contributes to the pathogenesis of Candida albicans keratitis. Front Med. 2022. https://doi.org/10.3389/fmed.2022.845129.
Acknowledgements
CP—Research productivity grant of National Council for scientific and Technological Development (CNPq).
Funding
The authors declare there is no funding to the research. CP receives a research productivity grant of National Council for Scientific and Technological Development (CNPq).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. CP: conceptualization, project administration, methodology, writing. LK-G: methodology, review. MNS: formal analysis, review.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical approval
Ethical approval was waived by the local Ethics Committee of University of São Paulo, Medical School in view of the retrospective nature of the study and all the procedures being performed were part of the routine care. The use of the material that constituted the casuistic was approved under the number 0317/11.
Additional information
Handling Editor: Anamelia Lorenzetti Bocca.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pagliari, C., Kanashiro-Galo, L. & Sotto, M.N. Inflammasome and Inflammatory Programmed Cell Death in Chromoblastomycosis. Mycopathologia 188, 63–70 (2023). https://doi.org/10.1007/s11046-022-00679-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11046-022-00679-w