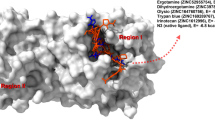
Abstract
Hepatitis is a medical condition characterized by inflammation of the liver. It is commonly caused by the hepatitis viruses A, B, C, D, and E. Hepatitis A virus (HAV) is highly contagious and can spread from infected individuals, through contaminated food, blood, or can also be water-borne. As per the statistics of World Health Organization (WHO), HAV infects about 1.4 million individuals each year globally. In this research work, we have focused on identifying natural product-based potential inhibitors for the two major enzymes of HAV namely 3C proteinase (3Cpro) and RNA-directed RNA polymerase (RdRP). The enzyme 3Cpro plays an important role in proteolytic activity that promotes viral maturation and infectivity. RNA-directed RNA polymerase facilitate viral replication and transcription. Structure-based virtual screening was carried out using NPACT database that contains a collection of 1574 curated plant-derived natural compounds that are validated by experiments. The screening procedure identified the phytochemical Mulberrofuran W, which could bind to both the targets 3Cpro and RdRP. The phytochemical Mulberrofuran W also had better binding affinity compared to the control compounds atropine and pyridinyl ester, which are previously identified inhibitors of HAV 3Cpro and RdRP, respectively. The Mulberrofuran W bound 3Cpro and RdRP complexes were subjected to 200 ns molecular dynamics simulations and were found to be stable and interacting with the active site of the enzymes throughout the course of complex MD simulations. In addition to DFT, MMGBSA studies were also performed to validate the identified potential inhibitor further. The identified phytochemical Mulberrofuran W can be considered as a new potential drug candidate and could be taken up for experimental evaluation against HAV infection.
Graphical abstract













Similar content being viewed by others
References
Kukan M, Haddad PS (2001) Role of hepatocytes and bile duct cells in preservation-reperfusion injury of liver grafts. Liver Transpl Off Publ Am Assoc Study Liver Dis Int Liver Transpl Soc 7:381–400. https://doi.org/10.1053/jlts.2001.23913
Stéphenne X, Najimi M, Sokal EM (2010) Hepatocyte cryopreservation: is it time to change the strategy? World. J Gastroenterol 16:1–14
Michalopoulos GK (2007) Liver regeneration. J Cell Physiol 213:286–300. https://doi.org/10.1002/jcp.21172
Gao B, Jeong W-I, Tian Z (2008) Liver: an organ with predominant innate immunity. Hepatology 47:729–736. https://doi.org/10.1002/hep.22034
Pebody RG, Leino T, Ruutu P et al (1998) Foodborne outbreaks of hepatitis A in a low endemic country: an emerging problem? Epidemiol Infect 120:55–59. https://doi.org/10.1017/s0950268897008340
Johnson Z, Thornton L, Tobin A et al (1995) An outbreak of hepatitis A among Irish haemophiliacs. Int J Epidemiol 24:821–828. https://doi.org/10.1093/ije/24.4.821
Nainan OV, Xia G, Vaughan G, Margolis HS (2006) Diagnosis of hepatitis A virus infection: a molecular approach. Clin Microbiol Rev 19:63–79. https://doi.org/10.1128/CMR.19.1.63-79.2006/ASSET/F2C09994-DD75-4B66-A7D9-0A53F3B326DD/ASSETS/GRAPHIC/ZCM0010621570003.JPEG
McKnight KL, Lemon SM (2018) Hepatitis A virus genome organization and replication strategy. Cold Spring Harb Perspect Med. https://doi.org/10.1101/cshperspect.a033480
Firquet S, Beaujard S, Lobert P-E et al (2015) Survival of enveloped and non-enveloped viruses on inanimate surfaces. Microbes Environ 30:140–144. https://doi.org/10.1264/jsme2.ME14145
Bird SW, Kirkegaard K (2015) Escape of non-enveloped virus from intact cells. Virology 479–480:444–449. https://doi.org/10.1016/j.virol.2015.03.044
Gupta SP (2018) Inhibition of viruses: promising targets and their importance. Stud Hepat Viruses. https://doi.org/10.1016/B978-0-12-813330-9.00003-X
Probst C, Jecht M, Gauss-Müller V (1999) Intrinsic signals for the assembly of hepatitis A virus particles: role of structural proteins VP4 and 2A. J Biol Chem 274:4527–4531. https://doi.org/10.1074/JBC.274.8.4527
