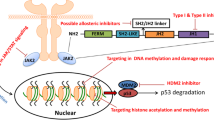
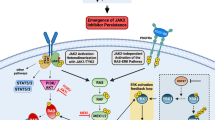
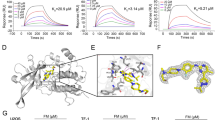
Abstract
The overactivation of Janus kinases 2 (JAK2) by gain-of-function mutations in the JAK2, Myeloproliferative leukemia virus oncogene, or Calreticulin genes are the most important factor in the development of Philadelphia-negative myeloproliferative neoplasms (MPNs). The discovery of the JAK2V617F mutation is a significant breakthrough in understanding the pathogenesis of MPNs, and inhibition of JAK2 abnormal activation has become one of the most effective strategies against MPNs. Currently, three JAK2 inhibitors for treating MPNs have been approved, and several are being evaluated in clinical trials. However, persistent challenges in terms of drug resistance and off-target effects remain unresolved. In this review, we introduce and classify the available JAK2 inhibitors in terms of their mechanisms and clinical considerations. Additionally, through an analysis of target points, binding modes, and structure–activity inhibitor relationships, we propose strategies such as combination therapy and allosteric inhibitors to overcome specific challenges. This review offers valuable insights into current trends and future directions for optimal management of MPNs using JAK2 inhibitors.




Similar content being viewed by others
References
Grinfeld J, Nangalia J, Green AR (2017) Molecular determinants of pathogenesis and clinical phenotype in myeloproliferative neoplasms. Haematologica 102:7–17. https://doi.org/10.3324/haematol.2014.113845
Grinfeld J, Nangalia J, Baxter EJ, Wedge DC, Angelopoulos N, Cantrill R et al (2018) Classification and personalized prognosis in myeloproliferative neoplasms. N Engl J Med 379:1416–1430. https://doi.org/10.1056/NEJMoa1716614
Silvennoinen O, Hubbard SR (2015) Molecular insights into regulation of JAK2 in myeloproliferative neoplasms. Blood 125:3388–3392. https://doi.org/10.1182/blood-2015-01-621110
Bose P, Masarova L, Verstovsek S (2020) Novel concepts of treatment for patients with myelofibrosis and related neoplasms. Cancers 12:2891. https://doi.org/10.3390/cancers12102891
Esposito MT, So CW (2014) DNA damage accumulation and repair defects in acute myeloid leukemia: implications for pathogenesis, disease progression, and chemotherapy resistance. Chromosoma 123:545–561. https://doi.org/10.1007/s00412-014-0482-9
Samuelson Bannow BT, Salit RB, Storer BE, Stevens EA, Wu D, Yeung C et al (2018) Hematopoietic cell transplantation for myelofibrosis: the dynamic international prognostic scoring system plus risk predicts post-transplant outcomes. Biol Blood Marrow Transplant 24:386–392. https://doi.org/10.1016/j.bbmt.2017.09.016
Guglielmelli P, Vannucchi AM (2020) Current management strategies for polycythemia vera and essential thrombocythemia. Blood Rev 42:100714. https://doi.org/10.1016/j.blre.2020.100714
Tefferi A (2018) Primary myelofibrosis: 2019 update on diagnosis, risk-stratification and management. Am J Hematol 93:1551–1560. https://doi.org/10.1002/ajh.25230
McMullin MF, Harrison CN, Ali S, Cargo C, Chen F, Ewing J et al (2019) A guideline for the diagnosis and management of polycythaemia vera. Br J Haematol 184:176–191. https://doi.org/10.1111/bjh.15648
Gangat N, Singh A, Szuber N, Begna K, Elliott M, Wolanskyj-Spinner A et al (2022) Site-specific venous thrombosis in essential thrombocythemia: impact on subsequent vascular events and survival. J Thromb Haemost 20:2439–2443. https://doi.org/10.1111/jth.15834
Venugopal S, Mascarenhas J (2020) Novel therapeutics in myeloproliferative neoplasms. J Hematol Oncol 13:162. https://doi.org/10.1186/s13045-020-00995-y
