Abstract
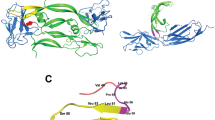
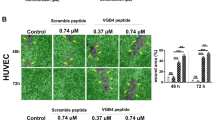
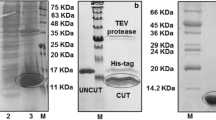
We report in the present study the design, synthesis and preliminary biological evaluations of linear, short-sized, and polymeric peptides asVEGF-A165 binding to NRP-1 and/or VEGF-R1 antagonists. These newly-synthesized peptides are structurally related to the VEGF-A165 domains encoded by exon-7 and -8. Thus, the key role of exon-7 encoded cysteine residues, and the relevance of the C-terminal peptide chemical function (amide or acid) were demonstrated. Indeed, COOH-terminal peptides showed a good selectivity for NRP-1, while CONH2-terminal peptides did not. Taking advantage of these results, we designed original VEGF-A165 binding to NRP-1 and VEGF-R1 polymeric peptide antagonists, which showed potent HUVEC anti-proliferative activity. Finally, this work paved the way for the design of future therapeutics targeting the VEGF-A165 angiogenic signaling pathway.


Similar content being viewed by others
References
Barr MP, Byrne AM, Duffy AM, Condron CM, Devocelle M, Harriott P, Bouchier-Hayes DJ, Harmey JH (2005) A peptide corresponding to the neuropilin-1-binding site of VEGF165 induces apoptosis of neuropilin-1-expressing breast tumour cells. Br J Cancer 92(2):328–333
Bielenberg DR, Pettaway CA, Takashima S, Klagsbrun M (2006) Neuropilins in neoplasms: expression, regulation, and function. Exp Cell Res 312:584–593
Binétruy-Tournaire R, Demangel C, Malavaud B, Vassy R, Rouyre S, Kraemer M, Plouët J, Derbin C, Perret G, Mazié JC (2000) Identification of a peptide blocking vascular endothelial growth factor (VEGF)-mediated angiogenesis. EMBO J 19(7):1525–1533
Biselli-Chicotte PM, Oliveira AR, Pavarino EC, Goloni-Bertollo EM (2012) VEGF gene alternative splicing: pro- and anti-angiogenic isoforms in cancer. J Cancer Res Clin Oncol 138(3):363–370
Borriello L, Montès M, Lepelletier Y, Leforban B, Liu WQ, Demange L, Delhomme B, Pavoni S, Jarray R, Boucher JL, Dufour S, Hermine O, Garbay C, Hadj-Slimane R, Raynaud F (2014) Structure-based discovery of a small non-peptidic neuropilins antagonist exerting in vitro and in vivo anti-tumor activity on breast cancer model. Cancer Lett 349(2):120–127
Caunt M, Mak J, Liang WC, Stawicki S, Pan Q, Tong RK, Kowalski J, Ho C, Reslan HB, Ross J, Berry L, Kasman I, Zlott C, Cheng Z, Le Couter J, Filvaroff EH, Plowman G, Peale F, French D, Carano R, Koch AW, Wu Y, Watts RJ, Tessier-Lavigne M, Bagri A (2008) Blocking neuropilin-2 function inhibits tumor cell metastasis. Cancer Cell 13(4):331–342
Cussac D, Vidal M, Leprince C, Liu W-Q, Cornille F, Tiraboschi G, Roques BP, Garbay C (1999) A Sos-derived peptidimer blocks the Ras signaling pathway by binding both Grb2 SH3 domains and display antiproliferative activity. FASEB J 13:31–38
Fairbrother WJ, Chample MA, Christinger HW, Keyt BA, Starovasnik MA (1998) Solution structure of the heparin-binding domain of vascular endothelial growth factor. Structure 6:637–648
Ferrara N (2001) Role of endothelial growth factor in regulation of phyysiological angiogenesis. Am J Physiol Cell Physiol 280(6):C1358–C1366
Ferrara N, Kerber RS (2005) Angiogenesis as a therapeutic target. Nature 438:967–974
Ferrara N, Gerber HP, Le Couter J (2003) The biology of VEGF and its receptors. Nat Med 9:669–676
Getz JA, Cheneval O, Craik DJ, Daugherty PS (2013) Design of a cyclotide antagonist of neuropilin-1 and -2 that potently inhibits endothelial cell migration. ACS Chem Biol 8:1147–1154
