Abstract
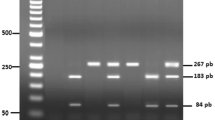
In this study, we aimed to determine the gene polymorphisms of antioxidant enzymes that determine or affect antioxidant activity in the occurrence of the disease and/or complications during and after the surgery in patients who were decided to undergo coronary artery bypass surgery due to coronary artery disease. Blood samples taken before operation in 26 coronary artery patients who were decided to be operated according to the international procedure and the phenol/chloroform method was used to isolate DNA. DNA samples were amplified by using polymerase chain reaction (PCR) method with specific primers for MnSOD, CAT, GPx-3 antioxidant gene regions. As a result of the increasing process, the PCR products for the purpose of determining gene polymorphism, NGOMIV SMA f and BSA I restriction enzymes were used for MNSOD, CAT and GPx-3 gene region, respectively. Allele frequencies were determined and compared by Chi square test. VV (46.15%) and VA (53.85%) genotype for MnSOD region, i TT (22.22%), TC (16.67%) and CC (61.11%) genotype for CAT region, and CC (12.50%), TC (25%) and TT (62.50%) genotypes for GPx-3 region were obtained. While there was no statistically significant significance in terms of genotypes obtained in MnSOD and GPx-3 gene regions (P > 0.05), a significant difference was found in the CAT gene region in terms of genotypes (P < 0.01). Although oxidative stress is important in relation to cardiovascular diseases and postoperative complications, virtually no study of antioxidant enzymes in gene polymorphism are included in the literature. Work is lacking in relation to the subject.

Similar content being viewed by others
References
Grundy SM, Pasternak R, Greenland P, Smith S Jr, Fuster V (1999) Assesment of cardiovascular risk by use of multiple risk factor assessment equation. A statement for healthcare professionals from the American Heart Association and the American College of cardiology. Circulation 100:1481–1492
Harrison D, Griendling KG, Landmesser U, Hornig B, Drexlar H (2003) Role of oxidative stress in atherosclerosis. Am J Cardio 91:7–11
Blankenberg S, Rupprechi MJ, Bickel C, Torzewski M, Hafner G, Tiret L et al (2003) Glutathione peroxidase 1 activity and cardiovascular events in patients with coronary artery disease. N Engl J Med 349(17):1605–1613
Cohn LH (2003) Fifty years of open heart surgery. Circulation 107:2168–2170
Lazar HL (2014) Should off-pump coronary artery bypass surgery be abandoned: a potential solution. J Thorac Cardiovasc Surg 148(6):2475–2476
Barzegar AOM, Sciesser CH, Taylor MK (2014) New reagents for detecting free radicals and oxidative stress. Org Blomol Chem 12(35):6757–6766
Gupta RK, Patel AK, Shah N, Chaudhary AK, Jha UK, Yadav UC, Gupta PK, Pakuval U (2014) Oxidative stress and antioxidants in disease and cancer: a review. Asian Pac Cancer Prev 15(11):4405–4409
Ray PD, Huang BW, Tsuji Y (2012) Reactive oxygen species (ROS) homeostasis and redox regulation in cellular signaling. Cell Signal 24(5):981–990
Trachootham D, Alexandre J, Huang P (2009) Targeting cancer cells by ROS-mediated mechanisms: a radical therapeutic approach? Nat Rev Drug Discov 8:579–591
Andersen JK (2004) Oxidative stress in neurodegeneration: cause or consequence? Nat Med 10:18–25
Shukla V, Mishra SK, Pant HC (2011) Oxidative stress in neurodegeneration. Adv Pharmacol Scl 572–634
Paravicini TM, Touyz RM (2006) Redox signaling in hypertension. Cardiovasc Res 247–258
Haigis MC, Yankner BA (2010) The aging stress response. Mol Cell 40(2):333–344
Ishikawa K, Takenaga K, Akimoto M, Koshikawa N, Yamaguchi A, Imanishi H et al (2008) ROS-generating mitochondrial DNA mutations can regulate tumor cell metastasis. Science 320: 661–664
