Abstract
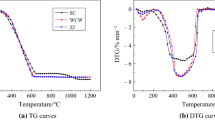
The study on CO2 gasification behavior helps to promote the clean and efficient utilization of high-alkali fuels. The high-alkali fuels feature high contents of alkali metals, and sometimes they are also rich in alkali earth metals and iron. Though the influences of minerals on gasification characteristics have been extensively studied, few researchers have selected minerals based on the ash composition of high-alkali fuel. Moreover, the complex interactions among minerals could lead to inaccurate conclusions. In this paper, the CO2 gasification behavior of chars originated from one high-alkali biomass and three high-alkali coals was studied. The effects of principal minerals related to the high-alkali fuels, which were determined through the new plasma ashing method and traditional muffle ashing method, were also further evaluated via synthetic coal char. The synthetic coal char was free from any intrinsic minerals. The results indicate that the gasification reactivities of chars from high-alkali fuels are positively associated with the reaction temperature (900–1200 °C). The biomass char possesses the highest gasification reactivity, and three coal char samples are inferior to various degrees. The related chemicals offer their catalytic activities at 1000 °C in the sequence of K/Na-containing chemicals > Fe-containing chemicals > Ca-containing chemicals. The shrinking core model (SCM) and two-dimensional growth of nuclei model (2DGM) were chosen to conduct the kinetic analysis. The reaction constants of chars from different high-alkali fuels agree with their gasification behavior. Generally, the 2DGM is more suitable than SCM to predict the conversion of selected char samples. In most cases, the additions of chemicals increase the reaction constants. When Fe2O3 and CaCO3 are added into synthetic coal char, the SCM is more accurate to describe the gasification behavior at 900 °C, but the addition of Na2SO4 makes the 2DGM a better one.









Similar content being viewed by others
References
Niu YQ, Tan HZ, Hui SE. Ash-related issues during biomass combustion: alkali-induced slagging, silicate melt-induced slagging (ash fusion), agglomeration, corrosion, ash utilization, and related countermeasures. Prog Energy Combust Sci. 2016;52:1–61.
Demirbas A. Combustion characteristics of different biomass fuels. Prog Energy Combust Sci. 2004;30(2):219–30.
Zhang S, Chen C, Shi D, Lü J, Wang J, Guo X, et al. Situation of combustion utilization of high sodium coal. Proc CSEE. 2013;33:1–12.
Safarian S, Unnþórsson R, Richter C. A review of biomass gasification modelling. Renew Sustain Energy Rev. 2019;110:378–91.
Toftegaard MB, Brix J, Jensen PA, Glarborg P, Jensen AD. Oxy-fuel combustion of solid fuels. Prog Energy Combust Sci. 2010;36(5):581–625.
Sun LT, Sun R, Yan YH, Yuan MF, Wu JQ. Investigation of air-MILD and oxy-MILD combustion characteristics of semicoke and bituminous coal mixtures in a 0.3 MW fuel-rich/lean fired furnace. Fuel Process Technol. 2022;231:107247.
Puig-Arnavat M, Bruno JC, Coronas A. Review and analysis of biomass gasification models. Renew Sustain Energy Rev. 2010;14(9):2841–51.
Xiang YL, Cai L, Guan YW, Liu WB, Cheng ZY, Liu ZX. Study on the effect of gasification agents on the integrated system of biomass gasification combined cycle and oxy-fuel combustion. Energy. 2020;206: 118131.
Chen DG, Zhang Z, Li ZS, Lv Z, Cai NS. Optimizing in-situ char gasification kinetics in reduction zone of pulverized coal air-staged combustion. Combust Flame. 2018;194:52–71.
Ding L, Dai ZH, Wei JT, Zhou ZJ, Yu GS. Catalytic effects of alkali carbonates on coal char gasification. J Energy Inst. 2017;90(4):588–601.
Mei YG, Wang ZQ, Fang YT, Huang JJ, Li WZ, Guo S, et al. CO2 catalytic gasification with NaAlO2 addition for its low-volatility and tolerant to deactivate. Fuel. 2019;242:160–6.
Ohtsuka Y, Tomita A. Calcium catalysed steam gasification of Yallourn brown coal. Fuel. 1986;65(12):1653–7.
Lahijani P, Zainal ZA, Mohamed AR, Mohammadi M. CO2 gasification reactivity of biomass char: catalytic influence of alkali, alkaline earth and transition metal salts. Bioresour Technol. 2013;144:288–95.
Zhang JB, Jiang PP, Gao FL, Ren ZY, Li R, Chen HY, et al. Fuel gas production and char upgrading by catalytic CO2 gasification of pine sawdust char. Fuel. 2020;280: 118686.