Goldwaser E, DeCourcy B, Demange L, Garbay C, Raynaud F, Hadj-Slimane R, Piquemal JP, Gresh N (2014) Conformational analysis of a polyconjugated protein-binding ligand by joint quantum chemistry and polarizable molecular mechanics. Addressing the issues of anisotropy, conjugation, polarization and multipole transferability. J Mol Model. doi:10.1007/s00894-014-2472-5
Grünewald FS, Prota AE, Giese A, Ballmer-Hofer K (2010) Structure-function analysis of VEGF receptor activation and the role of coreceptors in angiogenic signaling. Biochim Biophys Acta 1804:567–580
Harper SJ, Bates DO (2008) VEGF-A splicing: the key to anti-angiogenictherapeutics ? Nat Rev Cancer 8(11):880–887
Jarvis A, Allerston CK, Jia H, Herzog B, Garza-Garcia A, Winfield N, Ellard K, Aqil E, Lynch R, Chapman C, Hartzoulakis B, Nally J, Stewart M, Cheng M, Menon M, Tickner M, Djordjevic S, Driscoll PC, Zachary I, Selwood DL (2010) Small molecule inhibitor of the neuropilin-1 vascular endothelial growth factor A (VEGF-A) interaction. J Med Chem 53(5):2215–2226
Jia H, Bagherzadeh A, Hartzoulakis B, Jarvis A, Löhr M, Shaikh S, Aqil R, Cheng L, Tickner M, Esposito D, Harris R, Driscoll PC, Selwood DL, Zachary IC (2006) Characterization of a bicyclic peptide neuropilin-1 (NP-1) antagonist (EG3287) reveals importance of vascular endothelial growth factor exon 8 for NP-1 binding and role of NP-1 in KDR signaling. J Biol Chem 281(19):13493–13502
Jubb AM, Strickland LA, Liu SD, Mak J, Schmidt M, Koeppen H (2012) Neuropilin-1 expression in cancer and development. J Pathol 226:50–60
Keck RG, Berleau L, Harris R, Keyth BA (1997) Disulphide structure of the heparin binding domain in vascular endothelial growth factor: characterization of post translational modifications in VEGF. Arch Biochem Biophys 344:103–113
Keyt BA, Berleau LT, Nguyen HV, Chen H, Heinsohn H, Vandeln R, Ferrara N (1996) The carboxyl terminal domain (111-165) of vascular endothelial growth factor is critical for its mitogenic potency. J Biol Chem 271:7788–7795
Koch S (2012) Neuropilin signaling in angiogenesis. Biochem Soc Trans 40(1):20–25
Liu WQ, Megale V, Borriello L, Leforban B, Montès M, Goldwaser E, Gresh N, Piquemal JP, Hadj-Slimane R, Hermine O, Garbay C, Raynaud F, Lepelletier Y, Demange L (2014) Synthesis and structure-activity relationship of non-peptidic antagonists of neuropilin-1 receptor. Bioorg Med Chem Lett 24(17):4254–4259
Mamluk R, Gechtman Z, Kutcher ME, Gasiunas N, Gallagher J, Klagsbrun M (2002) Neuropilin-1 binds vascular endothelial growth factor 165, placenta growth factor-2 and heparin via its b1b2 domain. J Biol Chem 277:24818–24825
Miller JW, Le Couter J, Strauss EC, Ferrara N (2013) Vascular endothelial growth factor A in intraocular vascular disease. Ophthalmology 120(1):106–114
Moroder L (2005) Isosteric replacement of sulfur with other chalcogens in peptides and proteins. J Pept Sci 11:187–214
Novoa A, Pellegrini-Moïse N, Bechet D, Barberi-Heyob M, Chapleur Y (2010) Sugar-besed peptidomimetics as potential inhibitors of the vascular endothelial growth factor binding to neuropilin-1. Bioorg Med Chem 1:3285–3298
Pan Q, Chanthery Y, Liang WC, Stawicki S, Mak J, Rathore N, Tong RK, Kowalski J, Yee SF, Pacheco G, Ross S, Cheng Z, Le Couter J, Plowman G, Peale F, Koch AW, Wu Y, Bagri A, Tessier-Lavigne M, Watts RJ (2007) Blocking neuropilin-1 function has an additive effect with anti-VEGF to inhibit tumor growth. Cancer Lett 11(1):53–67
Parker MW, Xu P, Li X, Vander Kooi CW (2012) Structural basis for selective vascular endothelial growth factor-A (VEGF-A) binding to neuropilin-1. J Biol Chem 287(14):11082–11089
Parker MW, Linkugel AD, Vander Kooi CW (2013) Effect of C-terminal sequence on competitive semaphoring binding to neuropilin-1. J Mol Biol 425:4405–4414
