Abstract
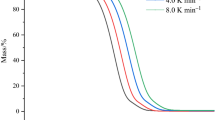
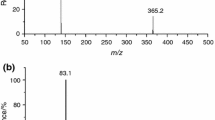
Due to their numerous properties as an advanced or specialized material, ionic liquids (ILs) have found an increasingly wide utilization in many fields. Moreover, mixtures of ILs can further improve their correlative properties. Therefore, it is significant that the thermal stability and exothermic behaviour of IL binary mixtures were studied. In this work, two typical representative imidazole ILs ([BMIM]BF4 and BMIM[NO3]) were selected, and their binary mixture thermal stability and thermal decomposition process of visualization were investigated by simultaneous thermogravimetric analyser (STA). Furthermore, their binary mixture exothermic behaviour and decomposition products were obtained by differential scanning calorimetry (DSC) and thermogravimetry coupled with Fourier transform infrared spectroscopy (TG-FTIR), respectively. The compound mode of imidazolium-based IL binary mixtures was divided into three categories: the molar fraction (7:3, 1:1, and 3:7) of parent pure ILs. We found that TG curves trend of IL binary mixtures was consistent with their parent pure ILs. Compared with TG curves, DSC curves of IL mixtures demonstrated that the behaviours of heat absorption and heat release at greater than 700.0 K in mixtures were different from their parent pure ILs. According to the infrared spectrum of gas products, the oxygen-containing functional group in BMIM[NO3] is likely to have some influence on the second-stage decomposition of sample 1#. Systematic thermal analysis results provided a significant reference for the exothermic behaviours and thermal degradation process of imidazolium-based ionic liquid binary mixture and a feasible research route for further investigations into the thermal decomposition characteristics of IL mixtures.








Similar content being viewed by others
References
Rogers RD, Seddon KR. Ionic liquids-solvents of the future? Science. 2003;302:792–3.
Yu CF, Liu SH, Xia R, Wu KF. Studies on the thermal stability and decomposition kinetics of 1-ethyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide via density functional theory and experimental methods. J Mole Liq. 2022;306:119422.
Wilkes JS. A short history of ionic liquids—from molten salts to neoteric solvents. Green Chem. 2002;4:73–80.
Liaw HJ, Chen CC, Chen YC, Chen JR, Huang SK, Liu SN. Relationship between flash point of ionic liquids and their thermal decomposition. Green Chem. 2012;14:2001–8.
Stepan AM, Michud A, Helisten S, Hummel M, Sixta H. IONCELL-P&F: pulp fractionation and fiber spinning with ionic liquids. Ind Eng Chem Res. 2016;55:8225–33.
Wang WT, Liu SH, Wang Y, Yu CF, Cheng YF, Shu CM. Thermal stability and exothermic behaviour of imidazole ionic liquids with different anion types under oxidizing and inert atmospheres. J Mole Liq. 2021;343:117691.
Patinha DJS, Tome LC, Florindo C, Soares HR, Coroadinha AS, Marrucho IM. New low-toxicity cholinium-based ionic liquids with perfluoroalkanoate anions for aqueous biphasic system implementation. ACS Sustain Chem Eng. 2016;4:2670–9.
Barreto GP, Rodríguez KD, Morales GE, Enríquez MFJ, Cañizo AI, Eyler GN. Effect of ionic liquid on the thermal decomposition of cyclic organic peroxides. Arab J Chem. 2019;12:4277–86.
Wang Y, Liu SH, Chiang CL, Zhang LY, Wang WT. The effect of oxygen on the thermal stability and decomposition behaviours of 1,3-dimethylimidazolium nitrate for application using STA, ARC and FTIR. Process Saf Environ Prot. 2022;162:513–9.
Bai Z, Wang C, Deng J, Kang F, Shu CM. Experimental investigation on using ionic liquid to control spontaneous combustion of lignite. Process Saf Environ Prot. 2020;142:138–49.
Chatel G, Pereira JFB, Debbeti V, Wang H, Rogers RD. Mixing ionic liquids – “simple mixtures” or “double salts”? Green Chem. 2014;16:2051–83.
Kemperman GJ, Roeters TA, Hilberink PW. Cleavage of aromatic methyl ethers by chloroaluminate ionic liquid reagents. J Org Chem. 2003;9:1681–6.
Studzińska S, Kowalkowski T, Buszewski B. Study of ionic liquid cations transport in soil. J Hazard Mater. 2009;168:1542–7.
Gao Y, Cheng L, Gao R, Hu G, Zhao J. Deep desulfurization of fuels using supported ionic liquid-polyoxometalate hybrid as catalyst: a comparison of different types of ionic liquids. J Hazard Mater. 2021;401:123267.
