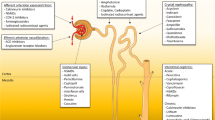
Abstract
The main goal of the treatment of patients is its effectiveness and safety. However, all currently prescribed drugs being used also have certain adverse effects, which might be seen as an unavoidable but necessary cost of pharmacotherapy. The kidney is the primary organ for xenobiotics elimination, making it particularly susceptible to the harmful effects of drugs and their metabolites during their excretion from the body. Moreover, certain medications have a preferential nephrotoxicity potential, which means that using them increases the risk of kidney injury. Drug nephrotoxicity is, therefore, both a significant problem and a complication of pharmacotherapy. It should be noted that, there is presently no accepted definition of drug-induced nephrotoxicity and no established diagnostic criteria. The current review briefly describes the pathogenic mechanism of drug-induced nephrotoxicity, the various basic drugs with nephrotoxicity potential and the renal biomarkers for the treatment of the drug-related kidney damage.

Similar content being viewed by others
Data Availability
Not applicable.
Abbreviations
- BUN:
-
Blood urea nitrogen
- PCT:
-
Proximal convoluted tubule
- NSAIDs:
-
Non-steroidal anti-inflammatory drugs
- ATN:
-
Acute tubular necrosis
- GFR:
-
Glomerular filtration rate
- FSGS:
-
Focal segmental glomerulosclerosis
- RTA:
-
Renal tubular acidosis
- CIN:
-
Contrast induced nephropathy
References
Ferguson MA, Vaidya VS, Bonventre JV (2008) Biomarkers of nephrotoxic acute kidney injury. Toxicology 245(3):182–193. https://doi.org/10.1016/j.tox.2007.12.024
Finn WF, Porter GA (2003) Urinary biomarkers and nephrotoxicity. Clinical nephrotoxins. Springer, Dordrecht, pp 621–655. https://doi.org/10.1007/1-4020-2586-6_33
Galley HF (2000) Can acute renal failure be prevented?. J R Coll Surg Edinb, 45(1)
Kohli HS, Bhaskaran MC, Muthukumar T, Thennarasu K, Sud K, Jha V, Gupta KL, Sakhuja V (2000) Treatment-related acute renal failure in the elderly: a hospital-based prospective study. Nephrol dialysis transplantation 15(2):212–217. https://doi.org/10.1093/ndt/15.2.212
Moisi MI, Bungau SG, Vesa CM, Diaconu CC, Behl T, Stoicescu M, Toma MM, Bustea C, Sava C, Popescu MI (2021) Framing cause-effect relationship of acute coronary syndrome in patients with chronic kidney disease. Diagnostics 11(8):1518
Naughton CA (2008) Drug-induced nephrotoxicity. Am Family Phys 78(6):743–750
Pazhayattil GS, Shirali AC (2014) Drug-induced impairment of renal function. Int J Nephrol renovascular disease 7:457
Rached E, Hoffmann D, Blumbach K, Weber K, Dekant W, Mally A (2008) Evaluation of putative biomarkers of nephrotoxicity after exposure to ochratoxin a in vivo and in vitro. Toxicol Sci 103(2):371–381. https://doi.org/10.1093/toxsci/kfn040
Kirtane AJ, Leder DM, Waikar SS, Chertow GM, Ray KK, Pinto DS, Karmpaliotis D, Burger AJ, Murphy SA, Cannon CP, Braunwald E (2005) Serum blood urea nitrogen as an independent marker of subsequent mortality among patients with acute coronary syndromes and normal to mildly reduced glomerular filtration rates. J Am Coll Cardiol 45(11):1781–1786
BAIDOG A, BUNGAU S, BEHL T, RATIU I, URSU ARONRAC, F., LAZAR, L. and, VESA CM (2020) INTERRELATIONSHIPS BETWEEN HYPERURICEMIA, METABOLIC SYNDROME AND CHRONIC KIDNEY DISEASE IN PATIENTS WITH DIABETES MELLITUS
Kumar A Fausto, Pathological basis of disease 2007;9: 417
Choudhury D, Ahmed Z (2006) Drug-associated renal dysfunction and injury. Nat Clin Pract Nephrol 2(2):80–91. https://doi.org/10.1038/ncpneph0076
