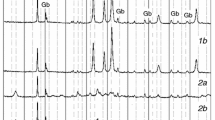
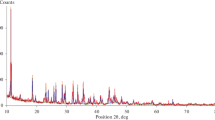
Abstract
Thermal decomposition mechanisms for cobaltoblödite Na2Co(SO4)2·4H2O and cupper–kröhnkite Na2Cu(SO4)2·2H2O have been developed using constant rate thermal analysis technique under controlled residual water vapor pressure above the sample \( P_{{{\text{H}}_{2} {\text{O}}}} = \, 5\;{\text{hPa}} \). The apparent activation energy of each dehydration process was measured by means of two CRTA curves without any presumption to the kinetic low. Correlation between structure and thermal behavior was highlighted. It has shown that dehydration of cobaltoblödite mineral occurs in two steps with formation of Na2Co(SO4)2·1.5H2O and Na2Co(SO4)2 as intermediate and final phases, respectively. The activation energies of both dehydration steps are 167 kJ mol−1 and 107 kJ mol−1, respectively. For cupper–kröhnkite mineral, its dehydration occurs in one step leading to Na2Cu(SO4)2. The corresponding activation energy was found equal to 71 kJ mol−1.












Similar content being viewed by others
References
Hoffmann SK, Goslar J, Hilczer W, Augustyniak-Jabłokow MA. Electron spin relaxation of vibronic Cu(H2O)6 complexes in K2Zn(SO4)2·6H2O single crystals. J Phys Condens Matter. 2001;13:707–18.
Mills SJ, Hatert F, Nickel EH, Ferraris G. The standardisation of mineral group hierarchies: application to recent nomenclature proposals. Eur J Mineral. 2009;21:1073–80.
Palache C, Warren CH. Krohnkite, natrochalcite (a new mineral), and other sulphates from Chile. Am J Sci. 1908;154:342–8.
Souamti A, Martín IR, Zayani L, Hernández-Rodríguez MA, Soler-Carracedo K, Lozano-Gorrín AD, Chehimi DBH. Blue up-conversion emission of Yb3+-doped langbeinite salts. Opt Mater. 2016;53:190–4.
Souamti A, Martín IR, Zayani L, Hernández-Rodríguez MA, Soler-Carracedo K, Lozano-Gorrín AD, Lalla E, Chehimi DBH. Synthesis, characterization and spectroscopic properties of a new Nd3+-doped Co-picromerite-type Tutton salt. J Lumin. 2016;177:93–8.
Souemti A, Lozano-Gorrín AD, Zayani L, Essayes SA, Abbassi M, Lalla E, Pérez-Rodríguez C, Chehimi DBH. Synthesis, characterization and electrical properties of both pure and cobalt-doped picromerite-type hydrated double salt K2Mg1−xCox(SO4)2·6H2O (x = 0, 0.4). J Electron Mater. 2016;45:4418–24.
Souamti A, Martín IR, Zayani L, Lozano-Gorrín AD, Chehimi DBH. Luminescence properties of Pr3+ ion doped Mg-picromerite Tutton salt. J Lumin. 2017;188:148–53.
Béjaoui O, Zayani L, Chehimi DBH. Synthesis and thermal behaviour of Na2SO4·MgSO4·4H2O. J Therm Anal Calorim. 2016;123:1205–11.
Reynaud M, Ati M, Boulineau S, Sougrati MT, Melot BC, Rousse G, Chotard JN, Tarascon JM. Bimetallic sulfates A2M(SO4)2·nH2O (A = Li, Na and M = transition metal): as new attractive electrode materials for Li-and Na-ion batteries. ECS Trans. 2013;50:11–9.
Van Tiggelen PJ. Détermination Par Résonance Magnétique Nucléaire de la Position des Atomes d’Hydrogène dans la Kröhnkite. Bull Soc Chim Belg. 1966;75:620–40.
Hawthorne FC, Ferguson RB. Refinement of the crystal structure of kröhnkite. Acta Crystallogr Sect B Struct Crystallogr Cryst Chem. 1975;31:1753–5.
Kasatkin AV, Nestola F, Plášil J, Marty J, Belakovskiy DI, Agakhanov AA, Mills SJ, Pedron D, Lanza A, Favaro M, Bianchin S, Lykova IS, Goliáš V, Birch WD. Manganoblödite, Na2Mn(SO4)2·4H2O and cobaltoblödite Na2Co(SO4)2·4H2O: two new members of the blödite group from the Blue Lizard mine, San Juan County, Utah, USA. Mineral Mag. 2013;77:367–83.
Stoilova D, Wildner M. Blödite-type compounds Na2Me(SO4)2·4H2O (Me = Mg Co, Ni, Zn): crystal structures and hydrogen bonding systems. J Mol Struct. 2004;706:57–63.
Sarma CRN, Satyanandam G, Murthy PG, Haranadh C. EPR investigation of krohnkite, Na2Cu(SO4)2·2H2O. J Phys C Solid State Phys. 1976;9:841–7.
Rouquerol J. Controlled transformation rate thermal analysis: the hidden face of thermal analysis. Thermochim Acta. 1989;144:209–24.
Rouquerol J, Bordère S, Rouquerol F. Controlled rate evolved gas analysis: recent experimental set-up and typical results. Thermochim Acta. 1992;203:193–202.
Rouquerol J. A general introduction to SCTA and to rate-controlled SCTA. J Therm Anal Calorim. 2003;72:1081–6.
Vágvölgyi V, Daniel L, Pinto C, Kristóf J, Frost R, Horváth E. Dynamic and controlled rate thermal analysis of attapulgite. J Therm Anal Calorim. 2008;92:589–94.
Vágvölgyi V, Frost R, Hales M, Locke A, Kristóf J, Horváth E. Controlled rate thermal analysis of hydromagnesite. J Therm Anal Calorim. 2008;92:893–7.
Vágvölgyi V, Hales M, Martens W, Kristóf J, Horváth E, Frost RL. Dynamic and controlled rate thermal analysis of hydrozincite and smithsonite. J Therm Anal Calorim. 2008;92:911–6.
Nahdi K, Rouquerol F, Ayadi MT. Mg(OH)2 dehydroxylation: a kinetic study by controlled rate thermal analysis (CRTA). Solid State Sci. 2009;11:1028–34.
Nahdi K, Ferid M, Ayadi MT. Thermal dehydration of CeP3O9·3H2O by controlled rate thermal analysis. J Therm Anal Calorim. 2009;96:455–61.
Nahdi K, Ferid M, Ayadi MT. Chemical preparation and thermal behavior of neodymium cyclotriphosphate pentahydrate NdP3O9·5H2O: a study by controlled rate thermal analysis (CRTA). Thermochim Acta. 2009;487:54–9.
Dhandapani M, Thyagu L, Prakash PA, Amirthaganesan G, Kandhaswamy MA, Srinivasan V. Synthesis and characterization of potassium magnesium sulphate hexahydrate crystals. Cryst Res Technol. 2006;41:328–31.
Acknowledgements
Funding was provided by Ministry of Higher Education and Scientific Research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bejaoui, O., Zayani, L. & Chehimi, D.B.H. Constant rate thermal analysis of cobaltoblödite and cupper–kröhnkite at low water vapor pressure 5 hPa. J Therm Anal Calorim 137, 25–31 (2019). https://doi.org/10.1007/s10973-018-7921-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-018-7921-4