Abstract
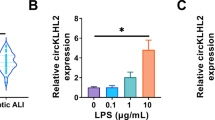
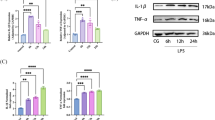
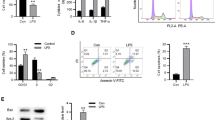
Sepsis is one of the leading causes of morbidity and mortality and a major cause of acute lung injury (ALI). carried by exosomes play a role in a variety of diseases. However,there are not many studies of exosomal miRNAs in sepsis and sepsis lung injury.miR-1298-5p and suppressor of cytokine signaling 6 (SOCS6) were silenced or overexpressed in human bronchial epithelial cells (BEAS-2B). PKH-67 Dye was used to trace exosome endocytosis. Cell permeability was evaluated by measuring trans-epithelial electrical resistance (TEER) and FITC dextran flux. ELISA kits were used for cytokine detection. Quantitative RT-PCR and western blots were used to evaluate gene expression. miR-1298-5p was elevated in exosomes from patients with sepsis lung injury (Sepsis_exo). Treatment of BEAS-2B cells using Sepsis_exo significantly inhibited cell proliferation, and induced cell permeability and inflammatory response. miR-1298-5p directly targeted SOCS6. Overexpressing SOCS6 reversed miR-1298-5p-induced cell permeability and inflammatory response. Inhibition of STAT3 blocked SOCS6-silencing caused significant increase of cell permeability and inflammation. Exosomes isolated from patients of sepsis lung injury increased cell permeability and inflammatory response in BEAS-2B cells through exosomal miR-1298-5p which targeted SOCS6 via STAT3 pathway. The findings highlight the importance of miR-1298-5p/SOCS6/STAT3 axis in sepsis lung injury and provide new insights into therapeutic strategies for sepsis lung injury.





Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
Abbreviations
- ALI:
-
Acute lung injury
- miRNAs:
-
MicroRNAs
- SOCS6:
-
Suppressor of cytokine signaling 6
- TEER:
-
Trans-epithelial electrical resistance
- Sepsis_exo:
-
Exosomes from patients with sepsis lung injury
- CIS:
-
Cytokine-inducible SH2-containing protein
- STAT3:
-
Signal transducer and activator of transcription 3
- EVs:
-
Extracellular vesicles
- GC:
-
Gastric cancer
- IBD:
-
Inflammatory bowel disease
References
Singer M, Deutschman CS, Seymour CW, Shankar-Hari M, Annane D, Bauer M, Bellomo R, Bernard GR, Chiche JD, Coopersmith CM, Hotchkiss RS, Levy MM, Marshall JC, Martin GS, Opal SM, Rubenfeld GD, van der Poll T, Vincent JL, Angus DC (2016) The third international consensus definitions for sepsis and septic shock (sepsis-3). JAMA 315:801–810
Sakr Y, Jaschinski U, Wittebole X, Szakmany T, Lipman J, Namendys-Silva SA, Martin-Loeches I, Leone M, Lupu MN, Vincent JL, Investigators I (2018) Sepsis in intensive care unit patients: worldwide data from the intensive care over nations audit. Open Forum Infect Dis 5:ofy313
Clemente G, Tuttolomondo A, Colomba D, Pecoraro R, Renda C, Della Corte V, Maida C, Simonetta I, Pinto A (2015) When sepsis affects the heart: a case report and literature review. World J Clin Cases 3:743–750
Gyawali B, Ramakrishna K, Dhamoon AS (2019) Sepsis: the evolution in definition, pathophysiology, and management. SAGE Open Med 7:2050312119835043
Gajic O, Dabbagh O, Park PK, Adesanya A, Chang SY, Hou P, Anderson H 3rd, Hoth JJ, Mikkelsen ME, Gentile NT, Gong MN, Talmor D, Bajwa E, Watkins TR, Festic E, Yilmaz M, Iscimen R, Kaufman DA, Esper AM, Sadikot R, Douglas I, Sevransky J, Malinchoc M, Illness USC (2011) Injury Trials Group: lung injury prevention study I. Early identification of patients at risk of acute lung injury: evaluation of lung injury prediction score in a multicenter cohort study. Am J Respir Crit Care Med 183:462–470
Maybauer MO, Maybauer DM, Herndon DN (2006) Incidence and outcomes of acute lung injury. N Engl J Med 354:416–417
Iscimen R, Cartin-Ceba R, Yilmaz M, Khan H, Hubmayr RD, Afessa B, Gajic O (2008) Risk factors for the development of acute lung injury in patients with septic shock: an observational cohort study. Crit Care Med 36:1518–1522
