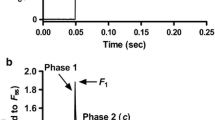
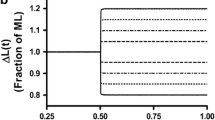
Abstract
The N-terminal extension of human cardiac troponin T (TnT), which modulates myofilament Ca2+ sensitivity, contains several hypertrophic cardiomyopathy (HCM)-causing mutations including S69F. However, the functional consequence of S69F mutation is unknown. The human analog of S69F in rat TnT is L71F (TnTL71F). Because the functional consequences due to structural changes in the N-terminal extension are influenced by the type of myosin heavy chain (MHC) isoform, we hypothesized that the TnTL71F-mediated effect would be differently modulated by α- and β-MHC isoforms. TnTL71F and wild-type rat TnT were reconstituted into de-membranated muscle fibers from normal (α-MHC) and propylthiouracil-treated rat hearts (β-MHC) to measure steady-state and dynamic contractile parameters. The magnitude of the TnTL71F-mediated attenuation of Ca2+-activated maximal tension was greater in α- than in β-MHC fibers. For example, TnTL71F attenuated maximal tension by 31% in α-MHC fibers but only by 10% in β-MHC fibers. Furthermore, TnTL71F reduced myofilament Ca2+ sensitivity by 0.11 pCa units in α-MHC fibers but only by 0.05 pCa units in β-MHC fibers. TnTL71F augmented rate constants of crossbridge recruitment and crossbridge detachment dynamics in α-MHC fibers but not in β-MHC fibers. Collectively, our data demonstrate that TnTL71F induces greater contractile deficits against α-MHC than against β-MHC background.




Similar content being viewed by others
References
Becker AE, Caruso G (1982) Myocardial disarray—a critical-review. Brit Heart J 47:527–538
Bulkley BH, Weisfeldt ML, Hutchins GM (1977) Asymmetric septal hypertrophy and myocardial fiber disarray. Features of normal, developing, and malformed hearts. Circulation 56:292–298
Campbell K (1997) Rate constant of muscle force redevelopment reflects cooperative activation as well as cross-bridge kinetics. Biophys J 72:254–262. doi:10.1016/S0006-3495(97)78664-8
Campbell KB, Chandra M, Kirkpatrick RD, Slinker BK, Hunter WC (2004) Interpreting cardiac muscle force–length dynamics using a novel functional model. Am J Physiol Heart Circ Physiol 286:H1535–H1545. doi:10.1152/ajpheart.01029.2003
Chandra M, Tschirgi ML, Tardiff JC (2005) Increase in tension-dependent ATP consumption induced by cardiac troponin T mutation. Am J Physiol Heart Circ Physiol 289:H2112–H2119. doi:10.1152/ajpheart.00571.2005
Chandra M, Tschirgi ML, Ford SJ, Slinker BK, Campbell KB (2007) Interaction between myosin heavy chain and troponin isoforms modulate cardiac myofiber contractile dynamics. Am J Physiol Regul Integr Comp Physiol 293:R1595–1607. doi:10.1152/ajpregu.00157.2007
Chandra V, Gollapudi SK, Chandra M (2015) Rat cardiac troponin T mutation (F72L)-mediated impact on thin filament cooperativity is divergently modulated by alpha- and beta-myosin heavy chain isoforms. Am J Physiol Heart Circ Physiol 309:H1260–H1270. doi:10.1152/ajpheart.00519.2015
Daniel TL, Trimble AC, Chase PB (1998) Compliant realignment of binding sites in muscle: transient behavior and mechanical tuning. Biophys J 74:1611–1621
de Tombe PP, Stienen GJ (1995) Protein kinase A does not alter economy of force maintenance in skinned rat cardiac trabeculae. Circ Res 76:734–741. doi:10.1161/01.RES.76.5.734
Ford SJ, Chandra M (2013) Length-dependent effects on cardiac contractile dynamics are different in cardiac muscle containing alpha- or beta-myosin heavy chain. Arch Biochem Biophys 535:3–13. doi:10.1016/j.abb.2012.10.011
Ford SJ, Chandra M, Mamidi R, Dong W, Campbell KB (2010) Model representation of the nonlinear step response in cardiac muscle. J Gen Physiol 136:159–177. doi:10.1085/jgp.201010467
Ford SJ, Mamidi R, Jimenez J, Tardiff JC, Chandra M (2012) Effects of R92 mutations in mouse cardiac troponin T are influenced by changes in myosin heavy chain isoform. J Mol Cell Cardiol 53:542–551. doi:10.1016/j.yjmcc.2012.07.018
Gollapudi SK, Chandra M (2012) Cardiomyopathy-Related Mutations in Cardiac Troponin C, L29Q and G159D, Have Divergent Effects on Rat Cardiac Myofiber Contractile Dynamics. Biochem Res Int 2012:824068. doi:10.1155/2012/824068
Gollapudi SK, Chandra M (2016) The effect of cardiomyopathy mutation (R97L) in mouse cardiac troponin T on the muscle length-mediated recruitment of crossbridges is modified divergently by alpha- and beta-myosin heavy chain. Arch Biochem Biophys 601:105–112. doi:10.1016/j.abb.2016.01.008
Gollapudi SK, Mamidi R, Mallampalli SL, Chandra M (2012) The N-terminal extension of cardiac troponin T stabilizes the blocked state of cardiac thin filament. Biophys J 103:940–948. doi:10.1016/j.bpj.2012.07.035