Donnelly MLL, Luke G, Mehrotra A et al (2001) Analysis of the aphthovirus 2A/2B polyprotein “cleavage” mechanism indicates not a proteolytic reaction, but a novel translational effect: a putative ribosomal “skip.” J Gen Virol 82:1013–1025. https://doi.org/10.1099/0022-1317-82-5-1013
Chen P, Wojdyla JA, Colasanti O et al (2022) Biochemical and structural characterization of hepatitis A virus 2C reveals an unusual ribonuclease activity on single-stranded RNA. Nucleic Acids Res 50:9470–9489. https://doi.org/10.1093/nar/gkac671
Pisani G, Beneduce F, Gauss-Müller V, Morace G (1995) Recombinant expression of hepatitis A virus protein 3A: interaction with membranes. Biochem Biophys Res Commun 211:627–638. https://doi.org/10.1006/BBRC.1995.1859
Bergmann EM, Cherney MM, Mckendrick J et al (1999) Crystal structure of an inhibitor complex of the 3C proteinase from hepatitis A virus (HAV) and implications for the polyprotein processing in HAV. Virology 265:153–163. https://doi.org/10.1006/viro.1999.9968
Yin J, Bergmann EM, Cherney MM et al (2005) Dual modes of modification of hepatitis A virus 3C protease by a serine-derived beta-lactone: selective crystallization and formation of a functional catalytic triad in the active site. J Mol Biol 354:854–871. https://doi.org/10.1016/j.jmb.2005.09.074
Jiang Y, Yin W, Xu HE (2021) RNA-dependent RNA polymerase: Structure, mechanism, and drug discovery for COVID-19. Biochem Biophys Res Commun 538:47–53. https://doi.org/10.1016/j.bbrc.2020.08.116
He J, Hu L, Huang X et al (2020) Potential of coronavirus 3C-like protease inhibitors for the development of new anti-SARS-CoV-2 drugs: Insights from structures of protease and inhibitors. Int J Antimicrob Agents 56:106055. https://doi.org/10.1016/J.IJANTIMICAG.2020.106055
Galindez G, Matschinske J, Rose TD et al (2021) Lessons from the COVID-19 pandemic for advancing computational drug repurposing strategies. Nat Comput Sci 1:33–41. https://doi.org/10.1038/s43588-020-00007-6
Mangal M, Sagar P, Singh H et al (2013) NPACT: Naturally Occurring Plant-based Anti-cancer Compound-Activity-Target database. Nucleic Acids Res 41:D1124–D1129. https://doi.org/10.1093/nar/gks1047
Yin J, Cherney MM, Bergmann EM et al (2006) An episulfide cation (thiiranium ring) trapped in the active site of HAV 3C proteinase inactivated by peptide-based ketone inhibitors. J Mol Biol 361:673–686. https://doi.org/10.1016/j.jmb.2006.06.047
Varadi M, Anyango S, Deshpande M et al (2021) NAR Breakthrough Article AlphaFold Protein Structure Database: massively expanding the structural coverage of protein-sequence space with high-accuracy models. Nucleic Acids Res. https://doi.org/10.1093/nar/gkab1061
Jumper J, Evans R, Pritzel A et al (2021) (2021) Highly accurate protein structure prediction with AlphaFold. Nat 5967873(596):583–589. https://doi.org/10.1038/s41586-021-03819-2
Laskowski RA, MacArthur MW, Moss DS, Thornton JM (1993) PROCHECK: a program to check the stereochemical quality of protein structures. J Appl Crystallogr 26:283–291. https://doi.org/10.1107/s0021889892009944
Abraham MJ, Murtola T, Schulz R et al (2015) GROMACS: High performance molecular simulations through multi-level parallelism from laptops to supercomputers. SoftwareX 1–2:19–25. https://doi.org/10.1016/J.SOFTX.2015.06.001
Huang J, Rauscher S, Nawrocki G et al (2016) charmm36m: an improved force field for folded and intrinsically disordered proteins. Nat Method 14:71–73. https://doi.org/10.1038/nMeth.4067
Berendsen HJC, Postma JPM, van Gunsteren WF, Hermans J (1981) Interaction models for water in relation to protein hydration. Intermol Forces 15:331–342. https://doi.org/10.1007/978-94-015-7658-1_21
Vrahatis MN, Androulakis GS, Lambrinos JN, Magoulas GD (2000) A class of gradient unconstrained minimization algorithms with adaptive stepsize. J Comput Appl Math 114:367–386. https://doi.org/10.1016/S0377-0427(99)00276-9
Berendsen HJC, Postma JPM, van Gunsteren WF et al (1998) Molecular dynamics with coupling to an external bath. J Chem Phys 81:3684. https://doi.org/10.1063/1.448118
Parrinello M, Rahman A (1998) Polymorphic transitions in single crystals: a new molecular dynamics method. J Appl Phys 52:7182. https://doi.org/10.1063/1.328693