Wang WT, Liu SH, Wang Y, Yu CF, Cheng YF, Shu CM. Thermal stability and exothermic behaviour of imidazole ionic liquids with different anion types under oxidising and inert atmospheres. J Mole Liq. 2021;343:117691.
Liu SH, Zhang B, Cao CR. Evaluation of thermal properties and process hazard of three ionic liquids through thermodynamic calculations and equilibrium methods. J Loss Prev Process Ind. 2020;68:104332.
Parajó JJ, Teijeira T, Fernández J, Salgado J, Villanueva M. Thermal stability of some imidazolium [NTf2] ionic liquids: Isothermal and dynamic kinetic study through thermogravimetric procedures. The J Chem Therm. 2017;112:105–13.
Smoll EJ, Tesa-Serrate MA, Purcell SM, D’Andrea L, Bruce DW, Slattery JM, Costen ML, Minton TK, McKendrick KG. Determining the composition of the vacuum-liquid interface in ionic-liquid mixtures. Faraday Discuss. 2018;206:497–522.
Barreto G, Eyler G. Thermal decomposition of 3,3,6,6,9,9-hexaethyl-1,2,4,5,7,8-hexaoxacyclononane in solution and its use in methyl methacrylate polymerization. Polym Bull. 2011;67:1–14.
Ahmed HM, Kamal MS, Al-Harthi M. Polymeric and low molecular weight shale inhibitors: a review. Fuel. 2019;251:187–217.
Kunze M, Jeong S, Appetecchi GB, Schonhoff M, Winter M, Passerini S. Mixtures of ionic liquids for low temperature electrolytes. Electrochim Acta. 2012;82:69–74.
Huang GS, Lin WC, He P, Pan Y, Shu CM. Thermal decomposition of imidazolium-based ionic liquid binary mixture: Processes and mechanisms. J Mole Liq. 2018;272:37–42.
Navarro P, Larriba M, García J, Rodríguez F. Thermal stability, specific heats, and surface tensions of ([emim][DCA] + [4empy][Tf2N]) ionic liquid mixtures. J Chem Thermodyn. 2014;76:152–60.
Pinto AM, Rodríguez H, Colón YJ, Arce A, Soto A. Absorption of carbon dioxide in two binary mixtures of ionic liquids. Ind Eng Chem Res. 2013;52:5975–84.
Fox DM, Awad WH, Gilman JW, Maupin PH, Long HCD, Trulove PC. Flammability, thermal stability, and phase change characteristics of several trialkylimidazolium salts. Green Chem. 2003;5:724–7.
Navarro P, Larriba M, García J, Rodríguez F. Thermal stability and specific heats of [emim][DCA] + [emim][TCM] mixed ionic liquids. Thermochim Acta. 2014;588:22–7.
Xu RJ, Liu SH, Wu KF, Yu CF. Effect of 1-butyl-3-MethylImidazolium bis(trifluoromethylsulfonyl)imide on spontaneous combustion of bituminous coal. J Therm Anal Calorim. 2023;148:4707–15.
Wu KF, Liu SH. Effect of two imidazole ionic liquids on the thermal stability of cumene hydroperoxide. J Therm Anal Calorim. 2023;148:5051–62.
Chen KY, Wu SH, Wang YW, Shu CM. Runaway reaction and thermal hazards simulation of cumene hydroperoxide by DSC. J Loss Prev Process Ind. 2008;21:101–9.
Qu BB, Liu SH, Guo RL, Chiang CL. Using dynamic and adiabatic methods for thermal hazard evaluation of styrene bulk polymerization initiated by AIBN. J Therm Anal Calorim. 2023;148:4791–9.
Xia R, Liu SH, Wang WT, et al. Influence of oxidizing gas atmosphere on thermal stability and safety risk of 1-buty-3-methylimidazolium tetrafluoroborate. J Therm Anal Calorim. 2023;148:4717–27.
Acknowledgements
The authors are grateful for the financial support from the National Natural Science Foundation of China, under Contract Number 52104177.
Author information
Authors and Affiliations
Contributions
S-HL: Writing—Original draft preparation (lead), Writing—Review & editing (lead), Visualization (lead), Funding Acquisition (lead). K-FW: Writing—Review & editing (supporting), Methodology (lead), Formal Analysis (supporting). R-JX: Methodology (supporting), Formal Analysis (supporting). C-FY: Editing (supporting), Methodology (supporting). YW: Editing (supporting), Formal Analysis (supporting).
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interest or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, SH., Wu, KF., Xu, RJ. et al. Studies on the thermal stability and exothermic behaviour of imidazolium-based ionic liquid binary mixture. J Therm Anal Calorim (2023). https://doi.org/10.1007/s10973-023-12702-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10973-023-12702-3