Schneider V, Lévesque LE, Zhang B, Hutchinson T, Brophy JM (2006) Association of selective and conventional nonsteroidal antiinflammatory drugs with acute renal failure: a population-based, nested case-control analysis. Am J Epidemiol 164(9):881–889. https://doi.org/10.1093/aje/kwj331
Markowitz GS, Perazella MA (2005) Drug-induced renal failure: a focus on tubulointerstitial disease. Clin Chim Acta 351(1–2):31–47. https://doi.org/10.1016/j.cccn.2004.09.005
Schnellmann RG, Kelly KJ (1999) Pathophysiology of nephrotoxic acute renal failure. Acute Ren Fail Phila Pa : Blackwell Sci, pp.1–14
Perazella MA (2005) Drug-induced nephropathy: an update. Exp Opin Drug Saf 4(4):689–706
Pisoni R, Ruggenenti P, Remuzzi G (2001) Drug-induced thrombotic microangiopathy. Drug Saf 24(7):491–501. https://doi.org/10.2165/00002018-200124070-00002
Rossert J (2001) Drug-induced acute interstitial nephritis. Kidney Int 60(2):804–817
Fored CM, Ejerblad E, Lindblad P, Fryzek JP, Dickman PW, Signorello LB, Lipworth L, Elinder CG, Blot WJ, McLaughlin JK, Zack MM (2001) Acetaminophen, aspirin, and chronic renal failure. N Engl J Med 345(25):1801–1808
Rodríguez-Iturbe B, García GG (2010) The role of tubulointerstitial inflammation in the progression of chronic renal failure. Nephron Clin Pract 116(2):c81–c88
Coco TJ, Klasner AE (2004) Drug-induced rhabdomyolysis. Curr Opin Pediatr 16(2):206–210
Huerta-Alardín AL, Varon J, Marik PE (2004) Bench-to-bedside review: rhabdomyolysis–an overview for clinicians. Crit Care 9(2):1–12. https://doi.org/10.1186/cc2978
Vanholder R, Sever MS, Erek E, Lameire N, Rhabdomyolysis (2000) J Am Soc Nephrol, 11(8), 1553–1561. https://doi.org/10.1681/ASN.V1181553
Schoolwerth AC, Sica DA, Ballermann BJ, Wilcox CS (2001) Renal considerations in angiotensin converting enzyme inhibitor therapy: a statement for healthcare professionals from the Council on the kidney in Cardiovascular Disease and the Council for high blood pressure research of the American Heart Association. Circulation 104(16):1985–1991. https://doi.org/10.1161/hc4101.096153
Palmer BF (2002) Renal dysfunction complicating the treatment of hypertension. N Engl J Med 347(16):1256–1261
D’Agati V (2003) March. Pathologic classification of focal segmental glomerulosclerosis. Seminars in nephrology, vol 23. WB Saunders, pp 117–134. 2
Jaffe JA, Kimmel PL (2006) Chronic nephropathies of cocaine and heroin abuse: a critical review. Clin J Am Soc Nephrol 1(4):655–667. https://doi.org/10.2215/CJN.00300106
Markowitz GS, Radhakrishnan JAI, Kambham N, Valeri AM, Hines WH, D’AGATI VD (2000) Lithium nephrotoxicity: a progressive combined glomerular and tubulointerstitial nephropathy. J Am Soc Nephrol 11(8):1439–1448. https://doi.org/10.1681/ASN.V1181439
Amoghimath S, Majagi SI (2017) Drug induced kidney disease. Hypertension 347(16):1256–1261
Roderick P, Roth M, Mindell J (2011) Prevalence of chronic kidney disease in England: findings from the 2009 health survey for England. J Epidemiol Community Health 65(Suppl 2):A12–A12
Rosner MH, Okusa MD (2006) Acute kidney injury associated with cardiac surgery. Clin J Am Soc Nephrol 1(1):19–32. https://doi.org/10.2215/CJN.00240605
Bonventre JV, Vaidya VS, Schmouder R, Feig P, Dieterle F (2010) Next-generation biomarkers for detecting kidney toxicity. Nat Biotechnol 28(5):436–440. https://doi.org/10.1038/nbt0510-436
Shao C, Li M, Li X, Wei L, Zhu L, Yang F, Jia L, Mu Y, Wang J, Guo Z, Zhang D (2011) A tool for biomarker discovery in the urinary proteome: a manually curated human and animal urine protein biomarker database. Mol Cell Proteom, 10(11)
Guder WG, Hofmann W (1992) Markers for the diagnosis and monitoring of renal tubular lesions. Clin Nephrol 38:3–7
Prinsen BH, der Velden MG, Kaysen GA, Straver HW, van Rijn HJ, Stellaard F, Berger R, Rabelink TJ (2001) Transferrin synthesis is increased in nephrotic patients insufficiently to replace urinary losses. J Am Soc Nephrol, 12, 1017–1025
