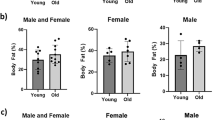
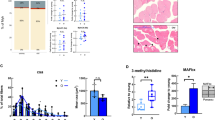
Abstract
Muscle damage resulting from physical activities such as exercise triggers an immune response crucial for tissue repair and recovery. This study investigates the immune cell profiles in muscle biopsies of individuals engaged in resistance exercise (RE) and explores the impact of age and sex on the immune response following exercise-induced muscle damage. Microarray datasets from muscle biopsies of young and old subjects were analyzed, focusing on the gene expression patterns associated with immune cell activation. Genes were compared with immune cell signatures to reveal the cellular landscape during exercise. Results show that the most significant modulated gene after RE was Folliculin Interacting Protein 2 (FNIP2) a crucial regulator in cellular homeostasis. Moreover, the transcriptome was stratified based on the expression of FNIP2 and the 203 genes common to the groups obtained based on sex and age. Gene ontology analysis highlighted the FLCN-FNIP1-FNIP2 complex, which exerts as a negative feedback loop to Pi3k-Akt-mTORC1 pathway. Furthermore, we highlighted that the young females exhibit a distinct innate immune cell activation signature compared to males after a RE session. Specifically, young females demonstrate a notable overlap with dendritic cells (DCs), M1 macrophages, M2 macrophages, and neutrophils, while young males overlap with M1 macrophages, M2 macrophages, and motor neurons. Interestingly, in elderly subjects, both sexes display M1 macrophage activation signatures. Comparison of young and elderly signatures reveals an increased M1 macrophage percentage in young subjects. Additionally, common genes were identified in both sexes across different age groups, elucidating biological functions related to cell remodeling and immune activation. This study underscores the intricate interplay between sex, age, and the immune response in muscle tissue following RE, offering potential directions for future research. Nevertheless, there is a need for further studies to delve deeper and confirm the dynamics of immune cells in response to exercise-induced muscle damage.












Similar content being viewed by others
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Abbas AR, Baldwin D, Ma Y et al (2005) Immune response in silico (IRIS): immune-specific genes identified from a compendium of microarray expression data. Genes Immun 6(4):319–331. https://doi.org/10.1038/sj.gene.6364173
Arnold L, Henry A, Poron F et al (2007) Inflammatory monocytes recruited after skeletal muscle injury switch into antiinflammatory macrophages to support myogenesis. J Exp Med 204(5):1057–1069. https://doi.org/10.1084/jem.20070075
Ashburner M, Ball CA, Blake JA et al (2000) Gene ontology: tool for the unification of biology. The Gene Ontology Consortium. Nat Genet 25(1):25–29. https://doi.org/10.1038/75556
Bodine SC (2022) The role of mTORC1 in the regulation of skeletal muscle mass. Fac Rev 11:32. https://doi.org/10.12703/r/11-32
Box GEP, Tiao GC (2011) Bayesian inference in statistical analysis. Wiley
Care MA, Barrans S, Worrillow L, Jack A, Westhead DR, Tooze RM (2013) A microarray platform-independent classification tool for cell of origin class allows comparative analysis of gene expression in diffuse large B-cell lymphoma. PLoS ONE 8(2):e55895. https://doi.org/10.1371/journal.pone.0055895
Castrogiovanni P, Li Volti G, Sanfilippo C et al (2018) Fasting and fast Food Diet play an Opposite Role in mice Brain Aging. Mol Neurobiol 55(8):6881–6893. https://doi.org/10.1007/s12035-018-0891-5