Gollapudi SK, Tardiff JC, Chandra M (2015) The functional effect of dilated cardiomyopathy mutation (R144W) in mouse cardiac troponin T is differently affected by alpha- and beta-myosin heavy chain isoforms. Am J Physiol Heart Circ Physiol 308:H884–H893. doi:10.1152/ajpheart.00528.2014
Gordon AM, Homsher E, Regnier M (2000) Regulation of contraction in striated muscle. Physiol Rev 80:853–924
Huxley AF (1957) Muscle structure and theories of contraction. Prog Biophys Biophys Chem 7:255–318
Lu QW, Wu XY, Morimoto S (2013) Inherited cardiomyopathies caused by troponin mutations. J Geriatr Cardiol 10:91–101. doi:10.3969/j.issn.1671-5411.2013.01.014
Luo Y, Cooke R, Pate E (1994) Effect of series elasticity on delay in development of tension relative to stiffness during muscle activation. Am J Physiol 267:C1598–C1606
Lynch TLt et al (2015) Oxidative stress in dilated cardiomyopathy caused by MYBPC3 mutation. Oxid Med Cell Longev 2015:424751. doi:10.1155/2015/424751
Mamidi R, Mallampalli SL, Wieczorek DF, Chandra M (2013) Identification of two new regions in the N-terminus of cardiac troponin T that have divergent effects on cardiac contractile function. J Physiol 591:1217–1234. doi:10.1113/jphysiol.2012.243394
Maron BJ, Sato N, Roberts WC, Edwards JE, Chandra RS (1979) Quantitative analysis of cardiac muscle cell disorganization in the ventricular septum. Comparison of fetuses and infants with and without congenital heart disease and patients with hypertrophic cardiomyopathy. Circulation 60:685–696. doi:10.1161/01.CIR.60.3.685
Martyn DA, Chase PB, Regnier M, Gordon AM (2002) A simple model with myofilament compliance predicts activation-dependent crossbridge kinetics in skinned skeletal fibers. Biophys J 83:3425–3434. doi:10.1016/S0006-3495(02)75342-3
Michael JJ, Gollapudi SK, Chandra M (2014) Effects of pseudo-phosphorylated rat cardiac troponin T are differently modulated by alpha- and beta-myosin heavy chain isoforms. Basic Res Cardiol 109:442. doi:10.1007/s00395-014-0442-9
Montgomery DE, Tardiff JC, Chandra M (2001) Cardiac troponin T mutations: correlation between the type of mutation and the nature of myofilament dysfunction in transgenic mice. J Physiol 536:583–592. doi:10.1111/j.1469-7793.2001.0583c.xd
Moss RL, Razumova M, Fitzsimons DP (2004) Myosin crossbridge activation of cardiac thin filaments: implications for myocardial function in health and disease. Circ Res 94:1290–1300. doi:10.1161/01.RES.0000127125.61647.4F
Nakayama Y, Shimizu G, Hirota Y, Saito T, Kino M, Kitaura Y, Kawamura K (1987) Functional and histopathologic correlation in patients with dilated cardiomyopathy: an integrated evaluation by multivariate analysis. J Am Coll Cardiol 10:186–192. doi:10.1016/S0735-1097(87)80178-X
Razumova MV, Bukatina AE, Campbell KB (2000) Different myofilament nearest-neighbor interactions have distinctive effects on contractile behavior. Biophys J 78:3120–3137. doi:10.1016/S0006-3495(00)76849-4
Rundell VL, Manaves V, Martin AF, de Tombe PP (2005) Impact of beta-myosin heavy chain isoform expression on cross-bridge cycling kinetics. Am J Physiol Heart Circ Physiol 288:H896–H903. doi:10.1152/ajpheart.00407.2004
Stelzer JE, Brickson SL, Locher MR, Moss RL (2007) Role of myosin heavy chain composition in the stretch activation response of rat myocardium. J Physiol 579:161–173. doi:10.1113/jphysiol.2006.119719
Stienen GJ, Zaremba R, Elzinga G (1995) ATP utilization for calcium uptake and force production in skinned muscle fibres of Xenopus laevis. J Physiol 482(Pt 1):109–122. doi:10.1113/jphysiol.1995.sp020503
Tardiff JC et al (1998) A truncated cardiac troponin T molecule in transgenic mice suggests multiple cellular mechanisms for familial hypertrophic cardiomyopathy. J Clin Inves 101:2800–2811. doi:10.1172/JCI2389
Varnava AM, Elliott PM, Baboonian C, Davison F, Davies MJ, McKenna WJ (2001) Hypertrophic cardiomyopathy: histopathological features of sudden death in cardiac troponin T disease. Circulation 104:1380–1384. doi:10.1161/hc3701.095952
Xu Q, Dewey S, Nguyen S, Gomes AV (2010) Malignant and benign mutations in familial cardiomyopathies: insights into mutations linked to complex cardiovascular phenotypes. J Mol Cell Cardiol 48:899–909. doi:10.1016/j.yjmcc.2010.03.005
Funding
This work was supported, in part, by National Institutes of Health Grant No. HL-075643 (to Dr. Murali Chandra).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Reda, S.M., Gollapudi, S.K. & Chandra, M. L71F mutation in rat cardiac troponin T augments crossbridge recruitment and detachment dynamics against α-myosin heavy chain, but not against β-myosin heavy chain. J Muscle Res Cell Motil 37, 215–223 (2016). https://doi.org/10.1007/s10974-016-9460-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10974-016-9460-6