Abstract
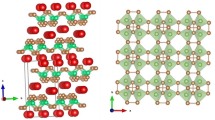
In this study, (C2H5NH3)2CdCl4 single crystals were grown, and their thermal properties were studied by the thermogravimetric analysis and differential scanning calorimetry. Phase transition temperature above room temperature was observed at 470 K. In addition, the cation dynamics in this crystal were investigated by 1H magic-angle spinning nuclear magnetic resonance (MAS NMR) and 13C cross-polarization (CP)/MAS NMR experiments. The Bloembergen–Purcell–Pound curves for 1H T1ρ and 13C T1ρ in C2H5NH3 cation exhibited minimum values, and these curves represent C2H5NH3 and C2H5 rotational motions. The activation energy for 1H in the C2H5NH3 cation is Ea = 22.63 kJ mol−1, whereas those for 13C in CH3 and CH2 are Ea = 18.05 and 19.14 kJ mol−1, respectively. Furthermore, the proton dynamics of C2H5NH3 undergo faster rotation than carbons. This implies that the molecular motion for 1H is enhanced at the C-end and N-end of an organic cation, whereas the molecular motion is not free at the main chain carbons of the organic cation.







Similar content being viewed by others
References
Elwej R, Hannachi N, Chaabane I, Oueslati A, Hlel F. Structural, characterization and AC conductivity of bis-2-amino-6-picolinium tetrachloromercurate, (C6H9N2)2HgCl4. Inorg Chim Acta. 2013;406:10.
Oueslati A, Bulou A, Calvayrac F, Adil K, Gargouri M, Hlel F. Infrared, polarized Raman and ab initio calculations of the vibrational spectra of [N(C3H7)4]2Cu2Cl6 crystals. Vib Spectrosc. 2013;64:10–20.
Oliveira LH, Moura AP, Longo E, Varela JA, Rosa ILV. Luminescent properties of hybrid materials prepared by the polymeric precursor method. J Alloys Compd. 2013;579:227–35.
Hajlaoui S, Chaabane I, Oueslati A, Guidara K, Bulou A. A theoretical study on the molecular structure and vibrational (FT-IR and Raman) spectra of new organic-inorganic compound [N(C3H7)4]2SnCl6. Spectrochim Acta Part A. 2014;117:225–33.
Lefi R, Naser FB, Guermazi H. Structural, optical properties and characterization of (C2H5NH3)2CdCl4, (C2H5NH3)2CuCl4 and (C2H5NH3)2Cd0.5Cu0.5Cl4 compounds. J Alloys Compd. 2017;696:1244.
Nasr FB, Lefi R, Guermazi H. Analysis of high temperature phase transitions of copper doped (C2H5NH3)2CdCl4 perovskite. J Mol Struct. 2018;1165:236.
Ohnishi A, Tanaka KI, Kitaura M, Otomo T, Yoshinari T. Optical spectra of inorganic–organic compounds (C2H5NH3)2CdCl4 in 3–30 eV range. J Phys Soc Jpn. 2001;70:3424–7.
Elseman AM, Shalan AE, Sajid S, Rashad MM, Hassan AM, Li M. Copper-substituted lead perovskite materials constructed with different halides for working (CH3NH3)2CuX4-based perovskite solar cells from experimental and theoretical view. ACS Appl Mater Interfaces. 2018;10:11699.
Aramburu JA, Garcia-Fernandez P, Mathiesen NR, Garcia-Lastra JM, Moreno M. Changing the usual interpretation of the structure and ground state of Cu2+-layered perovskite. J Phys Chem C. 2018;122:5071.
Geick R, Strobel K. Symmetry of the lattice vibrations in perovskite-type layer structures. J Phys C Solid State Phys. 1977;10:4221–39.
Peyrard M, Remoissenet M. Optical phonons line broadening at the first order phase transition of (C2H5NH3)2CdCl4: a spin-phonon coupled system. J Chem Phys. 1979;71:2732.
Levstik A, Filipic C. Dielectric study of the (C2H5NH3)2CdCl4 monocrystal. Phys Stat Sol (a). 1980;58:k165.
Yoshinari T, Matsuyama T, Yamaoka H, Aoyagi K. Optical properties of electron trapped center in layered ionic crystals (CH3NH3)2CdCl4 and (C2H5NH3)2CdCl4 irradiated with X-rays at 15 K. J Phys Soc Jpn. 1989;58:4222.
Moral BA, Rodriguez F. Dependence of the change transfer spectra of (C2H5NH3)2CdCl4:Cu2+ with hydrogstatic pressure:structural changes…. J Phys Chem Solids. 1997;58:1487.
Ohnishi A, Tanaka KI, Yoshinari T. Exciton self-trapping in two dimensional system of (C2H5NH3)2CdCl4 single crystal. J Phys Soc Jpn. 1999;68:288.
Ohwada K, Ishii K, Inami T, Murakami Y, Shobu T, Ohsumi H, Ikeda N, Ohishi Y. Structural properties and phase transition of hole-orbital-ordered (C2H5NH3)2CuCl4 studied by resonant and non-resonant X-ray scatterings under high pressure. Phys Rev B. 2005;72:14123–33.
Rao CNR, Cheetham AK, Thirumurugan A. Hybrid inorganic–organic materials: a new family in condensed matter physics. J Phys Condens Matter. 2008;20:083202.
Zhang W, Xiong RG. Ferroelectric metal–organic frameworks. Chem Rev. 2012;112:1163–95.
Mohamed CB, Karoui K, Saidi S, Guidara K, Rhaiem AB. Electrical properties, phase transitions and conduction mechanisms of the [(C2H5)NH3]2CdCl4 compound. Physica B. 2014;451:87–95.
Yadav R, Swain D, Kundu PP, Nair HS, Narayana C, Elizabeth S. Dielectric and Raman investigations of structural phase transitions in (C2H5NH3)2CdCl4. Phys Chem Chem Phys. 2015;17:12207.
Chapuis G. X-ray study of the first-order phase transition Pcab-Bmab in (CH3CH2NH3)2CdCl4. Phys Stat Sol (a). 1977;43:203.
Hagemann H, Bill H. Raman investigation on structural phase transitions in (C2H5NH3)2CdCl4. Chem Phys Lett. 1982;93:582–5.
Lim AR, Kim SW, Joo YL. Structural phase transitions and ferroelastic properties of perovskite-type layered (CH3NH3)2CdCl4. J Appl Phys. 2017;121:215501.
Wieser ME. Atomic weights of the elements 2005 (IUPAC technical report). Pure Appl Chem. 2005;78:2051–66.
Koenig JL. Spectroscopy of polymers. New York: Elsevier; 1999.
Mcbrierty VJ, Packer KJ. Nuclear magnetic resonance in solid polymers. Cambridge: Cambridge University Press; 1993.
Abragam A. The principles of nuclear magnetism. Oxford: Oxford University Press; 1961.
Acknowledgements
This research was supported by the Basic Science Research program through the National Research Foundation of Korea (NRF), funded by the Ministry of Education (2018R1D1A1B07041593).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors decalre that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lim, A.R. Thermal decomposition and structural dynamics in perovskite (C2H5NH3)2CdCl4 crystals. J Therm Anal Calorim 142, 2243–2249 (2020). https://doi.org/10.1007/s10973-019-09187-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-019-09187-4