Abstract
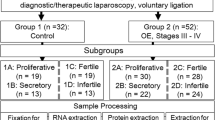
The estrogen-metabolizing activities of cytochrome P450 (CYP) enzymes have been implicated in endometriosis. However, their regulation in various sources of endometrial tissue under different hormonal conditions has not been clarified. Our objective was to study the hormone regulation of a specific CYP enzyme, namely CYP3A4, in control (n = 15) and endometriosis patients (n = 42). To this end, we evaluated mRNA expression (using real-time PCR) of CYP3A4 in tissue samples classified according to the phase of menstrual cycle at which they were obtained as confirmed by the related circulating hormone levels. Protein expression was also evaluated by Western Blot. In order to further investigate the hormonal regulation of CYP3A4, stromal cells from ovarian endometriotic lesions were cultured with the prevailing hormones of the distinct phases of the menstrual cycle. We observed that all control and endometriosis tissues express CYP3A4. Nevertheless, changes in CYP3A4 gene expression related to cycle phase were only seen in the control eutopic endometrium and not in samples from endometriosis patients, with an increase in the luteal phase. Stromal cells isolated from ovarian endometriotic lesions expressed CYP3A4 and their exposure to luteal phase-mimicking hormones (estradiol + progesterone) reduced CYP3A4 mRNA in parallel with a diminished expression of the corresponding receptors, estrogen receptor alpha and progesterone receptor. Our findings suggest that steroid hormones are able to regulate CYP3A4 mRNA expression, although the circulating levels of these hormones can only regulate control endometrium and not endometriosis tissues, probably because of dysregulated local steroid concentration in these latter samples.



Similar content being viewed by others
References
Guengerich FP (1999) Cytochrome P-450 3A4: regulation and role in drug metabolism. Annu Rev Pharmacol Toxicol 39:1–17. doi:10.1146/annurev.pharmtox.39.1.1
Stresser DM, Kupfer D (1998) Prosubstrates of CYP3A4, the major human hepatic cytochrome P450: transformation into substrates by other P450 isoforms. Biochem Pharmacol 55:1861–1871
Yamazaki H, Shimada T (1997) Progesterone and testosterone hydroxylation by cytochromes P450 2C19, 2C9, and 3A4 in human liver microsomes. Arch Biochem Biophys 346:161–169. doi:10.1006/abbi.1997.0302
Lee AJ, Kosh JW, Conney AH, Zhu BT (2001) Characterization of the NADPH-dependent metabolism of 17beta-estradiol to multiple metabolites by human liver microsomes and selectively expressed human cytochrome P450 3A4 and 3A5. J Pharmacol Exp Ther 298:420–432
Waxman DJ, Chang T (1995) Hormonal regulation of liver cytochrome P450 enzymes. In: Montellano P (ed) Cytochrome P450: structure, mechanism, and biochemistry. Plenum Press, New York, pp 391–417
de Waziers I, Cugnenc PH, Yang CS, Leroux JP, Beaune PH (1990) Cytochrome P 450 isoenzymes, epoxide hydrolase and glutathione transferases in rat and human hepatic and extrahepatic tissues. J Pharmacol Exp Ther 253:387–394
Zanger UM, Schwab M (2013) Cytochrome P450 enzymes in drug metabolism: regulation of gene expression, enzyme activities, and impact of genetic variation. Pharmacol Ther 138:103–141. doi:10.1016/j.pharmthera.2012.12.007
Choi SY, Koh KH, Jeong H (2013) Isoform-specific regulation of cytochromes P450 expression by estradiol and progesterone. Drug Metab Dispos 41:263–269. doi:10.1124/dmd.112.046276
Zheng Y, Tabbaa ZM, Khan Z, Schoolmeester JK, El-Nashar S, Famuyide A, Keeney GL, Daftary GS (2014) Epigenetic regulation of uterine biology by transcription factor KLF11 via posttranslational histone deacetylation of cytochrome p450 metabolic enzymes. Endocrinology 155:4507–4520. doi:10.1210/en.2014-1139
Sarkar MA, Vadlamuri V, Ghosh S, Glover DD (2003) Expression and cyclic variability of CYP3A4 and CYP3A7 isoforms in human endometrium and cervix during the menstrual cycle. Drug Metab Dispos 31:1–6
Hevir N, Ribic-Pucelj M, Rizner TL (2013) Disturbed balance between phase I and II metabolizing enzymes in ovarian endometriosis: a source of excessive hydroxy-estrogens and ROS? Mol Cell Endocrinol 367:74–84. doi:10.1016/j.mce.2012.12.019
Correa LF, Zheng Y, Delaney AA, Khan Z, Shenoy CC, Daftary GS (2016) TGF-beta Induces endometriotic progression via a noncanonical, KLF11-mediated mechanism. Endocrinology 157:3332–3343. doi:10.1210/en.2016-1194
Delvoux B, Groothuis P, D’Hooghe T, Kyama C, Dunselman G, Romano A (2009) Increased production of 17beta-estradiol in endometriosis lesions is the result of impaired metabolism. J Clin Endocrinol Metab 94:876–883. doi:10.1210/jc.2008-2218
Huhtinen K, Desai R, Stahle M, Salminen A, Handelsman DJ, Perheentupa A, Poutanen M (2012) Endometrial and endometriotic concentrations of estrone and estradiol are determined by local metabolism rather than circulating levels. J Clin Endocrinol Metab 97:4228–4235. doi:10.1210/Jc.2012-1154