Zhang F, Xu DP, Wang YG, Argyle MD, Fan MH. CO2 gasification of Powder River Basin coal catalyzed by a cost-effective and environmentally friendly iron catalyst. Appl Energy. 2015;145:295–305.
Wang CA, Zhu X, Liu X, Lv Q, Zhao L, Che DF. Correlations of chemical properties of high-alkali solid fuels: a comparative study between Zhundong coal and biomass. Fuel. 2018;211:629–37.
Guo S, Jiang YF, Liu T, Zhao JT, Huang JJ, Fang YT. Investigations on interactions between sodium species and coal char by thermogravimetric analysis. Fuel. 2018;214:561–8.
Wang YW, Wang ZQ, Huang JJ, Fang YT. Investigation into the characteristics of Na2CO3-catalyzed steam gasification for a high-aluminum coal char. J Therm Anal Calorim. 2018;131(2):1213–20.
Wang CA, Zhao L, Liu CC, Gao XY, Li GY, Che DF. Comparative study on ash characteristics of various high-alkali fuels using different ashing methods and temperatures. Fuel. 2021;299: 120912.
Qin YH, He YY, Ren WP, Gao MJ, Wiltowski T. Catalytic effect of alkali metal in biomass ash on the gasification of coal char in CO2. J Therm Anal Calorim. 2020;139(5):3079–89.
Mukherjee S, Srivastava SK. Minerals transformations in northeastern region coals of India on heat treatment. Energy Fuels. 2006;20(3):1089–96.
Wang CA, Zhao L, Yuan MB, Liu CC, Wang CW, Zhao L, et al. Effects of ashing method and blending on ash characteristics of pyrolyzed and gasified semi-cokes. Fuel. 2020;271: 117607.
Wang CA, Zhao L, Yuan MB, Du YB, Zhu CZ, Liu YH, et al. Effects of minerals containing sodium, calcium, and iron on oxy-fuel combustion reactivity and kinetics of Zhundong coal via synthetic coal. J Therm Anal Calorim. 2020;139(1):261–71.
Wang CA, Du YB, Che DF. Reactivities of coals and synthetic model coal under oxy-fuel conditions. Thermochim Acta. 2013;553:8–15.
Xu J, Yang Y, Li YW. Recent development in converting coal to clean fuels in China. Fuel. 2015;152:122–30.
Kok MV. Simultaneous thermogravimetry–calorimetry study on the combustion of coal samples: Effect of heating rate. Energy Convers Manag. 2012;53(1):40–4.
Kok MV, Topa E. Thermal characterization and model-free kinetics of biodiesel sample. J Therm Anal Calorim. 2015;122(2):955–61.
Xin HH, Wang HT, Kang WJ, Di CC, Qi XY, Zhong XX, et al. The reburning thermal characteristics of residual structure of lignite pyrolysis. Fuel. 2020;259: 116226.
Zhao RD, Qin JG, Chen TJ, Wang LL, Wu JH. Experimental study on co-combustion of low rank coal semicoke and oil sludge by TG-FTIR. Waste Manag. 2020;116:91–9.
Yadav D, Saha S, Sahu G, Chavan PD, Datta S, Chauhan V, et al. A comparative review on thermal behavior of feedstocks during gasification via thermogravimetric analyzer. J Therm Anal Calorim. 2023;148(2):329–54.
Wang CA, Feng QQ, Mao QS, Wang CW, Li GY, Che DF. Oxy-fuel co-combustion performances and kinetics of bituminous coal and ultra-low volatile carbon-based fuels. Int J Energy Res. 2021;45(2):1892–907.
Zuo HB, Geng WW, Zhang JL, Wang GW. Comparison of kinetic models for isothermal CO2 gasification of coal char-biomass char blended char. Int J Miner, Metall, Mater. 2015;22(4):363–70.
He Q, Guo QH, Ding L, Wei JT, Yu GS. CO2 gasification of char from raw and torrefied biomass: Reactivity, kinetics and mechanism analysis. Bioresour Technol. 2019;293: 122087.
Saha S, Sahu G, Chavan PD, Datta S. Gasification reactivity of high ash Indian coals in varying concentrations of CO2. Int J Oil, Gas Coal Technol. 2018;18(1–2):163–86.
Li L, Wang ZQ, Zhao R, Mei YG, Shi WJ, Liu ZY, et al. The different catalytic effects of Na species on char gasification and the reasons for this different. J Therm Anal Calorim. 2022;147(10):5687–99.