Pearson TA, Mensah GA, Alexander RW et al (2003) Markers of inflammation and cardiovascular disease: application to clinical and public health practice: a statement for healthcare professionals from the centers for disease control and prevention ant the American Heart Association. Circulation 107(3):499–511
Doğan A, Türker FS (2017) Cardiopulmonary bypass and oxidative stress. Chem Res J 2(6):156–162
Batinic-Haberle I, Reboucas JS, Spasojevich I (2010) Superoxide dismutase mimics: chemistry, pharmacology and therapeutic potential. Antioxid Redox Signal 13:877–918
Ambrosone CB, Freudenheim JL, Thompson PA, Browman E, Vena JE, Marshall JR, Graham S, Laughlin R, Nemoto T, Shields PG (1999) Manganase superoxide dismutase (MnSOD) genetic polymorphisms, dietary antioxidants, risk of breast cancer. Cancer Res 59:602–606
Zhou XF, Cui J, De S Al, Chazaro I, Farrer LA, Manolis AJ, Gavras H, Baldwin CT (2005) Polymorphisms in the promoter region of catalase gene and essential hypertension. Dis Markers 21:3–7
Shuvalova YA, Kaminnyi AI, Meshkov AN, Shirokov RO, Samko AN (2012) Association between polymorphisms of eNOS and GPx-1 genes activity of free-radical processes an in-stent restenosis. Mol Cell Biochem 370:241–249
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: A laboratory manual. 2nd ed. Cold Spring Harbor Laboratory Pres, Cold Spring Harbor. pp 9.16–9.19
Forsberg L, Lyrenas L, Faire UD, Morgenstern R (2001) A common functional C–T substitution polymorphism in the promoter region of the human catalase gene influences transcription factor binding, reporter gene transcription and is correlated to blood catalase levels. Free Radical Biol Med 30(5):500–505
Voetsch B, Jin RC, Bierl C, Benke KS, Kenet G, Simioni P, Ottaviano F, Damasceno BP, Annichino-Bizacchi JM, Handy DE, Loscalzo J (2007) Promoter polymorphisms in the plasma glutathione peroxidase (GPx-3) gene: a novel risk factor for arterial ischemic stroke among young adults and children. Stroke 38(1):41–49
Don RH, Cox PT, Wainwright BJ, Baker K, Mattick JS (1991) Touchdown’ PCR to circumvent spurious priming during gene amplification. Nucl Acids Res 19:4008
Ohata T, Mitsuno M, Yamamura M, Tanaka H, Kobayashi Y, Ryomoto M, Yoshioka Y, Miyomoto Y (2007) Minimal cardiopulmonary bypass attenuates neutrophil activation and cytokine release in coronary artery bypass grafting. J Artif Organs 10:92–95
Karahalil B, Kesimci E, Emerce E, Gumus T, Kanbak O (2011) The impact of OGG1, MTH1 and MnSOD gene polymorphisms on 8-hydroxy-2′-deoxyguanosine and cellular superoxide dismutase activity in myocardial ischemia-reperfusion. Mol Biol Rep 38:2427–2435
Neomoto M, Nishimura R, Sasaki T, Hiki Y, Miyashita Y, Nishioka M, Fujimoto K, Sakuma T, Ohashi T, Fukuda K, Eto Y, Tajima N (2007) Genetic association of glutathione peroxidase-1 with coronary artery calcification in type 2 diabetes: a case control study with multi-slice computed tomography. Cardiovasc Diabetol 6:23–27
Wickremasinghe D, Peiris H, Chandrasena LG, Senaratne V, Perera R (2016) Case control feasibility study assessing the association between severity of coronary artery disease with Glutathione Peroxidase 1 (GPX-1) and GPX-1 polymorphism. BMC Cardiovasc Disord 16:111
Türker FS, Doğan A, Ozan G, Kıbar K, Erışır M (2016) Change in free radical and antioxidant enzyme levels in patients undergoing open heart surgery with cardiopulmonary bypass. Oxid Med Cell Longev 2016:1783728
Venardos KM, Perkins A, Headrick J, Kaye DM (2007) Myocardial ischemia-reperfusion injury, antioxidant enzyme systems, and selenium: a review. Curr Med Chem 14:1539–1549
Acknowledgements
This study was funded by Cumhuriyet Üniversitesi (Grant no 1) and Bitlis Eren Üniversitesi (Grant no 2).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Doğan, A., Özşensoy, Y. & Türker, F.S. MnSOD, CAT and GPx-3 genetic polymorphisms in coronary artery disease. Mol Biol Rep 46, 841–845 (2019). https://doi.org/10.1007/s11033-018-4539-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-018-4539-3