Catrogiovanni P, Musumeci G, Giunta S, Imbesi R, Di Rosa M (2020) The expression levels of CHI3L1 and IL15Rα correlate with TGM2 in duodenum biopsies of patients with celiac disease. Inflamm Res 69(9):925–935. https://doi.org/10.1007/s00011-020-01371-9
Centini R, Tsang M, Iwata T et al (2018) Loss of Fnip1 alters kidney developmental transcriptional program and synergizes with TSC1 loss to promote mTORC1 activation and renal cyst formation. PLoS ONE 13(6):e0197973. https://doi.org/10.1371/journal.pone.0197973
Chazaud B, Sonnet C, Lafuste P et al (2003) Satellite cells attract monocytes and use macrophages as a support to escape apoptosis and enhance muscle growth. J Cell Biol 163(5):1133–1143. https://doi.org/10.1083/jcb.200212046
Cheadle C, Vawter MP, Freed WJ, Becker KG (2003) Analysis of microarray data using Z score Transformation. J Mol Diagn 5(2):73–81
Chen QR, Song YK, Wei JS et al (2008) An integrated cross-platform prognosis study on neuroblastoma patients. Genomics 92(4):195–203. https://doi.org/10.1016/j.ygeno.2008.05.014
Clausen L, Stein A, Grønbæk-Thygesen M et al (2020) Folliculin variants linked to Birt-Hogg-Dubé syndrome are targeted for proteasomal degradation. PLoS Genet 16(11):e1009187. https://doi.org/10.1371/journal.pgen.1009187
Clough E, Barrett T (2016) The Gene expression Omnibus Database. Methods Mol Biol 1418:93–110. https://doi.org/10.1007/978-1-4939-3578-9_5
Damas F, Libardi CA, Ugrinowitsch C (2018) The development of skeletal muscle hypertrophy through resistance training: the role of muscle damage and muscle protein synthesis. Eur J Appl Physiol 118(3):485–500. https://doi.org/10.1007/s00421-017-3792-9
Davis S, Meltzer PS (2007) GEOquery: a bridge between the Gene expression Omnibus (GEO) and BioConductor. Bioinformatics 23(14):1846–1847. https://doi.org/10.1093/bioinformatics/btm254
de Martín Garrido N, Aylett CHS (2020) Nutrient Signaling and Lysosome Positioning Crosstalk Through a Multifunctional Protein, Folliculin. Frontiers in Cell and Developmental Biology.;8. Accessed March 1, 2024. https://www.frontiersin.org/articles/https://doi.org/10.3389/fcell.2020.00108
de Salles B, Simão R, Miranda F, Novaes J, Lemos A, Willardson J (2009) Rest interval between sets in Strength Training. Sports Med (Auckland NZ) 39:765–777. https://doi.org/10.2165/11315230-000000000-00000
Dennis RA, Przybyla B, Gurley C et al (2006) Aging alters macrophage properties in human skeletal muscle both at rest and in response to acute resistance exercise. FASEB J 20(4):A385–A385. https://doi.org/10.1096/fasebj.20.4.A385
Di Rosa M, Sanfilippo C, Libra M, Musumeci G, Malaguarnera L (2015) Different pediatric brain tumors are associated with different gene expression profiling. Acta Histochem 117(4–5):477–485. https://doi.org/10.1016/j.acthis.2015.02.010
Di Rosa M, Giallongo C, Romano A et al (2020) Immunoproteasome genes are modulated in CD34+JAK2V617F mutated cells from primary myelofibrosis patients. Int J Mol Sci 21(8):2926. https://doi.org/10.3390/ijms21082926
Egashira K, Inou T, Hirooka Y et al (1993) Effects of age on endothelium-dependent vasodilation of resistance coronary artery by acetylcholine in humans. Circulation 88(1):77–81. https://doi.org/10.1161/01.cir.88.1.77
Fernández LP, Deleyto-Seldas N, Colmenarejo G et al (2022) Folliculin-interacting protein FNIP2 impacts on overweight and obesity through a polymorphism in a conserved 3’ untranslated region. Genome Biol 23(1):230. https://doi.org/10.1186/s13059-022-02798-5
Freidenreich DJ, Volek JS (2012) Immune responses to resistance exercise. Exerc Immunol Rev 18:8–41