Fein AM, Calalang-Colucci MG (2000) Acute lung injury and acute respiratory distress syndrome in sepsis and septic shock. Crit Care Clin 16:289–317
Goodman RB, Pugin J, Lee JS, Matthay MA (2003) Cytokine-mediated inflammation in acute lung injury. Cytokine Growth Factor Rev 14:523–535
Lotem J, Sachs L (2002) Cytokine control of developmental programs in normal hematopoiesis and leukemia. Oncogene 21:3284–3294
Sasi W, Sharma AK, Mokbel K (2014) The role of suppressors of cytokine signalling in human neoplasms. Mol Biol Int 2014:630797
Kabir NN, Sun J, Ronnstrand L, Kazi JU (2014) SOCS6 is a selective suppressor of receptor tyrosine kinase signaling. Tumour Biol 35:10581–10589
Lai RH, Wang MJ, Yang SH, Chen JY (2009) Genomic organization and functional characterization of the promoter for the human suppressor of cytokine signaling 6 gene. Gene 448:64–73
Yoon S, Yi YS, Kim SS, Kim JH, Park WS, Nam SW (2012) SOCS5 and SOCS6 have similar expression patterns in normal and cancer tissues. Tumour Biol 33:215–221
Wu Q, Luo G, Yang Z, Zhu F, An Y, Shi Y, Fan D (2014) miR-17-5p promotes proliferation by targeting SOCS6 in gastric cancer cells. FEBS Lett 588:2055–2062
Zhu JG, Dai QS, Han ZD, He HC, Mo RJ, Chen G, Chen YF, Wu YD, Yang SB, Jiang FN, Chen WH, Sun ZL, Zhong WD (2013) Expression of SOCSs in human prostate cancer and their association in prognosis. Mol Cell Biochem 381:51–59
Letellier E, Schmitz M, Baig K, Beaume N, Schwartz C, Frasquilho S, Antunes L, Marcon N, Nazarov PV, Vallar L, Even J, Haan S (2014) Identification of SOCS2 and SOCS6 as biomarkers in human colorectal cancer. Br J Cancer 111:726–735
Filgueiras LR Jr, Martins JO, Serezani CH, Capelozzi VL, Montes MB, Jancar S (2012) Sepsis-induced acute lung injury (ALI) is milder in diabetic rats and correlates with impaired NFkB activation. PLoS ONE 7:e44987
Zhang Y, Xie Y, Zhang L, Zhao H (2020) MicroRNA-155 participates in smoke-inhalation-induced acute lung injury through inhibition of SOCS-1. Molecules 25:1022
Wang H, Lafdil F, Kong X, Gao B (2011) Signal transducer and activator of transcription 3 in liver diseases: a novel therapeutic target. Int J Biol Sci 7:536–550
Yu H, Pardoll D, Jove R (2009) STATs in cancer inflammation and immunity: a leading role for STAT3. Nat Rev Cancer 9:798–809
Wang C, Zhou G, Zeng Z (2014) Effects of peroxisome proliferator-activated receptor-beta/delta on sepsis induced acute lung injury. Chin Med J (Engl) 127:2129–2137
Ikegami M, Falcone A, Whitsett JA (1985) STAT-3 regulates surfactant phospholipid homeostasis in normal lung and during endotoxin-mediated lung injury. J Appl Physiol 2008(104):1753–1760
Jones MR, Quinton LJ, Simms BT, Lupa MM, Kogan MS, Mizgerd JP (2006) Roles of interleukin-6 in activation of STAT proteins and recruitment of neutrophils during Escherichia coli pneumonia. J Infect Dis 193:360–369
Kalluri R, LeBleu VS (2020) The biology, function, and biomedical applications of exosomes. Science 367:eaau6977
Lv LL, Cao YH, Ni HF, Xu M, Liu D, Liu H, Chen PS, Liu BC (2013) MicroRNA-29c in urinary exosome/microvesicle as a biomarker of renal fibrosis. Am J Physiol Renal Physiol 305:F1220-1227
Valadi H, Ekstrom K, Bossios A, Sjostrand M, Lee JJ, Lotvall JO (2007) Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol 9:654–659
Benz F, Tacke F, Luedde M, Trautwein C, Luedde T, Koch A, Roderburg C (2015) Circulating microRNA-223 serum levels do not predict sepsis or survival in patients with critical illness. Dis Markers 2015:384208
Roderburg C, Luedde M, Vargas Cardenas D, Vucur M, Scholten D, Frey N, Koch A, Trautwein C, Tacke F, Luedde T (2013) Circulating microRNA-150 serum levels predict survival in patients with critical illness and sepsis. PLoS ONE 8:e54612
Wang C, Zhou G, Zeng Z (2014) Effects of peroxisome proliferator-activated receptor-β/δ on sepsis induced acute lung injury. Chin Med J (Engl) 127:2129–2137