Guy A, Bidet A, Ling C, Caumont C, Boureau L, Viallard J-F et al (2021) Novel findings of splenic extramedullary hematopoiesis during primary myelofibrosis, post-essential thrombocythemia, and post-polycythemia vera myelofibrosis. Virchows Arch 479:755–764. https://doi.org/10.1007/s00428-021-03110-9
Takenaka K, Shimoda K, Akashi K (2018) Recent advances in the diagnosis and management of primary myelofibrosis. Korean J Intern Med 33:679–690. https://doi.org/10.3904/kjim.2018.033
Santos FP, Tam CS, Kantarjian H, Cortes J, Thomas D, Pollock R et al (2014) Splenectomy in patients with myeloproliferative neoplasms: efficacy, complications and impact on survival and transformation. Leuk Lymphoma 55:121–127. https://doi.org/10.3109/10428194.2013.794269
Pettit K, Odenike O (2017) Novel therapies for myelofibrosis. Curr Hematol Malig Rep 12:611–624. https://doi.org/10.1007/s11899-017-0403-0
Tefferi A, Pardanani A (2015) Myeloproliferative neoplasms a contemporary review. JAMA Oncol 1:97–105. https://doi.org/10.1001/jamaoncol.2015.89
Cervantes F, Pereira A (2012) Prognostication in primary myelofibrosis. Curr Hematol Malig Rep 7:43–49. https://doi.org/10.1007/s11899-011-0102-1
Holmström MO, Hasselbalch HC, Andersen MH (2020) Cancer immune therapy for Philadelphia chromosome-negative chronic myeloproliferative neoplasms. Cancers (Basel) 12:1763. https://doi.org/10.3390/cancers12071763
Geyer HL, Mesa RA (2014) Therapy for myeloproliferative neoplasms: when, which agent, and how? Blood 124:3529–3537. https://doi.org/10.1182/blood-2014-05-577635
Greenfield G, McMullin MF, Mills K (2021) Molecular pathogenesis of the myeloproliferative neoplasms. J Hematol Oncol 14:103. https://doi.org/10.1186/s13045-021-01116-z
How J, Hobbs GS, Mullally A (2019) Mutant calreticulin in myeloproliferative neoplasms. Blood 134:2242–2248. https://doi.org/10.1182/blood.2019000622
Sawyers C, Pikman Y, Lee BH, Mercher T, McDowell E, Ebert BL et al (2006) MPLW515L is a novel somatic activating mutation in myelofibrosis with myeloid metaplasia. PLoS Med 3:270. https://doi.org/10.1371/journal.pmed.0030270
Lucet IS, Fantino E, Styles M, Bamert R, Patel O, Broughton SE et al (2006) The structural basis of Janus kinase 2 inhibition by a potent and specific pan-Janus kinase inhibitor. Blood 107:176–183. https://doi.org/10.1182/blood-2005-06-2413
Xin P, Xu X, Deng C, Liu S, Wang Y, Zhou X et al (2020) The role of JAK/STAT signaling pathway and its inhibitors in diseases. Int Immunopharmacol 80:106210. https://doi.org/10.1016/j.intimp.2020.106210
Gao Q, Liang X, Shaikh AS, Zang J, Xu W, Zhang Y (2018) JAK/STAT signal transduction: promising attractive targets for immune, inflammatory and hematopoietic diseases. Curr Drug Targets 19:487–500. https://doi.org/10.2174/1389450117666161207163054
Meyer SC, Levine RL (2014) Molecular pathways: molecular basis for sensitivity and resistance to JAK kinase inhibitors. Clin Cancer Res 20:2051–2059. https://doi.org/10.1158/1078-0432.CCR-13-0279
Levine RL, Wadleigh M, Cools J, Ebert BL, Wernig G, Huntly BJP et al (2005) Activating mutation in the tyrosine kinase JAK2 in polycythemia vera, essential thrombocythemia, and myeloid metaplasia with myelofibrosis. Cancer Cell 7:387–397. https://doi.org/10.1016/j.ccr.2005.03.023
Kralovics R, Passamonti F, Buser AS, Teo S-S, Tiedt R, Passweg JR et al (2005) A gain-of-function mutation of JAK2 in myeloproliferative disorders. N Engl J Med 352:1779–1790. https://doi.org/10.1056/NEJMoa051113
Baxter EJ, Scott LM, Campbell PJ, East C, Fourouclas N, Swanton S et al (2005) Acquired mutation of the tyrosine kinase JAK2 in human myeloproliferative disorders. Lancet 365:1054–1061. https://doi.org/10.1016/s0140-6736(05)71142-9