Aghajanova L, Velarde MC, Giudice LC (2010) Altered gene expression profiling in endometrium: evidence for progesterone resistance. Semin Reprod Med 28:51–58. doi:10.1055/s-0029-1242994
Bulun SE, Cheng YH, Yin P, Imir G, Utsunomiya H, Attar E, Innes J, Julie Kim J (2006) Progesterone resistance in endometriosis: link to failure to metabolize estradiol. Mol Cell Endocrinol 248:94–103. doi:10.1016/j.mce.2005.11.041
Singh MN, Stringfellow HF, Taylor SE, Ashton KM, Ahmad M, Abdo KR, El-Agnaf OM, Martin-Hirsch PL, Martin FL (2008) Elevated expression of CYP1A1 and gamma-SYNUCLEIN in human ectopic (ovarian) endometriosis compared with eutopic endometrium. Mol Hum Reprod 14:655–663. doi:10.1093/molehr/gan056
Vandesompele J, De Preter K, Pattyn F, Poppe B, Roy Roy N, De Paepe A, Speleman F (2002) Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol 3:RESEARCH0034
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25:402–408. doi:10.1006/meth.2001.1262
Piccinato CA, Neme RM, Torres N, Sanches LR, Cruz Derogis PB, Brudniewski HF, e Silva JC, Ferriani RA (2016) Increased expression of CYP1A1 and CYP1B1 in ovarian/peritoneal endometriotic lesions. Reproduction 151:683–692. doi:10.1530/REP-15-0581
Klemmt PAB, Carver JG, Koninckx P, McVeigh EJ, Mardon HJ (2007) Endometrial cells from women with endometriosis have increased adhesion and proliferative capacity in response to extracellular matrix components: towards a mechanistic model for endometriosis progression. Hum Reprod 22:3139–3147. doi:10.1093/humrep/dem262
Huhtinen K, Saloniemi-Heinonen T, Keski-Rahkonen P, Desai R, Laajala D, Stahle M, Hakkinen MR, Awosanya M, Suvitie P, Kujari H, Aittokallio T, Handelsman DJ, Auriola S, Perheentupa A, Poutanen M (2014) Intra-tissue steroid profiling indicates differential progesterone and testosterone metabolism in the endometrium and endometriosis lesions. J Clin Endocrinol Metab 99:E2188–E2197. doi:10.1210/jc.2014-1913
Stasinopoulos DM, Rigby RA (2007) Generalized additive models for location scale and shape (GAMLSS) in R. J Stat Softw 23:1–46
Piccinato CA, Neme RM, Torres N, Sanches LR, Derogis PB, Brudniewski HF, e Silva JC, Ferriani RA (2016) Effects of steroid hormone on estrogen sulfotransferase and on steroid sulfatase expression in endometriosis tissue and stromal cells. J Steroid Biochem Mol Biol 158:117–126. doi:10.1016/j.jsbmb.2015.12.025
Ricci MS, Toscano DG, Mattingly CJ, Toscano WA Jr (1999) Estrogen receptor reduces CYP1A1 induction in cultured human endometrial cells. J Biol Chem 274:3430–3438
Bofinger DP, Feng L, Chi L, Love J, Stephen FD, Sutter TR, Osteen KG, Costich TG, Batt RE, Koury ST, Olson JR (2001) Effect of TCDD exposure on CYP1A1 and CYP1B1 expression in explant cultures of human endometrium. Toxicol Sci 62:299–314
Maglich JM, Stoltz CM, Goodwin B, Hawkins-Brown D, Moore JT, Kliewer SA (2002) Nuclear pregnane x receptor and constitutive androstane receptor regulate overlapping but distinct sets of genes involved in xenobiotic detoxification. Mol Pharmacol 62:638–646
Acknowledgement
The authors would like to acknowledge Albina Verceze for hormonal measurements and Gabriela dos S. Hidalgo and Marta Diniz, for technical assistance during sample collection and storage. Thanks to Isis Mozetic for artwork assistance, Prof. Ises de Almeida Abrahamsohn and Prof. Sergio Podgaec for critically reading the manuscript. Special thanks also to Elivane da Silva Victor and Deisy Morselli Victor for statistical consulting. This work was supported by the São Paulo Research Foundation (FAPESP: 2010/02412-6) and CNPq/INCT-Hormones.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
11010_2016_2899_MOESM1_ESM.tif
Supplementary Figure 1. Representative blots of CYP3A4 protein levels in different sites. Protein extract of the same samples were analyzed by Western Blot. Blots of protein expression of CYP3A4 in endometria and lesions are represented. Samples: 1 and 2: control endometrium, 3 and 4: eutopic endometrium, 5: non deep-infiltrating lesion, 7 and 8: deep-infiltrating lesions. (TIFF 1289 kb)
11010_2016_2899_MOESM2_ESM.tif
Supplementary Figure 2. Dose–response of estradiol in the CYP3A4 mRNA expression in stromal cells. The gene expression levels of ESR1, PR and CYP3A4 were analyzed by real time PCR in ovarian endometriotic stromal cells treated with estradiol and vehicle. Relative gene expression levels were normalized by Genorm algorithm and to the vehicle-treated ovarian stromal cells (control). No statistical differences between groups are observed (P > 0.05). (TIFF 4605 kb)
Rights and permissions
About this article
Cite this article
Piccinato, C.A., Neme, R.M., Torres, N. et al. Is cytochrome P450 3A4 regulated by menstrual cycle hormones in control endometrium and endometriosis?. Mol Cell Biochem 427, 81–89 (2017). https://doi.org/10.1007/s11010-016-2899-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-016-2899-3