Peng W, Jiang R, Li Y, Chen J, Shao Q, Qian K, Liu F (2021) Exosomes derived from human-induced pluripotent mesenchymal stem cells inhibit the pyrolysis of alveolar macrophages. Zhonghua Wei Zhong Bing Ji Jiu Yi Xue 33:43–48
Zheng JP, Dai YM, Chen Z, Chen Q, Zheng Y, Lin X, Cui TJ (2020) Circular RNA circ-ABCB10 promotes non-small cell lung cancer proliferation and inhibits cell apoptosis through repressing KISS1. Eur Rev Med Pharmacol Sci 24:2518–2524
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25:402–408
Van Den Broucke S, Vanoirbeek J, Alfaro-Moreno E, Hoet P (2020) Contribution of mast cells in irritant-induced airway epithelial barrier impairment in vitro. Toxicol Ind Health 36:823–834
Li G, Sun L, Mu Z, Liu S, Qu H, Xie Q, Hu B (2020) MicroRNA-1298-5p inhibits cell proliferation and the invasiveness of bladder cancer cells via down-regulation of connexin 43. Biochem Cell Biol 98:227–237
Wang CM, Cheng BH, Xue QJ, Chen J, Bai B (2017) MiR-1298 affects cell proliferation and apoptosis in C6 cells by targeting SET domain containing 7. Int J Immunopathol Pharmacol 30:264–271
Cai G, Qiao S, Chen K (2015) Suppression of miR-221 inhibits glioma cells proliferation and invasion via targeting SEMA3B. Biol Res 48:37
Fan H, Goodwin AJ, Chang E, Zingarelli B, Borg K, Guan S, Halushka PV, Cook JA (2014) Endothelial progenitor cells and a stromal cell-derived factor-1alpha analogue synergistically improve survival in sepsis. Am J Respir Crit Care Med 189:1509–1519
Real JM, Ferreira LRP, Esteves GH, Koyama FC, Dias MVS, Bezerra-Neto JE, Cunha-Neto E, Machado FR, Salomao R, Azevedo LCP (2018) Exosomes from patients with septic shock convey miRNAs related to inflammation and cell cycle regulation: new signaling pathways in sepsis? Crit Care 22:68
Zhou Y, Dang J, Chang KY, Yau E, Aza-Blanc P, Moscat J, Rana TM (2016) miR-1298 inhibits mutant KRAS-driven tumor growth by repressing FAK and LAMB3. Cancer Res 76:5777–5787
Qiu ZK, Liu N, Zhao SF, Ding AP, Cheng G, Qiu WS, Qi WW (2018) MiR-1298 expression correlates with prognosis and inhibits cell proliferation and invasion of gastric cancer. Eur Rev Med Pharmacol Sci 22:1672–1679
Ruibin W, Zheng X, Chen J, Zhang X, Yang X, Lin Y (2018) Micro RNA-1298 opposes the effects of chronic oxidative stress on human trabecular meshwork cells via targeting on EIF4E3. Biomed Pharmacother 100:349–357
Nguyen AV, Wu YY, Liu Q, Wang D, Nguyen S, Loh R, Pang J, Friedman K, Orlofsky A, Augenlicht L, Pollard JW, Lin EY (2013) STAT3 in epithelial cells regulates inflammation and tumor progression to malignant state in colon. Neoplasia 15:998–1008
Lovato P, Brender C, Agnholt J, Kelsen J, Kaltoft K, Svejgaard A, Eriksen KW, Woetmann A, Odum N (2003) Constitutive STAT3 activation in intestinal T cells from patients with Crohn’s disease. J Biol Chem 278:16777–16781
Acknowledgement
We would like to thank TopEdit (www.topeditsci.com) for English language editing of this manuscript.
Author information
Authors and Affiliations
Contributions
J.M. designed this projected and wrote the manuscript; J.M., L.Y.X. performed the experiments; Q.H.S., X.Y.W., B.L. analyzed the data and edited diagrams. All authors have contributed to, read, and agreed upon the final contents of the manuscript for submission.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Ethical approval
The protocol for the present study was approved by the Ethics Committee of Shanghai Pulmonary Hospital (Shanghai, China), and it conforms to the provisions of the Declaration of Helsinki in 1995. All participants have provided their written informed consent to participate in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ma, J., Xu, LY., Sun, QH. et al. Inhibition of miR-1298-5p attenuates sepsis lung injury by targeting SOCS6. Mol Cell Biochem 476, 3745–3756 (2021). https://doi.org/10.1007/s11010-021-04170-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-021-04170-w