Fan W, Cao W, Shi J, Gao F, Wang M, Xu L et al (2023) Contributions of bone marrow monocytes/macrophages in myeloproliferative neoplasms with JAK2(V617F) mutation. Ann Hematol 102:1745–1759. https://doi.org/10.1007/s00277-023-05284-5
Edahiro Y, Araki M, Komatsu N (2020) Mechanism underlying the development of myeloproliferative neoplasms through mutant calreticulin. Cancer Sci 111:2682–2688. https://doi.org/10.1111/cas.14503
Shires KL, Rust AJ, Harryparsad R, Coburn JA, Gopie RE (2023) JAK2/STAT5 pathway mutation frequencies in South African BCR/ABL negative MPN patients. Hematol Oncol Stem Cell Ther 16:291–302. https://doi.org/10.56875/2589-0646.1064
Yang LPH, Keating GM (2012) Ruxolitinib in the treatment of myelofibrosis. Drugs 72:2117–2127. https://doi.org/10.2165/11209340-000000000-00000
Plosker GL (2015) Ruxolitinib: a review of its use in patients with myelofibrosis. Drugs 75:297–308. https://doi.org/10.1007/s40265-015-0351-8
Ostojic A, Vrhovac R, Verstovsek S (2011) Ruxolitinib: a new JAK1/2 inhibitor that offers promising options for treatment of myelofibrosis. Future Oncol 7:1035–1043. https://doi.org/10.2217/fon.11.81
Holik H, Krečak I, Lucijanić M, Samardžić I, Pilipac D, Ljubičić IV et al (2023) Hip and knee osteoarthritis in patients with chronic myeloproliferative neoplasms: a cross-sectional study. Life (Basel) 13:1388. https://doi.org/10.3390/life13061388
Baghdassarian H, Blackstone SA, Clay OS, Philips R, Matthiasardottir B, Nehrebecky M et al (2023) Variant stat4 and response to ruxolitinib in an autoinflammatory syndrome. N Engl J Med 388:2241–2252. https://doi.org/10.1056/NEJMoa2202318
Neumann T, Schneidewind L, Weigel M, Plis A, Vaizian R, Schmidt CA et al (2019) Ruxolitinib for therapy of graft-versus-host disease. Biomed Res Int. https://doi.org/10.1155/2019/8163780
Chovatiya R, Paller AS (2021) JAK inhibitors in the treatment of atopic dermatitis. J Allergy Clin Immunol 148:927–940. https://doi.org/10.1016/j.jaci.2021.08.009
Barraco F, Greil R, Herbrecht R, Schmidt B, Reiter A, Willenbacher W et al (2020) Real-world non-interventional long-term post-authorisation safety study of ruxolitinib in myelofibrosis. Br J Haematol 191:764–774. https://doi.org/10.1111/bjh.16729
Talpaz M, Kiladjian JJ (2021) Fedratinib, a newly approved treatment for patients with myeloproliferative neoplasm-associated myelofibrosis. Leukemia 35:1–17. https://doi.org/10.1038/s41375-020-0954-2
Waksal JA, Tremblay D, Mascarenhas J (2021) Clinical utility of fedratinib in myelofibrosis. Onco Targets Ther 14:4509–4521. https://doi.org/10.2147/OTT.S267001
Palmer J, Mesa R (2020) The role of fedratinib for the treatment of patients with primary or secondary myelofibrosis. Ther Adv Hematol. https://doi.org/10.1177/2040620720925201
Duong VH, Komrokji RS (2014) The role of pacritinib in the management of myelofibrosis. Expert Rev Hematol 7:325–332. https://doi.org/10.2217/fon.15.200
Lamb YN (2022) Pacritinib: first approval. Drugs 82:831–838. https://doi.org/10.1007/s40265-022-01718-y
Tremblay D, Mascarenhas J (2018) Pacritinib to treat myelofibrosis patients with thrombocytopenia. Expert Rev Hematol 11:707–714. https://doi.org/10.1080/17474086.2018.1500456
Xu L, Feng J, Gao G, Tang H (2019) Momelotinib for the treatment of myelofibrosis. Expert Opin Pharmacother 20:1943–1951. https://doi.org/10.1080/14656566.2019.1657093
Verstovsek S, Chen C-C, Egyed M, Ellis M, Fox L, Goh YT et al (2021) Momentum: momelotinib vs danazol in patients with myelofibrosis previously treated with JAKi who are symptomatic and anemic. Future Oncol 17:1449–1458. https://doi.org/10.2217/fon-2020-1048
Ma L, Clayton JR, Walgren RA, Zhao B, Evans RJ, Smith MC et al (2013) Discovery and characterization of LY2784544, a small-molecule tyrosine kinase inhibitor of JAK2V617F. Blood Cancer J 3:e109. https://doi.org/10.1038/bcj.2013.6