Miyamoto S, Kollman PA (1992) Settle: an analytical version of the SHAKE and RATTLE algorithm for rigid water models. J Comput Chem 13:952–962. https://doi.org/10.1002/JCC.540130805
Darden T, York D, Pedersen L (1993) Particle mesh Ewald: an N·log(N) method for Ewald sums in large systems. J Chem Phys 98:10089–10092. https://doi.org/10.1063/1.464397
Humphrey W, Dalke A, Schulten K (1996) VMD: Visual molecular dynamics. J Mol Graph 14:33–38. https://doi.org/10.1016/0263-7855(96)00018-5
Jones G, Willett P, Glen RC et al (1997) Development and validation of a genetic algorithm for flexible docking11Edited by FE Cohen. J Mol Biol 267:727–748. https://doi.org/10.1006/jmbi.1996.0897
Schneider G, Böhm H-J (2002) Virtual screening and fast automated docking methods. Drug Discov Today 7:64–70. https://doi.org/10.1016/s1359-6446(01)02091-8
Zhang C, Freddolino PL, Zhang Y (2017) COFACTOR: improved protein function prediction by combining structure, sequence and protein-protein interaction information. Nucleic Acids Res 45:291–299. https://doi.org/10.1093/nar/gkx366
Yang J, Roy A, Zhang Y (2013) Structural bioinformatics Protein-ligand binding site recognition using complementary binding-specific substructure comparison and sequence profile alignment. Bioinformatics 29:2588–2595. https://doi.org/10.1093/bioinformatics/btt447
QikProp (2020) Schrödinger Release 2020–4; Schrödinger, LLC: New York, NY
Frisch MJ, Trucks GW, Schlegel HB et al (2016) G16_D09: Gaussian 16, Revision D09. Gaussian Inc, Wallin
Zheng Y, Zheng M, Ling X et al (2013) Design, synthesis, quantum chemical studies and biological activity evaluation of pyrazole-benzimidazole derivatives as potent Aurora A/B kinase inhibitors. Bioorganic Med Chem Lett 23:3523–3530. https://doi.org/10.1016/j.bmcl.2013.04.039
Vanommeslaeghe K, Hatcher E, Acharya C et al (2010) CHARMM general force field: A force field for drug-like molecules compatible with the CHARMM all-atom additive biological force fields. J Comput Chem 31:671–690. https://doi.org/10.1002/JCC.21367
Bj G, Ap R, Km E et al (2006) Bio3d: an R package for the comparative analysis of protein structures. Bioinformatics 22:2695–2696. https://doi.org/10.1093/BIOINFORMATICS/BTL461
Zhang L, Lin D, Sun X et al (2020) Crystal structure of SARS-CoV-2 main protease provides a basis for design of improved α-ketoamide inhibitors. Science 368:409–412. https://doi.org/10.1126/science.abb3405
Wang C, Wang C, Li Q et al (2017) Crystal Structure and thermostability characterization of enterovirus D68 3D(pol). J Virol 91:e00876. https://doi.org/10.1128/JVI.00876-17
van der Linden L, Vives-Adrián L, Selisko B et al (2015) The RNA template channel of the RNA-dependent RNA polymerase as a target for development of antiviral therapy of multiple genera within a virus family. PLoS Pathog 11:1004733. https://doi.org/10.1371/journal.ppat.1004733
Vives-Adrian L, Lujan C, Oliva B et al (2014) The crystal structure of a cardiovirus RNA-dependent RNA polymerase reveals an unusual conformation of the polymerase active site. J Virol 88:5595–5607. https://doi.org/10.1128/JVI.03502-13
Thompson AA, Peersen OB (2004) Structural basis for proteolysis-dependent activation of the poliovirus RNA-dependent RNA polymerase. EMBO J 23:3462–3471. https://doi.org/10.1038/sj.emboj.7600357
Li L, Wang M, Chen Y et al (2020) Structure of the enterovirus D68 RNA-dependent RNA polymerase in complex with NADPH implicates an inhibitor binding site in the RNA template tunnel. J Struct Biol 211:107510. https://doi.org/10.1016/j.jsb.2020.107510
Alqahtani MS, Kazi M, Ma A, Mz A (2021) Advances in oral drug delivery. Front Pharmacol 12:618411. https://doi.org/10.3389/fphar.2021.618411
Debing Y, Neyts J, Thibaut HJ (2013) Molecular biology and inhibitors of hepatitis A virus. Med Res Rev 34:895–917. https://doi.org/10.1002/med.21292
Huitema C, Zhang J, Yin J et al (2008) Heteroaromatic ester inhibitors of hepatitis A virus 3C proteinase: evaluation of mode of action. Bioorg Med Chem 16:5761–5777. https://doi.org/10.1016/j.bmc.2008.03.059
Biziagos E, Crance JM, Passagot J, Deloince R (1990) Inhibitory effects of atropine, protamine, and their combination on hepatitis A virus replication in PLC/PRF/5 cells. Antimicrob Agents Chemother 34:1112–1117. https://doi.org/10.1128/AAC.34.6.1112