Tencer J, Bakoush O, Torffvit O (2000) Diagnostic and prognostic significance of proteinuria selectivity index in glomerular diseases. Clin Chim Acta 297(1–2):73–83
Herget-Rosenthal S, Poppen D, Husing J, Marggraf G, Pietruck F, Jakob HG, Philipp T, Kribben A (2004) Prognostic value of tubular proteinuria and enzymuria in nonoliguric acute tubular necrosis. Clin Chem 50(3):552–558. https://doi.org/10.1373/clinchem.2003.027763
Emeigh Hart SG, Lavin A, Bounous DI, Macgregor JT, Harpur E (2005) Assessment of renal injury in vivo: traditional and novel biomarkers. Toxicol Sci 84:2–3
Trof RJ, Di Maggio F, Leemreis J, Groeneveld AJ (2006) Biomarkers of acute renal injury and renal failure. Shock 26(3):245–253
Gorriz JL, Martinez-Castelao A (2012) Proteinuria: detection and role in native renal disease progression. Transplantation reviews 26(1):3–13. https://doi.org/10.1016/j.trre.2011.10.002
Alchi B, Nishi S, Kondo D, Kaneko Y, Matsuki A, Imai N, Ueno M, Iguchi S, Sakatsume M, Narita I, Yamamoto T (2005) Osteopontin expression in acute renal allograft rejection. Kidney Int 67(3):886–896. https://doi.org/10.1111/j.1523-1755.2005.00153.x
Vaidya VS, Ferguson MA, Bonventre JV (2008) Biomarkers of acute kidney injury. Annu Rev Pharmacol Toxicol 48:463–493
Bailly V, Zhang Z, Meier W, Cate R, Sanicola M, Bonventre JV (2002) Shedding of kidney injury molecule-1, a putative adhesion protein involved in renal regeneration. J Biol Chem 277(42):39739–39748
Sales GTM, Foresto RD (2020) Drug-induced nephrotoxicity. Revista da Associação Médica Brasileira 66:s82–s90
Htike NL, Santoro J, Gilbert B, Elfenbein IB, Teehan G (2012) Biopsy-proven vancomycin-associated interstitial nephritis and acute tubular necrosis. Clin Exp Nephrol 16(2):320–324. https://doi.org/10.1007/s10157-011-0559-1
van Hal SJ, Paterson DL, Lodise TP (2013) Systematic review and meta-analysis of vancomycin-induced nephrotoxicity associated with dosing schedules that maintain troughs between 15 and 20 milligrams per liter. Antimicrob Agents Chemother 57(2):734–744. https://doi.org/10.1128/AAC.01568-12
Öktem F, Arslan MK, Ozguner F, Candir Ö, Yilmaz HR, Ciris M, Uz E (2005) In vivo evidences suggesting the role of oxidative stress in pathogenesis of vancomycin-induced nephrotoxicity: protection by erdosteine. Toxicology 215(3):227–233. https://doi.org/10.1016/j.tox.2005.07.009
Bird ST, Etminan M, Brophy JM, Hartzema AG, Delaney JA (2013) Risk of acute kidney injury associated with the use of fluoroquinolones. CMAJ 185(10):E475–E482. https://doi.org/10.1503/cmaj.121730
Stratta P, Lazzarich E, Canavese C, Bozzola C, Monga G (2007) Ciprofloxacin crystal nephropathy. Am J Kidney Dis 50(2):330–335. https://doi.org/10.1053/j.ajkd.2007.05.014
Justo JA, Bosso JA (2015) Adverse reactions associated with systemic polymyxin therapy. Pharmacotherapy: The Journal of Human Pharmacology and Drug Therapy 35(1):28–33. https://doi.org/10.1002/phar.1493
Mistro S, Maciel IDM, de Menezes RG, Maia ZP, Schooley RT, Badaró R (2012) Does lipid emulsion reduce amphotericin B nephrotoxicity? A systematic review and meta-analysis. Clin Infect Dis 54(12):1774–1777. https://doi.org/10.1093/cid/cis290
Singh HP, Singh TG, Singh R (2021) Evaluation of the renoprotective effect of syringic acid against nephrotoxicity induced by cisplatin in rats. J Appl Pharm Sci 11(1):080–085
Miller RP, Tadagavadi RK, Ramesh G, Reeves WB (2010) Mechanisms of cisplatin nephrotoxicity. Toxins 2(11):2490–2518. https://doi.org/10.3390/toxins2112490