Yan XY, Hu JJ, Zhang QG, Zhao SH, Dang JT, Wang W. Chemical-looping gasification of corn straw with Fe-based oxygen carrier: thermogravimetric analysis. Bioresour Technol. 2020;303: 122904.
Fermoso J, Gil MV, Pevida C, Pis JJ, Rubiera F. Kinetic models comparison for non-isothermal steam gasification of coal–biomass blend chars. Chem Eng J. 2010;161(1):276–84.
Wang GW, Zhang JL, Hou XM, Shao JG, Geng WW. Study on CO2 gasification properties and kinetics of biomass chars and anthracite char. Bioresour Technol. 2015;177:66–73.
Hancock J, Sharp J. Method of comparing solid-state kinetic data and its application to the decomposition of kaolinite, brucite, and BaCO3. J Am Ceram Soc. 2006;55:74–7.
Hu Q, Yang HP, Xu HS, Wu ZQ, Lim CJ, Bi XT, et al. Thermal behavior and reaction kinetics analysis of pyrolysis and subsequent in-situ gasification of torrefied biomass pellets. Energy Convers Manag. 2018;161:205–14.
Wang T, Tang LF, Raheem A, Chen XL, Wang FC. Study on CO2 gasification characteristics of pyrolysis char from pinewood block and pellet. Biomass Convers Biorefin 2021.
Liu HM, Sullivan RM, Hanson JC, Grey CP, Martin JD. Kinetics and mechanism of the β- to α-CuAlCl4 phase transition: A time-resolved 63Cu MAS NMR and powder X-ray diffraction study. J Am Chem Soc. 2001;123(31):7564–73.
Piotrowski K, Mondal K, Wiltowski T, Dydo P, Rizeg G. Topochemical approach of kinetics of the reduction of hematite to wüstite. Chem Eng J. 2007;131(1):73–82.
Betancur M, Natalia Arenas C, Daniel Martínez J, Victoria Navarro M, Murillo R. CO2 gasification of char derived from waste tire pyrolysis: kinetic models comparison. Fuel. 2020;273:117745.
Kumari N, Saha S, Sahu G, Chauhan V, Roy R, Datta S, et al. Comparison of CO2 gasification reactivity and kinetics: petcoke, biomass and high ash coal. Biomass Convers Biorefin. 2022;12(6):2277–90.
Wang G, Zhang J, Hou X, Shao J, Geng W. Study on CO2 gasification properties and kinetics of biomass chars and anthracite char. Biores Technol. 2015;177:66–73.
Mathews JP, Krishnamoorthy V, Louw E, Tchapda AHN, Castro-Marcano F, Karri V, et al. A review of the correlations of coal properties with elemental composition. Fuel Process Technol. 2014;121:104–13.
Krishnamoorthy V, Pisupati SV. A critical review of mineral matter related issues during gasification of coal in fixed, fluidized, and entrained flow gasifiers. Energies. 2015;8(9):10430–63.
Li C, Wang Y, Lin X, Tian Z, Wu X, Yang Y, et al. Influence of inherent minerals on CO2 gasification of a lignite with high ash content. J Fuel Chem Technol. 2017;45(7):780–8.
Lü L, Li C, Zhang GQ, Hu XW, Liang B. Decomposition behavior of CaSO4 during potassium extraction from a potash feldspar-CaSO4 binary system by calcination. Chin J Chem Eng. 2018;26(4):838–44.
Szekely J, Evans JW. A structural model for gas-solid reactions with a moving boundary. Chem Eng Sci. 1970;25(6):1091–107.
Adamon DGF, Fagbémi LA, Bensakhria A, Sanya EA. Comparison of kinetic models for carbon dioxide and steam gasification of rice husk char. Waste Biomass Valoriz. 2019;10(2):407–15.
Acknowledgements
The authors acknowledge the financial support from the National Natural Science Foundation of China (No. 52176129).
Author information
Authors and Affiliations
Contributions
Conceptualization: LZ, CW; Data curation: LZ; Formal analysis: LZ; Funding acquisition: CW, DC; Investigation: LZ, ML, PZ; Methodology: LZ; Project administration: CW, DC; Resources: CW, DC; Software: LZ; Supervision: CW; Validation: CW; Visualization: LZ; Writing—Original Draft: LZ; Writing—Review and Editing: CW.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhao, L., Wang, C., Luo, M. et al. CO2 gasification behavior of chars from high-alkali fuels and effects of Na, K, Ca, and Fe species via synthetic coal char. J Therm Anal Calorim 148, 6423–6437 (2023). https://doi.org/10.1007/s10973-023-12159-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-023-12159-4