Berdeja J, Palandri F, Baer MR, Quick D, Kiladjian JJ, Martinelli G et al (2018) Phase 2 study of gandotinib (LY2784544) in patients with myeloproliferative neoplasms. Leuk Res 71:82–88. https://doi.org/10.1016/j.leukres.2018.06.014
Nakaya Y, Shide K, Niwa T, Homan J, Sugahara S, Horio T et al (2011) Efficacy of NS-018, a potent and selective JAK2/Src inhibitor, in primary cells and mouse models of myeloproliferative neoplasms. Blood Cancer J 1:e29. https://doi.org/10.1038/bcj.2011.29
Liu J, Lv B, Yin H, Zhu X, Wei H, Ding Y (2020) A phase I, randomized, double-blind, placebo-controlled, single ascending dose, multiple ascending dose and food effect study to evaluate the tolerance, pharmacokinetics of jaktinib, a new selective janus kinase inhibitor in healthy Chinese volunteers. Front Pharmacol 11:604314. https://doi.org/10.3389/fphar.2020.604314
Davis RR, Li B, Yun SY, Chan A, Nareddy P, Gunawan S et al (2021) Structural insights into jak2 inhibition by ruxolitinib, fedratinib, and derivatives Thereof. J Med Chem 64:2228–2241. https://doi.org/10.1021/acs.jmedchem.0c01952
Bader MS, Meyer SC (2022) JAK2 in Myeloproliferative neoplasms: still a protagonist. Pharmaceuticals 15:160. https://doi.org/10.3390/ph15020160
Hasselbalch HC (2013) The role of cytokines in the initiation and progression of myelofibrosis. Cytokine Growth Factor Rev 24:133–145. https://doi.org/10.1016/j.cytogfr.2013.01.004
Koppikar P, Bhagwat N, Kilpivaara O, Manshouri T, Adli M, Hricik T et al (2012) Heterodimeric JAK-STAT activation as a mechanism of persistence to JAK2 inhibitor therapy. Nature 489:155–159. https://doi.org/10.1038/nature11303
Deshpande A, Reddy MM, Schade GO, Ray A, Chowdary TK, Griffin JD et al (2012) Kinase domain mutations confer resistance to novel inhibitors targeting JAK2V617F in myeloproliferative neoplasms. Leukemia 26:708–715. https://doi.org/10.1038/leu.2011.255
Downes CEJ, McClure BJ, Rehn J, Breen J, Bruning JB, Yeung DT et al (2020) Acquired mutations within the jak2 kinase domain confer resistance to jak inhibitors in an in vitro model of a high-risk acute lymphoblastic leukemia. Blood 136:5–6. https://doi.org/10.1182/blood-2020-133491
Bhagwat N, Levine RL, Koppikar P (2013) Sensitivity and resistance of JAK2 inhibitors to myeloproliferative neoplasms. Int J Hematol 97:695–702. https://doi.org/10.1007/s12185-013-1353-5
Brkic S, Meyer SC (2021) Challenges and perspectives for therapeutic targeting of myeloproliferative neoplasms. Hemasphere. https://doi.org/10.1097/HS9.0000000000000516
Khan I, Huang Z, Wen Q, Stankiewicz MJ, Gilles L, Goldenson B et al (2013) AKT is a therapeutic target in myeloproliferative neoplasms. Leukemia 27:1882–1890. https://doi.org/10.1038/leu.2013.167
Lu X, Smaill JB, Ding K (2020) New promise and opportunities for allosteric kinase inhibitors. Angew Chem Int Ed Engl 59:13764–13776. https://doi.org/10.1002/anie.201914525
Kung JE, Jura N (2019) Prospects for pharmacological targeting of pseudokinases. Nat Rev Drug Discov 18:501–526. https://doi.org/10.1038/s41573-019-0018-3
Byrne DP, Foulkes DM, Eyers PA (2017) Pseudokinases: update on their functions and evaluation as new drug targets. Future Med Chem 9:245–265. https://doi.org/10.4155/fmc-2016-0207
Wu P, Clausen MH, Nielsen TE (2015) Allosteric small-molecule kinase inhibitors. Pharmacol Ther 156:59–68. https://doi.org/10.1016/j.pharmthera.2015.10.002
Wrobleski ST, Moslin R, Lin S, Zhang Y, Spergel S, Kempson J et al (2019) Highly selective inhibition of tyrosine kinase 2 (tyk2) for the treatment of autoimmune diseases: discovery of the allosteric inhibitor BMS-986165. J Med Chem 62:8973–8995. https://doi.org/10.1021/acs.jmedchem.9b00444