Hasumi H, Baba M, Hasumi Y et al (2015) Folliculin-interacting proteins Fnip1 and Fnip2 play critical roles in kidney tumor suppression in cooperation with Flcn. Proc Natl Acad Sci U S A 112(13):E1624–1631. https://doi.org/10.1073/pnas.1419502112
Kraemer WJ, Staron RS, Hagerman FC et al (1998) The effects of short-term resistance training on endocrine function in men and women. Eur J Appl Physiol Occup Physiol 78(1):69–76. https://doi.org/10.1007/s004210050389
Laffont S, Seillet C, Guéry JC (2017) Estrogen receptor-dependent regulation of dendritic cell development and function. Front Immunol 8:108. https://doi.org/10.3389/fimmu.2017.00108
Ling NXY, Kaczmarek A, Hoque A et al (2020) mTORC1 directly inhibits AMPK to promote cell proliferation under nutrient stress. Nat Metab 2(1):41–49. https://doi.org/10.1038/s42255-019-0157-1
López de Padilla CM, Vallejo AN, McNallan KT et al (2007) Plasmacytoid dendritic cells in inflamed muscle of patients with juvenile dermatomyositis. Arthritis Rheum 56(5):1658–1668. https://doi.org/10.1002/art.22558
Mahoney DJ, Safdar A, Parise G et al (2008) Gene expression profiling in human skeletal muscle during recovery from eccentric exercise. Am J Physiol Regul Integr Comp Physiol 294(6):R1901–1910. https://doi.org/10.1152/ajpregu.00847.2007
Martinez FO, Gordon S (2014) The M1 and M2 paradigm of macrophage activation: time for reassessment. F1000Prime Rep 6:13. https://doi.org/10.12703/P6-13
Martinez FO, Gordon S, Locati M, Mantovani A (2006) Transcriptional profiling of the human monocyte-to-macrophage differentiation and polarization: new molecules and patterns of gene expression. J Immunol 177(10):7303–7311. https://doi.org/10.4049/jimmunol.177.10.7303
Martins L, Gallo CC, Honda TSB et al (2020) Skeletal muscle healing by M1-like macrophages produced by transient expression of exogenous GM-CSF. Stem Cell Res Ther 11(1):473. https://doi.org/10.1186/s13287-020-01992-1
Mehmood R, El-Ashram S, Bie R, Dawood H, Kos A (2017) Clustering by fast search and merge of local density peaks for gene expression microarray data. Sci Rep 7:45602. https://doi.org/10.1038/srep45602
Miles MP, Kraemer WJ, Grove DS et al (2002) Effects of resistance training on resting immune parameters in women. Eur J Appl Physiol 87(6):506–508. https://doi.org/10.1007/s00421-002-0683-4
Minari ALA, Thomatieli-Santos RV (2022) From skeletal muscle damage and regeneration to the hypertrophy induced by exercise: what is the role of different macrophage subsets? Am J Physiology-Regulatory Integr Comp Physiol 322(1):R41–R54. https://doi.org/10.1152/ajpregu.00038.2021
Mounier R, Lantier L, Leclerc J, Sotiropoulos A, Foretz M, Viollet B (2011) Antagonistic control of muscle cell size by AMPK and mTORC1. Cell Cycle 10(16):2640–2646. https://doi.org/10.4161/cc.10.16.17102
Ogura Y, Iemitsu M, Naito H et al (2011) Single bout of running exercise changes LC3-II expression in rat cardiac muscle. Biochem Biophys Res Commun 414(4):756–760. https://doi.org/10.1016/j.bbrc.2011.09.152
Panci G, Chazaud B (2021) Inflammation during post-injury skeletal muscle regeneration. Semin Cell Dev Biol 119:32–38. https://doi.org/10.1016/j.semcdb.2021.05.031
Perandini LA, Chimin P, Lutkemeyer D, da Câmara S (2018) Chronic inflammation in skeletal muscle impairs satellite cells function during regeneration: can physical exercise restore the satellite cell niche? FEBS J 285(11):1973–1984. https://doi.org/10.1111/febs.14417
Raue U, Trappe TA, Estrem ST et al (2012) Transcriptome signature of resistance exercise adaptations: mixed muscle and fiber type specific profiles in young and old adults. J Appl Physiol (1985) 112(10):1625–1636. https://doi.org/10.1152/japplphysiol.00435.2011