Pavet V, Beyrath J, Pardin C, Morizot A, Lechner MC, Briand JP, Wendland M, Maison W, Fournel S, Micheau O, Guichard G, Groenemeyer H (2010) Multivalent DR5 peptides activates the TRAIL death pathway and exert tumoricidal activity. Cancer Res 70(3):1101–1110
Pellet-Many C, Frankel P, Jia H, Zachary I (2008) Neuropilins: structure, function and role in diseases. Biochem J 411:211–226
Perret GY, Starzec A, Hauet N, Vergote J, Le Pecheur M, Vassy R, Léger G, Verbeke KA, Bormans G, Nicolas P, Verbruggen AM, Moretti JL (2004) In vitro evaluation and biodistribution of a 99mTc-labelled anti-VEGF peptide targeting neuropilin-1. Nucl Med Biol 31:575–581
Prud’homme GJ, Glinka Y (2012) Neuropilins are multifunctional coreceptors involved in tumor initiation, growth metastasis and immunity. Oncotarget 3(9):921–939
Starzec A, Ladam P, Vassy R, Badache S, Bouchemal N, Navaza A, DuPenhoat H, Perret GY (2007) Structure-function analysis of the antiangiogenic ATWLPPR peptide inhibiting VEGF165 binding to neuropilin-1 and molecular dynamics simulations of the ATWLPPR/neuropilin-1 complex. Peptides 28(12):2397–2402
Starzec A, Miteva MA, Ladam P, Villoutreix BO, Perret GY (2014) Discovery of novel inhibitors of vascular endothelial growth factor-A-Neuropilin-1 interaction by structure-based virtual screening. Bioorg Med Chem 22(15):4042–4048
Teesalu T, Sugahara KN, Kotamraju VR, Ruoslahti E (2009) C-End rule peptides mediate neuropilin-1-dependent cell, vascular, and tissue penetration. Proc Natl Acad Sci USA 106(38):16157–16162
Thomas N, Pernot M, Vanderesse R, Becuwe P, Kamarulzaman E, DaSilva D, François A, Frochot C, Guillemin F, Barberi-Heyob M (2010) Photodynamic therapy targeting neuropilin-1: interest of pseudopeptides with improved stability properties. Biochem Pharmacol 80(2):226–235
Vander Kooi CW, Jusino MA, Perman B, Neau DB, Bellamy HD, Leahy DJ (2007) Structural basis for ligand and heparin binding to neuropilin B domains. Proc Natl Acad Sci USA 104(5):6152–6157
Von Wronski MA, Raju N, Pillai R, Bogdan NJ, Marinelli ER, Nanjappan P, Ramalingam K, Arunachalam T, Eaton S, Linder K, Yan F, Pochon S, Tweedle MF, Nunn AD (2006) Tuftsin binds neuropilin-1 through a sequence similar to that encoded by exon 8 of vascular endothelial growth factor. J Biol Chem 281(9):5702–5710
Walsh DA, McWilliams DF, Turley MJ, Dixon MR, Fransès RE, Mapp PI, Wilson D (2010) Angiogenesis and nerve growth factor at the osteochondral junction in rheumatoid and osteoarthritis. Rheumatology 49:1852–1861
Wild JRL, Staton CA, Chapple K, Corfe BM (2012) Neuropilins: expression and roles in the epithelium. Int J Exp Pathol 93(2):81–103
Xin Y, Bai S, Damico-Beyer LA, Jin D, Liang WC, Wu Y, Theil FP, Joshi A, Lu Y, Lowe J, Maia M, Brachmann RK, Xiang H (2012) Anti-neuropilin-1 (MNRP1685A): unexpected pharmacokinetic differences across species, from preclinical models to humans. Pharm Res 29(9):2512–2521
Acknowledgments
This study was supported by Grant from the “Institute National du Cancer” (INCa “ANGIOMED”, 2007–2010 Grant). LB was supported by a “Leonardo da Vinci” Grant (Unipharma graduate, Noopolis Fundation, Rome, Italy) for her research. The authors are thankful to Drs. Rachid Benhida and Nadine Martinet for the critical reviewing of this article.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Liu, WQ., Borriello, L., Allain, B. et al. New Peptides Structurally Related to VEGF-A165 Exon-7 and -8 Encoded Domains Antagonize Its Binding to NRP-1 and VEGF-R1. Int J Pept Res Ther 21, 117–124 (2015). https://doi.org/10.1007/s10989-014-9436-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10989-014-9436-6