Le AM, Puig L, Torres T (2022) Deucravacitinib for the treatment of psoriatic disease. Am J Clin Dermatol 23:813–822. https://doi.org/10.1007/s40257-022-00720-0
Singh J (2022) The ascension of targeted covalent inhibitors. J Med Chem 65:5886–5901. https://doi.org/10.1021/acs.jmedchem.1c02134
Baillie TA (2016) Targeted covalent inhibitors for drug design. Angew Chem Int Ed Engl 55:13408–13421. https://doi.org/10.1002/anie.201601091
Kavanagh ME, Horning BD, Khattri R, Roy N, Lu JP, Whitby LR et al (2022) Selective inhibitors of JAK1 targeting an isoform-restricted allosteric cysteine. Nat Chem Biol 18:1388–1398. https://doi.org/10.1038/s41589-022-01098-0
Ishikawa C, Senba M, Mori N (2018) Anti-adult T-cell leukemia/lymphoma activity of cerdulatinib, a dual SYK/JAK kinase inhibitor. Int J Oncol 53:1681–1690. https://doi.org/10.3892/ijo.2018.4513
Shen P, Wang Y, Jia X, Xu P, Qin L, Feng X et al (2022) Dual-target janus kinase (jak) inhibitors: comprehensive review on the JAK-based strategies for treating solid or hematological malignancies and immune-related diseases. Eur J Med Chem 239:114551. https://doi.org/10.1016/j.ejmech.2022.114551
Li X, Li X, Liu F, Li S, Shi D (2021) Rational multitargeted drug design strategy from the perspective of a medicinal chemist. J Med Chem 64:10581–10605. https://doi.org/10.1021/acs.jmedchem.1c00683
Tian XY, Liu L (2012) Drug discovery enters a new era with multi-target intervention strategy. Chin J Integr Med 18:539–542. https://doi.org/10.1007/s11655-011-0900-2
Passamonti F, Maffioli M (2018) The role of JAK2 inhibitors in MPNs 7 years after approval. Blood 131:2426–2435. https://doi.org/10.1182/blood-2018-01-791491
Cervantes F, Martinez-Trillos A (2013) Myelofibrosis: an update on current pharmacotherapy and future directions. Expert Opin Pharmacother 14:873–884. https://doi.org/10.1517/14656566.2013.783019
Xu N, Luo J, Luo D, Liang H, Tan Y, Liu Q et al (2021) Targeting metabolic dysregulation for ruxolitinib failure in MPN. Blood 138:4321–4321. https://doi.org/10.1182/blood-2021-150291
Sant’Antonio E, Bonifacio M, Breccia M, Rumi E (2019) A journey through infectious risk associated with ruxolitinib. Br J Haematol 187:286–295. https://doi.org/10.1111/bjh.16174
Mascarenhas J, Hoffman R, Talpaz M, Gerds AT, Stein B, Gupta V et al (2018) Pacritinib vs best available therapy, including ruxolitinib, in patients with myelofibrosis: a randomized clinical trial. JAMA Oncol 4:652–659. https://doi.org/10.1001/jamaoncol.2017.5818
Verstovsek S, Talpaz M, Ritchie E, Wadleigh M, Odenike O, Jamieson C et al (2017) A phase I, open-label, dose-escalation, multicenter study of the JAK2 inhibitor NS-018 in patients with myelofibrosis. Leukemia 31:393–402. https://doi.org/10.1038/leu.2016.215
Zhang Y, Zhou H, Jiang Z, Wu D, Zhuang J, Li W et al (2022) Safety and efficacy of jaktinib in the treatment of Janus kinase inhibitor-naive patients with myelofibrosis: results of a phase II trial. Am J Hematol 97:1510–1519. https://doi.org/10.1002/ajh.26709
Funding
Financial support from Sichuan Science and Technology Program of China (2023NSFSC1692 and 2022YFS0614), and Luzhou Science and Technology Program of China (2022-SYF-46).
Author information
Authors and Affiliations
Contributions
XL, BW and YW wrote the main manuscript text. BW, YY, YL and XL prepared the articles collection. JW and YW supervised and advanced the process. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, X., Wang, B., Liu, Y. et al. JAK2 inhibitors for the treatment of Philadelphia-negative myeloproliferative neoplasms: current status and future directions. Mol Divers (2023). https://doi.org/10.1007/s11030-023-10742-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11030-023-10742-3