Reddy TBK, Riley R, Wymore F et al (2009) TB database: an integrated platform for tuberculosis research. Nucleic Acids Res 37(Database issue):D499–508. https://doi.org/10.1093/nar/gkn652
Sanfilippo C, Malaguarnera L, Di Rosa M (2016) Chitinase expression in Alzheimer’s disease and non-demented brains regions. J Neurol Sci 369:242–249. https://doi.org/10.1016/j.jns.2016.08.029
Sanfilippo C, Nunnari G, Calcagno A et al (2017) The chitinases expression is related to simian immunodeficiency Virus Encephalitis (SIVE) and in HIV encephalitis (HIVE). Virus Res 227:220–230. https://doi.org/10.1016/j.virusres.2016.10.012
Sanfilippo C, Pinzone MR, Cambria D et al (2018) OAS Gene Family expression is Associated with HIV-Related Neurocognitive disorders. Mol Neurobiol 55(3):1905–1914. https://doi.org/10.1007/s12035-017-0460-3
Sanfilippo C, Castrogiovanni P, Imbesi R et al (2019) Middle-aged healthy women and Alzheimer’s disease patients present an overlapping of brain cell transcriptional profile. Neuroscience 406:333–344. https://doi.org/10.1016/j.neuroscience.2019.03.008
Sanfilippo C, Castrogiovanni P, Vinciguerra M et al (2023) Neuro-immune deconvolution analysis of OAS3 as a transcriptomic central node in HIV-associated neurocognitive disorders. J Neurol Sci 446:120562. https://doi.org/10.1016/j.jns.2023.120562
Sartori R, Romanello V, Sandri M (2021) Mechanisms of muscle atrophy and hypertrophy: implications in health and disease. Nat Commun 12(1):330. https://doi.org/10.1038/s41467-020-20123-1
Scott JW, Hawley SA, Green KA et al (2004) CBS domains form energy-sensing modules whose binding of adenosine ligands is disrupted by disease mutations. J Clin Invest 113(2):274–284. https://doi.org/10.1172/JCI19874
Sica A, Mantovani A (2012) Macrophage plasticity and polarization: in vivo veritas. J Clin Invest 122(3):787–795. https://doi.org/10.1172/JCI59643
Singer EA, Bratslavsky G, Middelton L, Srinivasan R, Linehan WM (2011) Impact of genetics on the diagnosis and treatment of renal cancer. Curr Urol Rep 12(1):47–55. https://doi.org/10.1007/s11934-010-0156-y
Sjöstedt E, Zhong W, Fagerberg L et al (2020) An atlas of the protein-coding genes in the human, pig, and mouse brain. Science 367(6482):eaay5947. https://doi.org/10.1126/science.aay5947
Smyth GK (2004) Linear models and empirical bayes methods for assessing differential expression in microarray experiments. Stat Appl Genet Mol Biol 3(Article3). https://doi.org/10.2202/1544-6115.1027
Solomon E, Davis-Anderson K, Hovde B et al (2021) Global transcriptome profile of the developmental principles of in vitro iPSC-to-motor neuron differentiation. BMC Mol Cell Biol 22(1):13. https://doi.org/10.1186/s12860-021-00343-z
Sonobe T, Inagaki T, Sudo M, Poole DC, Kano Y (2010) Sex differences in intracellular ca(2+) accumulation following eccentric contractions of rat skeletal muscle in vivo. Am J Physiol Regul Integr Comp Physiol 299(4):R1006–1012. https://doi.org/10.1152/ajpregu.00623.2009
Szklarczyk D, Gable AL, Lyon D et al (2019) STRING v11: protein-protein association networks with increased coverage, supporting functional discovery in genome-wide experimental datasets. Nucleic Acids Res 47(D1):D607–D613. https://doi.org/10.1093/nar/gky1131
The Gene Ontology Consortium (2017) Expansion of the Gene Ontology knowledgebase and resources. Nucleic Acids Res 45(D1):D331–D338. https://doi.org/10.1093/nar/gkw1108
Thul PJ, Åkesson L, Wiking M et al (2017) A subcellular map of the human proteome. Science 356(6340):eaal3321. https://doi.org/10.1126/science.aal3321