Pabla N, Murphy RF, Liu K, Dong Z (2009) The copper transporter Ctr1 contributes to cisplatin uptake by renal tubular cells during cisplatin nephrotoxicity. Am J Physiology-Renal Physiol 296(3):F505–F511. https://doi.org/10.1152/ajprenal.90545.2008
Launay-Vacher V, Rey JB, Isnard-Bagnis C, Deray G, Daouphars M (2008) Prevention of cisplatin nephrotoxicity: state of the art and recommendations from the European Society of Clinical Pharmacy Special Interest Group on Cancer Care. Cancer Chemother Pharmacol 61(6):903–909. https://doi.org/10.1007/s00280-008-0711-0
Ciarimboli G, Holle SK, Vollenbröcker B, Hagos Y, Reuter S, Burckhardt G, Bierer S, Herrmann E, Pavenstädt H, Rossi R, Kleta R (2011) New clues for nephrotoxicity induced by ifosfamide: preferential renal uptake via the human organic cation transporter 2. Mol Pharm 8(1):270–279. https://doi.org/10.1021/mp100329u
Widemann BC, Adamson PC (2006) Understanding and managing methotrexate nephrotoxicity. Oncologist 11(6):694–703. https://doi.org/10.1634/theoncologist.11-6-694
Saland J, Leavey PJ, Bash RO, Hansch E, Arbus GS, Quigley R (2002) Effective removal of methotrexate by high-flux hemodialysis. Pediatr Nephrol 17(10):825–829. https://doi.org/10.1007/s00467-002-0946-7
Buchen S, Ngampolo D, Melton RG, Hasan C, Zoubek A, Henze G, Bode U, Fleischhack G (2005) Carboxypeptidase G2 rescue in patients with methotrexate intoxication and renal failure. Br J Cancer 92(3):480–487. https://doi.org/10.1038/sj.bjc.6602337
Murphy SW, Barrett BJ, Parfrey PS (2000) Contrast nephropathy. J Am Soc Nephrol 11(1):177–182. https://doi.org/10.1681/ASN.V111177
Naesens M, Kuypers DR, Sarwal M (2009) Calcineurin inhibitor nephrotoxicity. Clin J Am Soc Nephrol 4(2):481–508. https://doi.org/10.2215/CJN.04800908
Brewster U, Perazella M (2007) Proton pump inhibitors and the kidney: critical. Clin Nephrol 68:65–72
Simpson IJ, Marshall MR, Pilmore H, Manley P, Williams L, Thein H, Voss D (2006) Proton pump inhibitors and acute interstitial nephritis: report and analysis of 15 cases. Nephrology 11(5):381–385. https://doi.org/10.1111/j.1440-1797.2006.00651.x
Chapman MJ, Carrie A (2005) Mechanisms of Statin-Induced Myopathy. Arteriosclerosis, thrombosis, and vascular biology. https://doi.org/10.1161/10.1161/01.ATV.0000194548.11901.a4
van Zyl-Smit R, Firth JC, Duffield M, Marais AD (2004) Renal tubular toxicity of HMG-CoA reductase inhibitors. Nephrol Dialysis Transplantation 19(12):3176–3179. https://doi.org/10.1093/ndt/gfh474
Dickenmann M, Oettl T, Mihatsch MJ (2008) Osmotic nephrosis: acute kidney injury with accumulation of proximal tubular lysosomes due to administration of exogenous solutes. Am J Kidney Dis 51(3):491–503. https://doi.org/10.1053/j.ajkd.2007.10.044
Acknowledgements
The authors are grateful to the Chitkara College of Pharmacy, Chitkara University, Rajpura, Patiala, Punjab, India for providing the necessary facilities to carry out the research work.
Funding
Nil.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Compliance with ethical standards
Not applicable.
Consent to participate
Not applicable.
Competing interests
There are no conflicts of interest.
Credit Author Statement
Conceptualization: Thakur Gurjeet Singh. Analyzed the data: Thakur Gurjeet Singh Wrote the manuscript: Veerta Sharma. Visualization: Thakur Gurjeet Singh Editing of the Manuscript: Veerta Sharma, Thakur Gurjeet Singh Critically reviewed the article: Thakur Gurjeet Singh. Supervision: Thakur Gurjeet Singh. All authors read and approved the final manuscript.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sharma, V., Singh, T.G. Drug induced nephrotoxicity- A mechanistic approach. Mol Biol Rep 50, 6975–6986 (2023). https://doi.org/10.1007/s11033-023-08573-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-023-08573-4