Geng CA, Ma YB, Zhang XM et al (2012) Mulberrofuran G and isomulberrofuran G from Morus alba L.: anti-hepatitis B virus activity and mass spectrometric fragmentation. J Agric Food Chem 60:8197–8202. https://doi.org/10.1021/JF302639B/SUPPL_FILE/JF302639B_SI_002.PDF
Hyun YL, Ji HY, Yang KR et al (2007) Inhibition of HCV replicon cell growth by 2-arylbenzofuran derivatives isolated from Mori Cortex Radicis. Planta Med 73:1481–1485. https://doi.org/10.1055/S-2007-990249/ID/3
Xia CL, Tang GH, Guo YQ et al (2019) Mulberry diels-alder-type adducts from Morus alba as multi-targeted agents for Alzheimer’s disease. Phytochemistry 157:82–91. https://doi.org/10.1016/J.PHYTOCHEM.2018.10.028
Paudel P, Yu T, Seong SH et al (2018) Protein tyrosine phosphatase 1B inhibition and glucose uptake potentials of Mulberrofuran G, Albanol B, and Kuwanon G from Root Bark of Morus alba L in Insulin-Resistant HepG2 Cells: an in vitro and in silico study. Int J Mol Sci 19:1542. https://doi.org/10.3390/IJMS19051542
Kim YS, Kim B, Kwon E-B et al (2022) Mulberrofuran G, a mulberry component, prevents SARS-CoV-2 infection by blocking the interaction between SARS-CoV-2 spike protein S1 Receptor-binding domain and human angiotensin-converting enzyme 2 receptor. Nutrients 14:4170. https://doi.org/10.3390/nu14194170
Miryala SK, Basu S, Naha A et al (2021) Identification of bioactive natural compounds as efficient inhibitors against Mycobacterium tuberculosis protein-targets: a molecular docking and molecular dynamics simulation study. J Mol Liq 341:117340. https://doi.org/10.1016/j.molliq.2021.117340
Gabrielli F, Alberti F, Russo C et al (2023) Treatment Options for Hepatitis A and E: A Non-Systematic Review. Viruses 15:1080. https://doi.org/10.3390/v15051080
Gunduz H, Karabay O, Tamer A et al (2003) N-acetyl cysteine therapy in acute viral hepatitis. World J Gastroenterol 9:2698–2700. https://doi.org/10.3748/wjg.v9.i12.2698
Crance JM, Deloince R, Leveque F et al (1994) Antiviral effect of recombinant interferon-alpha on hepatitis A virus replication in human liver cells. C R Acad Sci III 317:94–97
Jiang W, Muhammad F, Ma P et al (2018) Sofosbuvir inhibits hepatitis A virus replication in vitro assessed by a cell-based fluorescent reporter system. Antiviral Res 154:51–57. https://doi.org/10.1016/j.antiviral.2018.04.007
Zakaria HM, Salem TA, El-Araby HA et al (2018) Steroid therapy in children with fulminant hepatitis A. J Viral Hepat 25:853–859. https://doi.org/10.1111/jvh.12873
Jiang X, Kanda T, Nakamoto S et al (2015) The JAK2 inhibitor AZD1480 inhibits hepatitis A virus replication in Huh7 cells. Biochem Biophys Res Commun 458:908–912. https://doi.org/10.1016/j.bbrc.2015.02.058
Acknowledgements
The authors are grateful to the management of SASTRA Deemed University for providing all the necessary facilities. The authors thankfully acknowledge the high-performance computational facility in the School of Computing at SASTRA Deemed University for providing computational resources to complete the simulation studies. AJ is thankful to Department of Science & Technology (DST), Government of India (No. DST: SB/YS/LS/2012). MS is thankful for the funding as Senior Research Fellow grant from ICMR-SRF (No. Fellowship/96/2022-ECD-II, IRIS ID No. 2021-11346/F96).
Funding
This work is not funded by external funding agency.
Author information
Authors and Affiliations
Contributions
MS: Data Curation, Investigation, Visualization, Validation, Writing, reviewing & editing—Original Draft. SB: Investigation, Visualization. AJ: Conceptualization, Writing—Review & Editing, Resources, Supervision, Funding acquisition.
Corresponding author
Ethics declarations
Competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethical approval
Not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sureshan, M., Brintha, S. & Jothi, A. Identification of Mulberrofuran as a potent inhibitor of hepatitis A virus 3Cpro and RdRP enzymes through structure-based virtual screening, dynamics simulation, and DFT studies. Mol Divers (2023). https://doi.org/10.1007/s11030-023-10679-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11030-023-10679-7