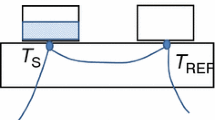
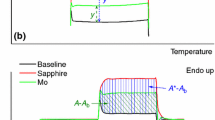
A method for enthalpy calibration of differential scanning calorimeters is described which makes it possible to eliminate the existing limits on the temperature range within which this metrological procedure can be carried out. The proposed method does not require use of standard samples of heats of fusion, so it is not limited by the thermal properties of certified materials. This method solves the problem of increasing the accuracy of measurements of specific enthalpy and heats of phase transitions of various substances for all types of differential scanning calorimeters. The effectiveness of the proposed method for solving the accuracy problems is confirmed and illustrated by the authors by comparing the results of measurements of heats of fusion of a number of metals. Results of measurements obtained on differential scanning calorimeters calibrated by the proposed method and on the same instruments, but calibrated in accordance with the generally accepted standard method of the International Union of Pure and Applied Chemistry, are compared. During the experiment, the advantages of the enthalpy calibration method developed here are identified in terms of saving the time and the costs necessary for calibration. The results can be used in work with differential scanning calorimeters, in the development of new materials, the monitoring of technological processes, production, input, and output control, as well as in the development of methods for studying various materials using differential scanning calorimeters.

Similar content being viewed by others
References
St. M. Sarge, E. Gmelin, G. W. H. Höhne, et al., Thermochim. Acta, 247, No. 2, 129–168 (1994), https://doi.org/10.1016/0040-6031(94)80118-5.
St. M. Sarge, G. W. H. Höhne, H. K. Cammenga, et al., Thermochim. Acta, 361, No. 1–2, 1–20 (2000), https://doi.org/10.1016/s0040-6031(00)00543-8.
E. Gmelin and St. M. Sarge, Pure & Appl. Chem., 67, No. 11, 1789–1800 (1995), https://doi.org/10.1351/pac199567111789.
G. D. Gatta, M. J. Richardson, St. M. Sarge, and S. Stølen, Pure & Appl. Chem., 78, No. 7, 1455–1476 (2006), https://doi.org/10.1351/pac200678071455.
D. G. Archer, J. Phys. Chem. Ref. Data, 22, 1441–1453 (1993), https://doi.org/10.1063/1.555931.
F. Grønvold, J. Chem. Thermodyn., 25, No. 9, 1133–1144 (1993), https://doi.org/10.1006/jcht.1993.1110.
D. G. Archer, J. Chem. & Eng. Data, 49, No. 5, 1364–1367 (2004), https://doi.org/10.1021/je049913p.
F. Grønvold and S. Stølen, Thermochim. Acta, 395, No. 1–2, 127–131 (2002), https://doi.org/10.1016/s0040-6031(02)00217-4.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Translated from Metrologiya, No. 4, pp. 40–52, October–December, 2021.
Rights and permissions
About this article
Cite this article
Kompan, T.A., Kulagin, V.I. & Vlasova, V.V. Enthalpy Calibration Method for Differential Scanning Calorimeters. Meas Tech 64, 999–1003 (2022). https://doi.org/10.1007/s11018-022-02035-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11018-022-02035-2