Tidball JG, Villalta SA (2010) Regulatory interactions between muscle and the immune system during muscle regeneration. American Journal of Physiology-Regulatory, Integrative and comparative physiology. Published Online March 10. https://doi.org/10.1152/ajpregu.00735.2009
Tiidus PM (2000) Estrogen and gender effects on muscle damage, inflammation, and oxidative stress. Can J Appl Physiol 25(4):274–287. https://doi.org/10.1139/h00-022
Uhlen M, Zhang C, Lee S et al (2017) A pathology atlas of the human cancer transcriptome. Science 357(6352):eaan2507. https://doi.org/10.1126/science.aan2507
Van den Bossche J, Leenen PJM (2021) Keep your macrophages fit for healthy aging. Cell Metab 33(3):468–470. https://doi.org/10.1016/j.cmet.2021.02.004
Wang J, Coombes KR, Highsmith WE, Keating MJ, Abruzzo LV (2004) Differences in gene expression between B-cell chronic lymphocytic leukemia and normal B cells: a meta-analysis of three microarray studies. Bioinformatics 20(17):3166–3178. https://doi.org/10.1093/bioinformatics/bth381
Wynn TA, Vannella KM (2016) Macrophages in tissue repair, regeneration, and fibrosis. Immunity 44(3):450–462. https://doi.org/10.1016/j.immuni.2016.02.015
Xiao J, Cao H, Chen J (2017) False discovery rate control incorporating phylogenetic tree increases detection power in microbiome-wide multiple testing. Bioinformatics 33(18):2873–2881. https://doi.org/10.1093/bioinformatics/btx311
Xiao L, Yin Y, Sun Z et al (2024) AMPK phosphorylation of FNIP1 (S220) controls mitochondrial function and muscle fuel utilization during exercise. Sci Adv 10(6):eadj2752. https://doi.org/10.1126/sciadv.adj2752
Yang W, Hu P (2018) Skeletal muscle regeneration is modulated by inflammation. J Orthop Translat 13:25–32. https://doi.org/10.1016/j.jot.2018.01.002
Yasrebi H, Sperisen P, Praz V, Bucher P (2009) Can survival prediction be improved by merging gene expression data sets? PLoS ONE 4(10):e7431. https://doi.org/10.1371/journal.pone.0007431
Zuberi K, Franz M, Rodriguez H et al (2013) GeneMANIA prediction server 2013 update. Nucleic Acids Res W115–122 41(Web Server issue). https://doi.org/10.1093/nar/gkt533
Acknowledgements
We would like to show our gratitude to the authors of microarray datasets made available online, for consultation and re-analysis. We would like to thank Ludovico Einaudi for inspiring us to write this manuscript.
Funding
The author Michelino Di Rosa was supported by the University Research Project Grant (PIACERI 2020–2022), Department of Biomedical and Biotechnological Sciences (BIOMETEC), University of Catania, Italy. The author Amer M. Alanazi was funded by the researchers supporting project number (RSP2024R261) King Saud University, Riyadh, Saudi Arabia. The funder/sponsor had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Author information
Authors and Affiliations
Contributions
Conceptualization, M.D.; Methodology, M.D. P.C., and C.S.; Investigation, C.S., P.C., M.D.; Data Curation, G.LV., R.I., G.L., D.T., N.V., R.P., L.G., I.B., A,M.A., V.M., F.C.; Writing – Original Draft, M.D., P.C, and C.S.; Writing - Visualization, M.D.; Supervision, M.D., G.M.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Castrogiovanni, P., Sanfilippo, C., Imbesi, R. et al. Skeletal muscle of young females under resistance exercise exhibits a unique innate immune cell infiltration profile compared to males and elderly individuals. J Muscle Res Cell Motil (2024). https://doi.org/10.1007/s10974-024-09668-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10974-024-09668-6